ABSTRACT
Radiation-induced cutaneous damage is a common sequela of radiation exposure, affecting up to 95 percent of patients who receive radiotherapy.[1][2][3] Most cases of radiation cutaneous damage are acute or chronic, based on the timing of presentation.
Assessment Summary:
See also 'Algorithm for Assessment of Radiation-Induced Cutaneous Damage' below.
History: A thorough history should be performed to document relevant risk factors. Cancer and radiation therapy history is especially relevant, and efforts should be made to document the treatment regimen including radiation dosage, type, delivery, and any use of chemotherapy.[4][5] Acute effects: occurs within 90 days of exposure; skin damage follows a progressive, dose-dependent course that begins hours to days after exposure. Effects peak then resolve unless severe. Late effects (also known as chronic reactions) presents at least 90 days after exposure. Chronic cutaneous damage is progressive and irreversible, with clinical manifestations more variable than the acute variety that appear months or years after exposure [6] and after doses of at least 45 Gy.[7]
Physical Examination: Acute damage manifestations range from transient erythema, to dry and moist desquamation, to eventual ulceration and necrosis [1][8][4][9][10][11][12] Chronic damage manifests as fibrosis, telangiectasias, pigmentation changes, ulceration, and necrosis.[8][4][5][13]
Diagnosis: Diagnosis is clinical. Physical exam findings consistent with acute or chronic radiation-induced cutaneous damage in the setting of radiotherapy is diagnostic. Histopathology can be used to confirm or rule out any potential mimics. However, care should be taken when biopsying wounds manifested as late effects of radiation because of delayed healing and the risk of worsening the clinical course.[5][14][15][16]
Differential diagnosis: Common differentials for acute effects include: contact dermatitis from bandages or adhesive on the irradiated skin, acute burns (sun, heat, cold, etc). For late effects, secondary malignancy should always be considered when evaluating non-healing wounds that have been previously irradiated. [17][18][7] Other, rarer presentations can resemble cutaneous radiation damage and should be considered when evaluating these patients
Grading systems: For acute effects, clinicians might opt for the CTCAE 4.03 or the RTOG/EORTC scale grades. For late effects, clinicians might opt for the LENT/SOMA or RTOG/EORTC scales. [19]
Ulcer healability: For radiation ulcers, classification according to potential to heal with conservative management alone is helpful in determining an adequate treatment plan. Ulcers that can be healed with conservative management only are categorized as "healable", ulcers that are likely not going to heal with conservative management only are "non-healable" and if co-existing medical conditions, drugs or circumstances will likely impede wound healing, ulcers are classified as "maintenance".[20]
Documentation: At minimum, assessment should be recorded weekly and include location of the lesions, laterality, date of onset, source of radiation, description of cutaneous changes and severity of lesions (e.g, area and depth limited to breakdown of skin, with fat layer exposed, with necrosis of muscle, with necrosis of bone), associated conditions, and treatment plan. For ulcers, medicare requires documentation of signs of improvement such as decrease in ulcer size/depth, exudate, inflammation, swelling, pain and necrotic tissue.
Treatment: see "Radiation-Induced Cutaneous Damage - Treatment, Prevention and Patient Education"
Prevention: see "Radiation-Induced Cutaneous Damage - Treatment, Prevention and Patient Education"
Patient Education: see "Patient Education - Radiation-Induced Cutaneous Damage - Acute Effects" and "Patient Education - Radiation-Induced Cutaneous Damage - Late Effects"
When to refer to specialists
- Nutritionist at initial evaluation
- Respective specialists if associated infectious, metabolic, hematologic, autoimmune, oncologic diseases are suspected
- Plastic surgeon for surgical wound coverage procedures
ICD-10 Coding
Algorithm for Assessment of Radiation-Induced Cutaneous Damage |
INTRODUCTION
Background
Radiation-induced cutaneous damage is a term used to characterize a variety of injuries caused by ionizing radiation. The damage can range from transient erythema and itching to chronic, nonhealing ulcers and exposure of the underlying tissues. These reactions are often painful and can persist beyond the duration of the treatment, affecting quality of life. If the damage is sufficiently severe, it may necessitate cessation of treatment, affecting radiation therapy outcomes. Advancements in delivery such as fractionation and guided methods of delivery have been developed to reduce damage to healthy tissue, but acute and late reactions due to radiation still comprise a significant burden.
Other terms for this condition include: radiation damage, radiation injury, cutaneous radiation injury, radiodermatitis, radiation skin injury, and radiation-induced skin injury.
Classification
Radiation-induced cutaneous damage can be classified as acute, consequential-late, chronic (also known as late), or other less common forms of radiation damage.[3]
-
Acute effects: present within 90 days of exposure and range from mild erythema and hyperpigmentation to severe desquamation and ulceration. Acute damage can also be caused by radiation burns and fluoroscopy.[21][22]
- Late effects (i.e. chronic reactions): present after more than 90 days after exposure. It is both progressive and irreversible - unlike acute damage, which typically peaks and then resolves unless severe.[1][3][23] Ulceration is a common finding in chronic damage, in addition to fibrosis, telangiectasia, lymphedema, and necrosis.[5]
- “Consequential-late”: If severe acute toxicity does not heal, it can result in clinical manifestations similar to those of chronic reactions such as fibrosis and ulceration, but are mechanistically similar to the acute process. This “consequential-late” damage is most common in the gastrointestinal and bladder mucosa, as well as the epidermis, which are particularly sensitive to disruption of the epithelium.[24] Consequential-late cutaneous non-healing ulcers may undergo malignant transformation. [1]
-
Less common forms of radiation damage include: eosinophilic, polymorphic, and pruritic eruption (EPPER), as well as radiation recall reaction. [25][26]
Epidemiology
Incidence
As radiation therapy has become a mainstay of cancer treatment, the incidence of radiation-induced cutaneous damage has increased dramatically:
- According to the NIH Cancer institute, the number of new cancer cases diagnosed per year is projected to reach approximately 1.8 million in 2018, up from 1.7 million in 2017. [27]
- It is estimated that about 70% of new cancer patients receive radiotherapy.[28][29] In 2017, over 1.2 million new cancer patients received radiotherapy as part of their treatment.[29][30]
- Between 85 to 95 percent of patients receiving radiation treatment will experience cutaneous radiation reactions such as redness, itching, and skin breakdown.[1][2][3]
-
- Moist desquamation from acute radiation reaction occurs in 20-25 percent of patients.[31]
- Late radiation damage is estimated to occur in 5-15 percent of cases with radiation exposure.[30]
- Fluoroscopic damage is much rarer, with a reported rate of 0.42 percent per patient risk.[21] Fluoroscopy, like radiotherapy, involves bombarding tissue with radiation, albeit with X-rays at much lower energy levels (kilo-volts compared to mega-volts). However, prolonged exposure can still cause similar skin toxicity that can present acutely or chronically.[22]
Risk factors
Risk factors for radiation-induced cutaneous damage can be intrinsic or extrinsic:
Intrinsic factors
Intrinsic factors that affect incidence of acute reactions:
- Area receiving radiation: areas of intertriginous skin such as the neck, axilla, inframammary folds, and the groin, have the highest risk of developing radiation-induced cutaneous damage due to the bolus effect, which allows for greater radiation to affect folds.[32]
- Larger breast size (bra cup size = D) confers a higher risk of acute radiodermatitis.[33]
- Skin tone has also been found to be a risk factor. Darker skinned patients were found to have much higher rates of post-radiotherapy skin reactions than did fair skinned of the same age and undergoing the same treatment regimen: 20 percent vs 8 percent.[34]
- Immunocompromised status secondary to HIV infection increases the risk of infection and mucosal injury.[35]
- Patients who have undergone breast reconstruction with implants prior to radiation treatment have elevated risk of acute radiation damage due to the inability of tissue with implants to effectively radiate heat.[36][37]
- Skin colonization by Staphylococcus aureus has been linked with increased risk of acute severe radiation dermatitis.[38]
Intrinsic factors that affect incidence of acute and late reactions:
- Connective tissue disorders such as lupus and scleroderma have traditionally been cited as risk factors for radiation damage, but currently available data from case series and a few retrospective studies are still insufficient to contraindicate radiotherapy in patients with those disorders. Nevertheless, a cautious approach and use of modern radiotherapy techniques is reasonable.[7][39][40]
- Genetic conditions that compromise DNA integrity and regenerability such as ataxia telangiectasia, xeroderma pigmentosum, Gorlin syndrome, Bloom syndrome, and fanconi anemia have all been linked to increased risk of radiation injury as well.[1][12][5][7] Single nucleotide polymorphisms in the ABCA1 and IL12RB2 genes were found to increase the risk of radiation dermatitis.[41]
- Other factors such as advanced age, female sex, obesity, smoking, sun exposure, comorbid disease such as diabetes mellitus, and skin disorders (such as acne, psoriasis, or atopic eczema) all predispose patients toward developing radiation-induced cutaneous damage.[1][12][5][7]
Extrinsic factors
extrinsic factors that affect incidence of both acute and late effects:
-
Radiation particle: the extent of damage depends on the radiation particle. Alpha and beta particles confer almost zero risk, whereas gamma, x-ray, and neutron beams confer increasing risk and are associated with radiation-induced cutaneous damage.[42]
-
Concurrent use of radiotherapy with chemotherapeutic agents such as actinomycin D, bleomycin, doxorubicin, 5-fluorouracil, methotrexate, as well as with biological agents such as epidermal growth factor receptor inhibitors have been correlated with increased risk of radiation dermatitis.[3][43] Use of paclitexel or docetaxel in conjunction with radiotherapy for treatment of breast cancer breast usage has been shown to increase the risk of skin damage.[44] Tamoxifen use during radiotherapy is believed to increase rates of fibrosis.[45] Concurrent chemotherapy with radiotherapy in breast cancer had higher rates of breast atrophy, fibrosis, hyperpigmentation, and telangiectasia than did chemotherapy followed by radiotherapy.[46]
-
Total radiation dose received: Increased total dose administered and greater body surface area increase the risk of skin damage.[4][8][5]. Tables 1 and 2 below illustrate expected reactions according to total radiation dose received.
Table 1. Total radiation dose received and expected acute radiation-induced effects.[1][8][4][9][10][11][12]
|
Onset dosage (Gy)
|
Observed skin findings
|
Definition
|
Pathogenesis
|
Time onset of findings
|
|
2
|
Transient erythema
|
Reddened skin that may be edematous, resolves within a few days
|
Histamine release secondary to keratinocyte death
|
Hours
|
|
6-10
|
Faint erythema and depilation
|
More defined redness, hair loss
|
Capillary destruction and RBC extravasation. Hair follicular cell death
|
7-10 days
|
|
12-25
|
Tender erythema, hyperpigmentation, dry desquamation
|
Painful redness; darkening of skin; flaking, pruritic, scaly skin
|
Melanocyte damage; increased mitosis in basal keratinocytes to regenerate damaged epithelium
|
3-4 weeks
|
|
30-40
|
Moist desquamation
|
Epithelial breakdown, serous drainage in areas of high friction
|
Regenerative potential of basal keratinocytes overwhelmed by damage
|
>4 weeks
|
|
>40
|
Ulceration
|
Dermal exposure, bleeding, bullae, fibrinous exudate, possible involvement of underlying structures
|
Destruction of epidermis
|
>4 weeks |
Table 2. Total radiation dose received and expected late radiation-induced effects [8][4][5][13]
|
Onset dosage (Gy)
|
Observed skin findings
|
Definition
|
Pathogenesis
|
Time onset of findings
|
|
>45
|
Delayed ulceration
|
Dermal exposure, bleeding, possible involvement of underlying structures
|
Impaired wound healing from microvascular dysfunction
|
Weeks after exposure
|
|
>45
|
Dermal necrosis/atrophy
|
Frank necrosis of epidermis and underlying dermis
|
Tissue destruction and impaired wound healing from microvascular dysfunction
|
Months after exposure
|
|
>45
|
Fibrosis
|
Hardening and sclerosis of affected tissue
|
Chronic inflammatory state, TGF-B1 upregulation, increased collagen and fibrin deposition
|
Months to years after exposure
|
|
>45
|
Telangiectasia
|
Dilated superficial cutaneous blood vessels
|
Endothelial and microvascular disruption
|
Months to years after exposure |
Etiology
Radiation-induced cutaneous damage is most commonly secondary to radiotherapy during cancer treatment. Less common causes include occupational exposure or accidental exposure such as during military conflict. [47]
Pathophysiology
Radiation induces DNA damage, which disrupts the existing tissue and compromises regenerative ability. The three most significant effects of radiation are: cellular depletion, disruption of the extracellular matrix, and induction of local hypoxia via disruption of the microvasculature.[48]
An overview of the pathophysiology involved in acute and late cutaneous damage is listed below. Figure 1 illustrates acute skin changes during a radiotherapy cycle. For details on the pathogenesis of each of the different types of cutaneous manifestation, see Tables 1 and 2 above.
Acute effects
- The first exposure to radiation causes direct tissue damage, generation of free radicals, ionization of cellular water, DNA damage in both the nucleus and mitochondria.[13][48] These effects are most significant in rapidly proliferating cells, such as those in the stratum basale, melanocytes, and hair follicular cells.[7][49]. This damage induces proinflammatory cytokines including TNF-?, IFN-?, IL-1, and IL-6, which potentiate the inflammatory response and decrease collagen deposition. [50]
- Reactions will continue to persist and worsen until they peak 7-10 days after the exposure as the irradiated basal cells migrate to the surface of the epidermis and are shed. Reepithelialization will commence 10-14 days after completion of treatment. Wounds usually heal within 4-6 weeks, but hyperpigmentation may persist.[4][9]
- Subsequent exposures compound the damage, preventing the tissue from recovering, leading to the development of acute radiation injuries. [51]
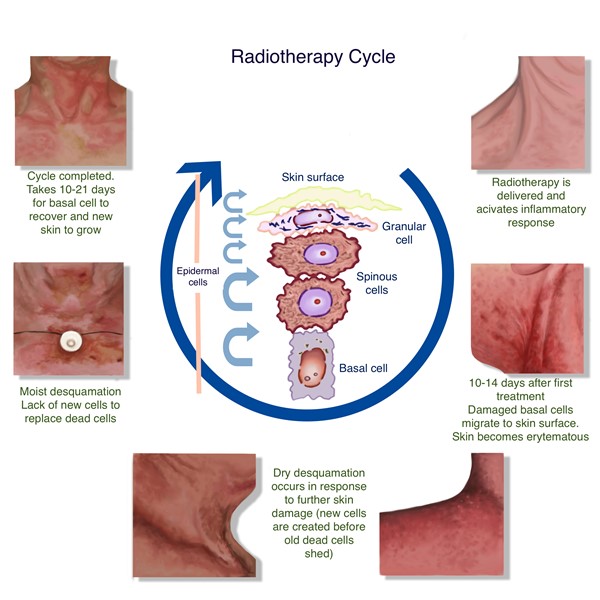
Figure 1. Acute skin changes during radiotherapy cycle. [9]
Late effects
- In a large animal model, radiation-induced damage to dermal stem cells has been found to upregulate apoptotic signals, which has been proposed to be responsible for long term damage and delayed wound healing.[52]
- Fibroblasts, which are key mediators of wound healing, demonstrate decreased replicative ability and poorer growth in the setting of radiation.[53]
- Longstanding inflammation results in TGF-ß1 upregulation, which increases fibrosis deposition, increases collagen expression, and decreases fibroblastic activity, all of which are believed to play a role in chronic radiation damage.[50][54]
- Radiation also increases microvascular permeability and disruption of the microvessels, leading to edema and alterations of the extracellular environment, which can negatively affect wound healing.[55][56]
ASSESSMENT
See algorithm for assessment of radiation-induced cutaneous damage below (Algorithm 1)

Algorithm 1. Algorithm for Assessment of Radiation-Induced Cutaneous Damage
History
An accurate and pertinent history is essential for any patient assessment. Inquiring about a history of radiation exposure is essential as it will shape decision making.
Chief Complaint and History of Previous Illness
- Age, gender, race: advanced age, female sex [7], and dark complexion increase risk [34]
- Location: confinement of lesion to irradiated area can be helpful in diagnosing radiation damage. Intertriginous areas, moist mucosa are at higher risk [32]
- Onset of lesion and duration of symptoms: differentiate between acute and late injuries via time of onset. Acute reactions present within 90 days of radiation[7], chronic or late reactions appear 90 days after exposure, consequential late injury are acute injuries (e.g., ulcer and fibrosis) that do not heal and have potential for malignant transformation [24]
- Pain: assess presence of local pain and level
- History of radiation exposure and type or radiation:
- Radiotherapy, accidental, fluoroscopy,or occupational.[21][22] If radiotherapy, identify prior illness and radiation parameters such as dosage, duration of treatment or exposure, fractionation, technique used and volume of tissue exposed [4][5]
- History of sun exposure [12]
- Treatments so far: History of prescribed and self-prescribed local and systemic medications.
Signs and symptoms of acute radiation syndrome: - Nausea, vomiting, anorexia, cramps, diarrhea, loss of consciousness, burning sensation within minutes to hours of exposure.[47]
- This is an emergency and requires prompt admission and monitoring
|
Medications/Surgery
- Chemotherapeutic agents that increase risk for radiation damage: actinomycin D, bleomycin, doxorubicin, 5-fluorouracil, methotrexate, and epidermal growth factor receptor inhibitors [43], tamoxifen [45]
- Timing of chemotherapeutic use and symptoms to determine if the patient is experiencing radiation recall
- Steroids: usage can delay healing [57]
- Surgeries in the same area that can affect wound healing
- Other oncological procedures such as biopsies or treatments for other cancers that could affect the damaged skin
Social History
- Tobacco use: usage can delay healing [12]
Review of Systems
- General: poor nutritional status, immunosuppression from HIV can delay healing [35],
- HEENT: oral mucosal involvement, xerostomia [14]
- Endocrine: obesity, diabetes can delay healing [7],
- Others: genetic disorders such as xeroderma pigmentosum [7], any skin conditions, breast reconstruction with implants [36][37]
Patient's and caregiver's concerns and psychosocial aspects
- Concerns and psychosocial aspects should be assessed and taken in consideration when creating a treatment plan:
- Evaluate patient's concerns: dryness, burning feelings, irritation, roughness, and hyperpigmentation are frequently reported symptoms of acute radiation dermatitis.[58] Pain level should be evaluated.
- ? Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"
- Evaluate psychosocial aspects of the patient, caregiver and family: cognitive, functional, emotional status, understanding of the condition and risk factors, preference for treatment method, motivation for adherence to the care plan, financial concerns
Physical Examination
A thorough exam is always preferred, but radiation injuries are typically discrete and confined to previously irradiated areas. These reactions are often painful and will be noticed by the patient, barring a loss of sensation or other injury that inhibits nociception. However, in the insensate or otherwise compromised patient, it is prudent to examine the whole body, paying particular attention to areas that have received radiation. It is important to note and document the size, depth, quality, and color of the wound, as this information will be used to gauge progress.
Local exam (skin changes/ulcer)
- Location: Skin folds and moist, intertriginous areas such as the axilla, inframammary folds, and groin are the most common locations.[32]
- Assess for acute cutaneous changes: Typical signs include erythema, hair loss, dry or wet desquamation, ulceration.[8][4] Skin damage follows a progressive, dose-dependent course that begins hours to days after exposure. Findings are summarized in Table 1 and also listed below:
-
- Low doses (<2 Gy) cause mild damage to the epithelium, resulting in erythema that typically resolves within a few days.
- Continued exposure/treatment (6-10 Gy) causes dryness, epilation, and more persistent erythema, that manifests within the second week of treatment.
- Doses of 12-25 Gy cause defined, tender erythema in the irradiated areas and pruritic, dry desquamation as the basal keratinocytes increase mitotic rate to compensate for the damage. These changes occur 3-4 weeks after initiation of treatment. Continued treatment of a month of more will manifest the most severe damage.
- High doses of 30-40 Gy cause disruption of the skin integrity, resulting in moist desquamation as the regenerative capability of the stratum basale is overwhelmed.
- Doses beyond 40 Gy cause ulceration, necrosis, and bleeding. The dermis will be exposed, and there may be fibrinous exudates and bullae present.[1][2][4][9][10][11][12] The damage will continue to persist and worsen until they peak 7-10 days after the exposure as the irradiated basal cells migrate to the surface of the epidermis and are shed. Reepithelialization will commence 10-14 days after completion of treatment. Wounds heal within 4-6 weeks, but hyperpigmentation may persist.[4][9]
- Assess for consequential late injury: acute injuries that did not heal resulting in ulcers or necrosis. These patients may also have gut and/or bladder mucosal involvement [1][24]
- Assess for chronic cutaneous changes (i.e., late effects): typical signs include delayed ulceration or necrosis (Figures 2 and 3), telangiectasia, fibrosis, lymphedema. [1][8][4] Chronic cutaneous injuries are more variable than the acute variety, manifesting months or years after exposure [6] and after doses of 45 Gy or more.[7]
-
- Hypo- or hyperpigmentation are frequently present due to melanocyte damage.
- Skin atrophy is common and loss of dermal appendages such as sebaceous glands and hair follicles may occur.
- Hyperkeratosis may be present, as well as telangiectasia and fibrosis.[3][5][7]
- Fibrosis can cause cutaneous retraction, pain, and increase the risk of ulceration and necrosis.[8][4][24]
- Assess for radiation recall: new symptoms of maculopapular, swelling, pruritus, desquamation, and ulceration in a previously irradiated patient undergoing chemotherapy is likely radiation recall.[1][26]
- Assess for Eosinophilic, Polymorphic, and Pruritic Eruption (EPPER): a reaction to radiation therapy that presents with erythema, papules, pruritus, wheals, or bullae. These findings may extend beyond the irradiated areas and are most commonly associated with breast and cervical cancer. This condition can emerge at any time after exposure to radiotherapy.[25]
- Assess for adequate blood supply: Radiation can frequently compromise tissue vasculature and hamper delivery of blood and nutrients needed for wound healing.
- If radiation damage is an non-healing ulcer on lower extremity, a non-invasive arterial testing modality is recommended to rule out combined peripheral arterial disease (PAD). Ankle brachial index (ABI), toe brachial index (TBI), handheld Doppler waveform analysis may be used. An ABI<0.8 indicates PAD. [59]
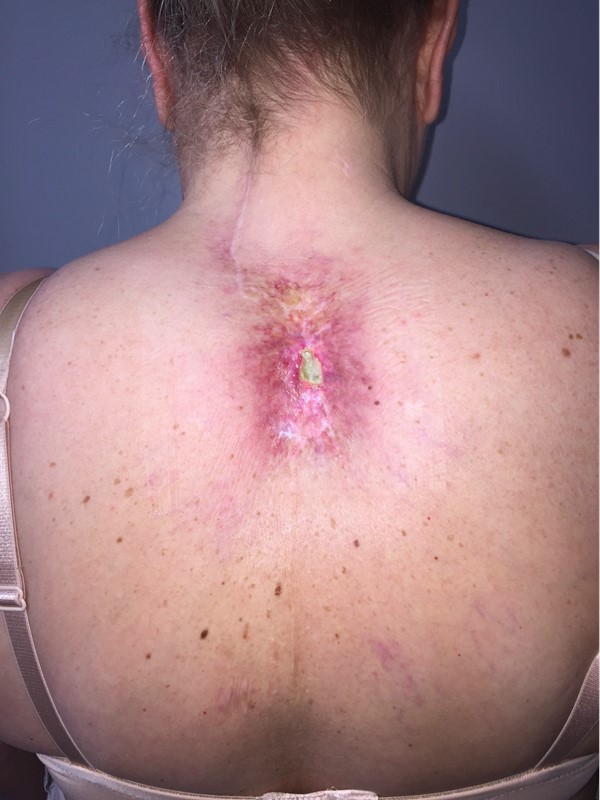
Figure 2: Chronic radiation-induced ulceration and fibrotic tissue of the upper back in a 54 year old woman, who received radiotherapy for sarcoma. This image was taken 9 years after completion of radiotherapy. The ulcer developed after a nearby cervical spine surgical procedure (see posterior surgical scar).
| 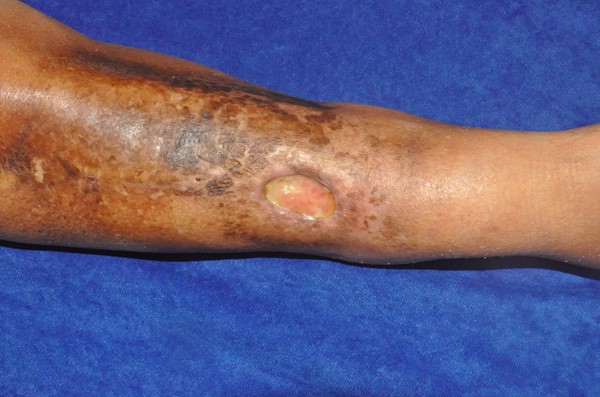 Figure 3: Chronic radiation-induced ulceration and hyperpigmentation of the leg in a 65 year old woman, who received radiotherapy for sarcoma. This image was taken several years after completion of radiotherapy Figure 3: Chronic radiation-induced ulceration and hyperpigmentation of the leg in a 65 year old woman, who received radiotherapy for sarcoma. This image was taken several years after completion of radiotherapy
|
Diagnosis
Acute effects
- A diagnosis of acute radiation-induced cutaneous damage can be done clinically in a patient with a history of radiation exposure and aforementioned skin findings (erythema, hair loss, pain, dry/wet desquamation, ulceration).[5][14]
- If the diagnosis is uncertain, histopathology can be employed for a definitive diagnosis. Histopathologic findings of acute radiation injuries feature keratinocyte apoptosis, vacuolization of the stratum basale, and edema.[15][16]
Late effects
- Chronic cutaneous injuries can be more challenging to diagnose due to their ability to manifest months or years after radiotherapy. The findings of dermal atrophy, pigmentation changes, hyperkeratosis, telangiectasia, fibrosis, and delayed ulceration in a patient with a history of radiation exposure are highly suggestive of a chronic radiation-induced cutaneous damage.([8][12]
- As with acute injuries, histopathology will provide an unambiguous diagnosis. Chronic wounds feature dermal sclerosis, epidermal hyperkeratosis, and dilated dermal vessels.[15] However, care should be taken when biopsying these wounds because of delayed healing and the risk of worsening the clinical course.[5]
Less common forms of radiation damage
- Eosinophilic Polymorphic and Pruritic Eruption Associated with Radiotherapy (EPPER): a pruritic, papular rash that may include vesicles or subdermal bullae that is not be confined to the irradiated fields, which can help distinguish it from other radiation reactions.
- Diagnosis can be confirmed histologically as well. Biopsy of the rash will show lymphocytic and histiocytic perivascular infiltrate with substantial eosinophils. In half of cases, immunofluorescence of the dermis will show perivascular IgM and C3, which can also help distinguish EPPER from bullous pemphigoid. [60]
Other exams that may be used to obtain “objective measures” of radiation-induced cutaneous damage include quantitative ultrasound, doppler perfusion imaging, reflectance colorimetry, and spectrophotometry.[8][61]
Differential Diagnosis
A diagnosis of radiation injury is usually made simply by the history and clinical presentation. However, there are several mimickers that are included in the differential diagnoses.
Acute effects
- Contact dermatitis from bandages or adhesive on the irradiated skin (Figure 4), which can range from erythema to necrosis. [62]
- Radiation port dermatophytosis: Tinea infection of the radiated site causes erythematous, expanding, pruritic lesions, which can mimic acute radiation injury. Diagnosis is confirmed with potassium hydroxide preparation or culture. [63]
- Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis (Figure 5): These syndromes mimic more severe presentations of acute radiodermatitis and feature bullae, skin sloughing, and can be diagnosed with biopsy showing complete epidermal necrosis. [64]
- Graft vs. Host Disease (GVHD): Acute onset GVHD presents with an erythematous, blanchable maculopapular, rash that often originates on the neck and hands, evolving to incorporate the torso and becoming mobilliform as it progresses.[65]
- Radiation recall injury (RRI): Six to 9 percent of patients who have had prior radiation treatment and begin chemotherapy may experience RRI, which resembles acute radiation injury. Patients have a mild to moderate pruritic, erythematous, maculopapular rash that occurs on areas that have been previously irradiated. The most common culprit medications are doxorubicin, taxanes such as paclitaxel and docetaxel, and gemcitabine.[26][66]
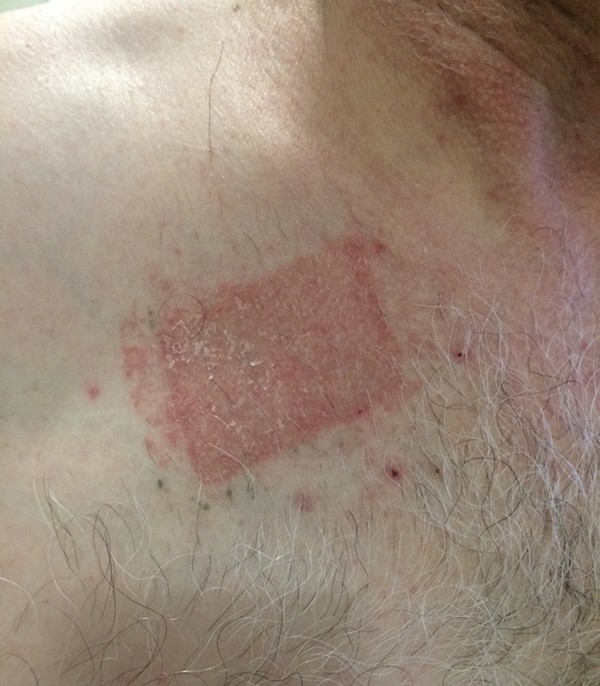
Figure 4: Contact dermatitis[67] | 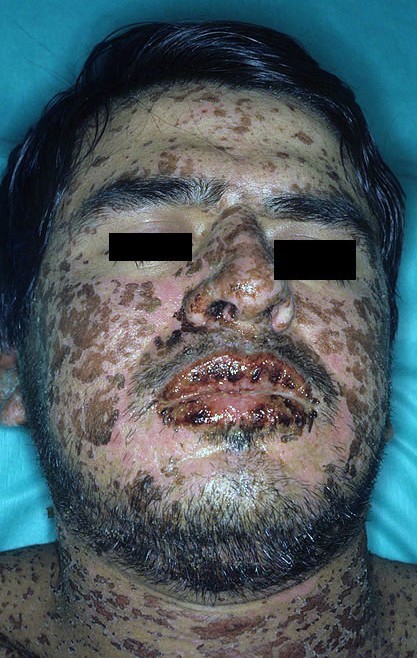
Figure 5. Steven-Johnson Syndrome [68] |
Late effects
- Malignancy: Radiotherapy increases the risk of secondary malignancies such as basal cell carcinoma [69], squamous cell carcinoma (Figure 6) and keratoacanthoma [12], which can mimic chronic cutaneous injury. Secondary malignancy must always be considered in the differential of a patient with history of cancer and radiation exposure. [17][18][7]
- Marjolin ulcer (Figure 7): A form of squamous cell carcinoma that arises in areas of chronic wounds or scars that features everted wound margins and ulceration. A biopsy is required for diagnosis. [70]
- Diabetic foot ulcer (Figure 8): Diabetic patients may suffer from diabetic ulcers, typically on the lower extremities. These wounds are chronic, non-healing, foul-smelling, and can be painful or insensate.[71] See topic "Diabetic Foot Ulcer - Introduction and Assessment"
- Radiation induced morphea: Radiation induced morphea, also called postirradiation morphea occurs months to years after treatment and features painful, disfiguring sclerotic plaques in the previously irradiated tissue, but can expand to involve non-irradiated tissue. [72]
- Nephrogenic Systemic Fibrosis (NSF): NSF, also known as nephrogenic fibrosis dermopathy, is an idiopathic condition that features widespread fibrosis of the cutaneous tissue and organs. [73]
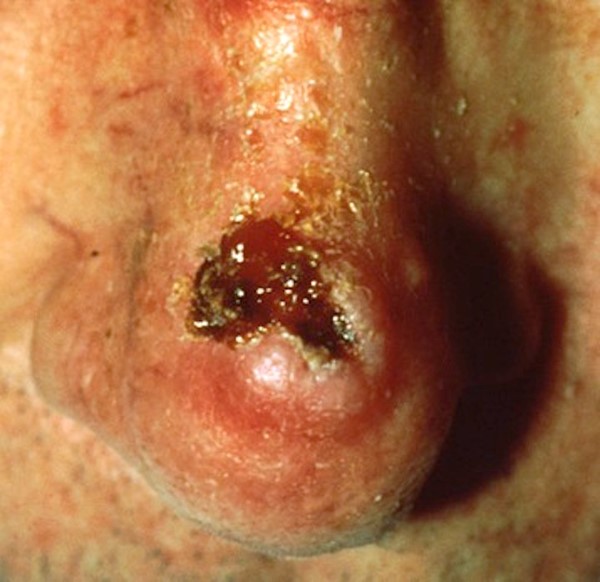
Figure 6. Squamous Cell Carcinoma [74] | 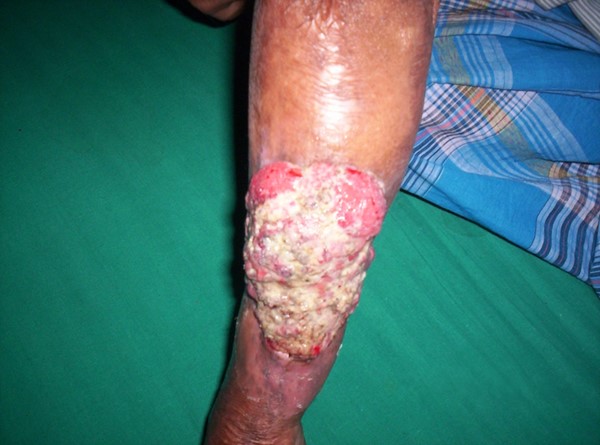
Figure 7. Marjolin ulcer[75] | 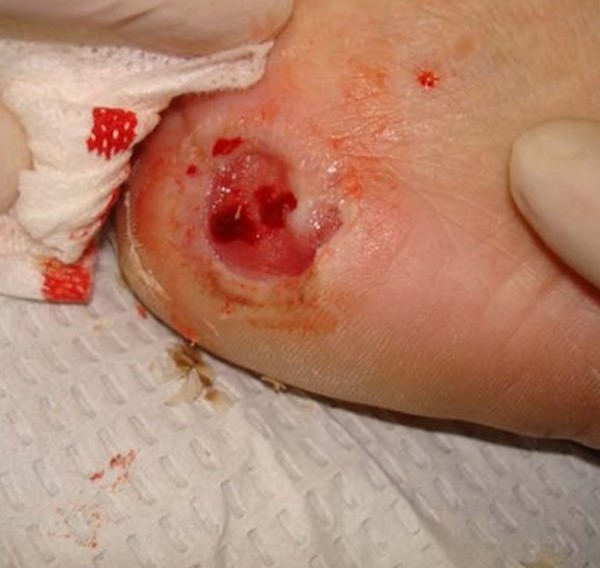
Figure 8. Diabetic foot ulcer [76] |
Grading systems
An accurate and objective means of grading radiation-induced cutaneous damage is crucial for proper management and communication among providers. Several systems exist, and currently there is no consensus on which method is superior [4], as reliability and validation data are sparse.[19]
The three most common grading criteria are (Table 3):
- The National Cancer Institute’s Common Technology Criteria for Adverse Events (CTCAE) version 4.03 [77],
- The Radiation Therapy Oncology Group (RTOG)/European Organization for Research and Treatment of Cancer (EORTC) scale [78],
- The Late Effects Normal Tissue Task Force/Subjective, Objective, Measurement, and Analytic (LENT/SOMA) scale for the classification of chronic dermatitis [79].
Table 3. Comparison among the most common radiation-induced cutaneous damage grading systems
|
Grading system
|
CTCAE 4.03
|
RTOG/EORTC
|
LENT/SOMA
|
|
Common application
|
Acute effects
|
Acute and late effects
|
Late effects
|
|
Elements assessed
|
Skin and subcutaneous tissue changes
|
Skin and subcutaneous tissue changes
|
Skin and subcutaneous tissue changes
Pain
|
|
Comments
|
Acute radiation injury assessed on a scale from 0 to 4, with increments of 1. Large increments of 1 unit often fail to capture subtle yet important skin changes [8][61]
|
Acute radiation injury assessed on a scale from 0 to 4, with increments of 1. Large increments of 1 unit often fail to capture subtle yet important skin changes [8][61]
|
Considered more precise than the RTOG/EORTC for the evaluation of late skin and subcutaneous toxicity . [80] 125I seed brachytherapy for parotid gland cancer. | Mao MH, Zhang J, Zhang JG et al. | 2017" target="_blank">[81]
|
Selecting grading systems
- For acute effects, clinicians might opt for the CTCAE 4.03 or the RTOG/EORTC scale grades. The CTCAE 4.03 seems to be the most frequently utilized scoring system in clinical practice for grading acute effects. [19] These two systems grade acute effects findings in a scale of 0 to 4 in increments of 1 unit, which may fail to capture subtle skin changes. To address this concern, newer grading scales with smaller increments (i.e., 0.5 unit) were developed, such as the Oncology Nursing Society (ONS) and Radiation Dermatitis Severity (RDS) scales.[4][8][61]. Also, Zenda et al. attempted to incorporate more objectivity in the CTCAE 4.03 scale by creation of a photo atlas documenting each grade. [82]
- For late effects, clinicians might opt for the LENT/SOMA or RTOG/EORTC scales. The LENT/SOMA scale is recommended for use in clinical trials, and is less practical for use in clinical practice.[19] A comparison of the LENT/SOMA and RTOG/EORTC scales in grading pigmentation, fibrosis, and telangiectasia found the LENT/SOMA scale to be more precise, but noted that both scales to misjudge wounds.[80] A subsequent comparison of the two scales in assessing late skin toxicity after radiotherapy for parotid cancer found the LENT/SOMA scale to be more representative of patients’ conditions, but noted that alterations of the criteria are necessary to improve precision.125I seed brachytherapy for parotid gland cancer. | Mao MH, Zhang J, Zhang JG et al. | 2017" target="_blank">[81]
We have created tables for both acute (Table 3) and chronic (Table 4) damage that combine the relevant scales, so as to facilitate grading of radiation-induced cutaneous damage:
Table 3: CTCAE 4.03 and RTOG/EORTC grading scales for acute radiation-induced cutaneous and subcutaneous damage [19][77][78]
CTCAE 4.03
| RTOG/EORTC
| Description |
| 0 | No symptoms or deviation from baseline
| No symptoms or deviation from baseline
|
| 1 | Light erythema or dry desquamation
| Erythema, dry desquamation, hypohydrosis, epilation
|
| 2 | Moderate to frank erythema, moist desquamation confined to intertriginous areas, edema
| Tender, frank erythema, patches of moist desquamation, edema
|
| 3 | Moist desquamation beyond intertriginous areas, bleeding caused by abrasions
| Confluent moist desquamation beyond intertriginous areas, pitting edema
|
| 4 | Life threatening injuries, full dermal thickness necrosis and/or ulceration, spontaneous bleeding; skin graft indicated
| Necrosis, ulceration, hemorrhage. Necrosis, ulceration are called consequential-late lesions if they fail to heal on their own
|
| 5 | Death
| Death associated with treatment |
Table 4: LENT/SOMA chronic radiation-induced cutaneous and subcutaneous damage grading scale [19][79][80]125I seed brachytherapy for parotid gland cancer. | Mao MH, Zhang J, Zhang JG et al. | 2017" target="_blank" style="background-color: rgb(255, 255, 255); font-size: 1em;">[81]
| LENT/SOMA | Grade 1 | Grade 2 | Grade 3 | Grade 4 |
Subjective
|
|
|
|
|
| Sensation | Occasional hypersensitivity, pruritus
| Intermittent, tolerable pain
| Constant and intense pain
| Refractory and debilitating pain
|
| Scaliness/ roughness | Present but asymptomatic
| Symptomatic
| Requires intervention
|
|
Objective
|
|
|
|
|
Telangiectasia
| <1 cm2
| 1-4 cm2
| >4 cm2
|
|
| Fibrosis | Palpable | Noticeably increased density and firmness, symptomatic
| Profoundly increased density, fixation, and rectraction, secondary dysfunction
| Total dysfunction
|
| Edema | Asymptomatic | Symptomatic | Symptomatic, secondary dysfunction
| Total dysfunction
|
Contraction and atrophy
| Present and asymptomatic
| Symptomatic (<10%)
| Secondary dysfunction (10-30%)
| Total dysfunction (=30%)
|
Pigmentation change
| Slight and transient
| Frank and permanent
|
|
|
| Ulceration | Epidermis only
| Dermis included
| Subcutaneous tissue included
| Exposed bone |
Alopecia (scalp)
| Slight
| Permanent patches
| Total and permanent
|
|
Ulcer Healability
- In case the radiation-induced cutaneous damage is a wound/ulcer, assess healability and prognosis:
- Determine wound prognosis: determine if wound is healable or non-healable with conservative treatment, or if co-existing medical conditions/drugs or circumstances will significantly hamper wound healing (“maintenance”) [20][83] See Table 5.
Table 5. Wound healability and prognosis. (* ) As determined by comprehensive patient assessment. Maintenance wounds have potential to heal, but patient or health system barriers compromise healing (**) For persons without diabetes, inadequate blood supply is objectively confirmed by ankle-brachial index (ABI) < 0.5, monophasic doppler waveform, skin perfusion pressure < 30mmHg, transcutaneous oxygen < 30mmHg, absolute systolic ankle pressure < 50 mmHg OR toe pressure < 30mmHg. For persons with diabetes, perform any other testing listed above besides ABI as ABI can be falsely elevated
| Can underlying cause be treated? | How is blood supply to the wound? | Co-existing medical conditions/drugs... | Then wound prognosis is...
|
| Yes | Adequate | Are not an obstacle for healing | Healable
|
| No (*) | Adequate | May or may not prevent healing | Maintenance
|
| No | Usually inadequate (**) | May inhibit healing | Non-healable |
Modified from Sibbald RG et al. 2011[20][83]
- Healable wounds: may be initially managed with an active wound care approach. Adjunctive therapy is considered if this approach fails
- Non-healable wounds: wounds are considered non-healable when active wound care approach is likely to fail without correction of underlying cause, optimization of blood supply and adequate bioburden control (e.g., wounds with frequent, recurrent infections that may need oral antibiotics in addition to topical antimicrobials).
- Radiation can frequently compromise tissue vasculature and hamper delivery of blood and nutrients needed for wound healing. In these cases, clinicians might opt for referral to plastic surgeon for evaluation of surgical wound coverage. Free flaps or flaps with pedicles originating in non-irradiated areas may represent the best treatment option.
- Maintenance wounds: managed with a “non-healable” wound care protocol until co-existing medical conditions/drugs or circumstances that inhibit healing are mitigated. Goal of wound care in the interim is to prevent further deterioration of wound, control bioburden, manage moisture and provide patient comfort.
- After determination of ulcer healability, see "Radiation-Induced Cutaneous Damage - Treatment, Prevention, Patient Education"
Documentation
When treating patients with radiation-induced cutaneous damage it is important to document the findings and features to plot the clinical course and determine management. The aforementioned grading scales (CTCAE, RTOG/EORTC, LENT/SOMA and others) can be helpful in tracking the progress of the skin damage and in providing a more objective measurement of progress.
Unfortunately, these scales are not frequently used in practice. Oddie et al. surveyed nurses working exclusively in the setting of irradiated wound care and found that 45.5 percent of those surveyed use no objective criteria at all.[84] A lack of consistent, objective documentation can negatively impact outcomes by inserting bias and potentially missing changes in the patient’s condition.
At minimum, assessment should be recorded weekly and include location of the lesions, laterality, date of onset, source of radiation, description of cutaneous changes and severity of lesions (e.g, area and depth limited to breakdown of skin, with fat layer exposed, with necrosis of muscle, with necrosis of bone), associated conditions, and treatment plan.
Medicare and other insurers expect documentation of signs of improvement in order to support medical necessity for many services/interventions/supplies:
- Evidence of improvement includes measurable changes (decreases) of some of the following:
- Drainage
- Inflammation
- Swelling
- Pain
- Wound dimensions (diameter, depth)
- Necrotic tissue/slough
- A wound that shows no improvement after 30 days requires a change to the plan of care, which may include physician reassessment of underlying infection, metabolic, nutritional, or vascular problems inhibiting wound healing, or a new treatment modality. See topic "Radiation-Induced Cutaneous Damage - Treatment, Prevention, Patient Education"
CODING
ICD-10
- L58.0 Acute radiodermatitis
- L58.1 Chronic radiodermatitis
- L58.9 Radiodermatitis, unspecified
- L59.8 Other specified disorders of the skin and subcutaneous tissue related to radiation
- L59.9 Disorder of the skin and subcutaneous tissue related to radiation, unspecified
For chronic ulcers due to radiation (late effect):
1. Identify and document first any documented underlying condition (ICD-10-CM documentation)
- L58.1 Chronic radiodermatitis
- L58.9 Radiodermatitis, unspecified
2. Select code for non-pressure chronic ulcer based on anatomic location (non-billable, need further description of laterality and severity)
- L97.1 Non-pressure chronic ulcer of thigh
- L97.2 Non-pressure chronic ulcer of calf
- L97.3 Non-pressure chronic ulcer of ankle
- L97.4 Non-pressure chronic ulcer of heel and midfoot
- L97.5 Non-pressure chronic ulcer of other part of foot
- L97.8 Non-pressure chronic ulcer of other part of lower leg
- L97.9 Non-pressure chronic ulcer of unspecified part of lower leg
- L98.41 Non-pressure chronic ulcer of buttock
- L98.42 Non-pressure chronic ulcer of back
- L98.49 Non-pressure chronic ulcer of skin of other sites
3. Specify laterality
- Right, left or unspecified
4. Specify ulcer severity:
- Limited to breakdown of skin
- With fat layer exposed
- With necrosis of muscle
- With necrosis of bone
- Unspecified severity