|
Algorithm for Management of Venous Ulcers | First Visit Checklist | Follow up Visit Checklist
|
TREATMENT
Overview
This topic covers management of venous leg ulcers (VLU) including treatment, prevention and a section for clinicians on patient education. For an introduction and assessment of venous leg ulcers (VLU) including epidemiology, risk factors, etiology, pathophysiology, history, physical examination, diagnosis, differential diagnoses, documentation and ICD-10 coding, see "Venous Ulcers - Introduction and Assessment". For clinical guidelines and quality measures specific to VLU, see "Venous Ulcers - Overview". For an overview of surgical interventions for management of chronic venous disease and resulting manifestations such as VLUs, see "Chronic Venous Disease - Surgical Management".
A customized treatment plan is created with patient input, based on a comprehensive assessment that aims to:
- Identify risk factors for VLU, amputation, delayed healing, recurrence
- Differentiate VLUs from other types of lower extremity ulcers, which may require different treatments
- Categorize VLUs as "simple", "complex" or of mixed arterial venous etiology. Categorization helps determine likely prognosis so that appropriate time frames for monitoring, reassessment and specialist referral can be established.[9]
-
- "Simple VLU" can be characterized as a VLU with wound area < 100 cm2 and onset < 6 months, with limited co-morbidities
- For "Complex VLU" and mixed arterial venous etiology, see section 'Characterization of VLUs' in "Venous Ulcers - Introduction and Assessment".
- Determine "healability", that is, the potential of the ulcer to heal with conservative treatment only (i.e., patient adheres to treatment and there are no factors that impede ability to heal). See 'Ulcer healability' in "Venous Ulcers - Introduction and Assessment".
An adequate treatment plan for VLUs should aim to [10]:
- Treat the cause: chronic venous insufficiency and other factors impeding healing
- Assess patient and caregiver concerns
- Provide effective local wound care
If a VLU does not show signs of improvement despite adequate treatment for 4 weeks, the treatment plan should be reassessed, with re-evaluation of differential diagnoses or consideration for adjunctive therapies. [11]
Of note, in all practice settings, it is essential to ensure that the treatment plan and care provided adheres to the specific facility's policies, procedures, protocols, and formulary.
See Algorithm for Management of Venous Ulcers (initial management and management for refractory ulcers) (Algorithm 1)
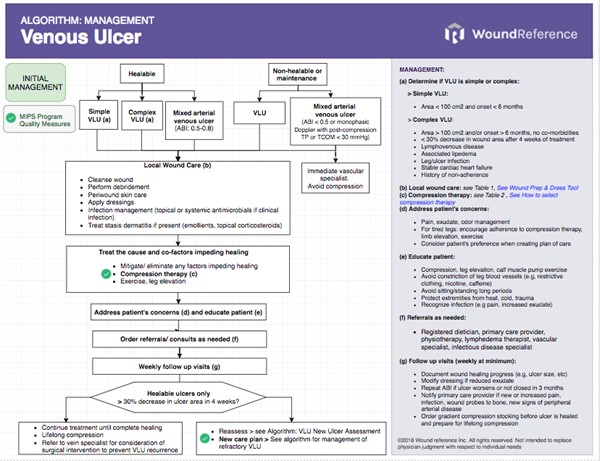
Algorithm 1. Algorithm for Management of Venous Ulcers (click link to enlarge)
Treatment Goals
Healable VLUs
- In general, the goals for healable VLUs are to reduce edema, improve ulcer healing, and prevent recurrence. Patient concerns that may not be initially obvious also need to be taken into account when creating a treatment or care plan.
- Typically, for healable VLUs, at least a 30% decrease in size in the first 4 weeks of treatment with standard care is expected. If a VLU does not reach this target, it has a 68% probability of failing to heal within 24 weeks. [6][12][13]
-
- A healable "simple" VLU (i.e, those with a good prognosis) is expected to be 100% healed within 12 weeks of adequate treatment. At minimum, it should be = 70% healed within 18 weeks. [9]
- A healable "complex" VLU (i.e. those that are likely to take longer to heal) is expected to be 100% healed within 18 weeks. At minimum: = 70% healed within 24 weeks.
- Healable VLUs that fail to reach a 30% decrease in size after 4 weeks of treatment with standard therapy need reassessment and consideration for adjunctive treatment options (See sections 'Plan Reassessment' and 'Adjunctive Treatment'). [2][5][11][14] Many adjunctive therapies are only covered by Medicare and other private insurers if VLUs fail to show evidence of healing with standard therapy for 4 weeks. [15]
Non-healable or maintenance VLU
- For maintenance or non-healable VLUs, goals include prevention of ulcer progression, management of pain, bioburden, exudate, and edema. Patient concerns and comfort should be prioritized when creating a treatment or care plan.
Treat the cause and co-factors impeding healing
For all patients with healable, non-healable or maintenance VLU:
- Address any identified factors that can affect wound healing to promote healing of existing VLUs and to prevent new or recurring ulcers.
- Treat venous hypertension with compression therapy, leg elevation and supervised exercises involving the calf-muscle pump. For non-healable or maintenance VLU, clinicians may opt for implementing these interventions to the extent acceptable by patient and family [11] (e.g., modified, low pressure compression therapy may be used to help manage edema and prevent VLU from getting larger).
- Oral pentoxifylline or micronized purified flavonoid fraction may be administered with compression therapy to promote healing of long-standing or large VLUs. [2][16][17]
For patients with healable VLU:
- Investigate surgically correctable arterial and venous conditions. For patients with venous reflux due to superficial venous incompetence, as demonstrated by duplex ultrasound, consultation with a vein specialist is recommended to consider early surgical treatment of the incompetent superficial veins.[18]
See details and evidence related to these interventions below.
Compression Therapy
Compression therapy plays a crucial role in the treatment of patients with venous leg ulcers and remains the cornerstone of VLU care.[19] Compression therapy associated with standard wound care has the potential to heal approximately 50% - 75% of VLUs. [20][21][22]
- Medicare Quality Payment Program, Quality Measure: "Adequate Compression of VLU at each treatment visit, appropriate to arterial supply"
-
1B
For VLU patients with no contraindications, we recommend compression over no compression therapy to promote wound healing (Grade 1B)
- Rationale: Clinical guidelines recommend compression therapy to treat VLU (level B evidence, moderate certainty).[11][14][22][23][24]Compression is the gold standard for VLU treatment. Compared with people with VLU who do not utilize any compression, people with VLU who utilize compression bandages or stockings are more likely to experience complete wound healing more quickly, have their VLU completely healed, experience less pain and better quality of life.[25] Despite benefits, issues such as patient adherence and clinician skills may interfere with its effectiveness. Many patients cannot tolerate, or do not adhere to, compression bandaging therapy.[26] Also, achieving the desired pressure with bandages can be difficult, as correct application is operator-dependent. As a result, many patients do not receive adequate compression therapy. [27][28][29] Skills can be improved by training professionals [28][30] and adherence can be improved by educating patients on the importance of compression therapy.[31] As for costs, compression bandages and certain gradient compression stockings are covered by Medicare Part B as long as used on patients with active VLUs. Multilayered compression systems with elastic components have been shown to be more cost-effective than usual care alone.[32][33]
Prior to application of compression therapy, presence of other ulcer etiologies and/or co-morbid conditions that affect how compression is applied should be carefully evaluated. Failure to do so can result in adverse effects and deterioration of the patient's condition. Peripheral artery disease (PAD) can be present in up to 25% of VLU patients [3][4] and may be ruled out with evaluation of ankle brachial index ratio (ABI) or audible handheld Doppler ultrasound with continuous waveform analysis. See section on 'Algorithms' in topic "How to Select Adequate Compression Therapy Pressure Levels and Products", and section on 'Noninvasive Arterial Tests' in topic "Arterial Ulcers - Introduction and Assessment".
- For patients with mixed arterial ulcers and severe PAD, compression therapy is not indicated. These patients should be referred to vascular specialist instead.
-
- All VLU patients should be screened for arterial disease using Doppler measurement or ankle-brachial pressure index (ABI) by trained staff before receiving compression therapy.[7] Compression on a limb compromised by arterial disease can lead to ischemic sequelae and tissue necrosis.[2]
- Arterial disease manifested by ABI below 0.8 is often considered clinically significant.
- For patients with VLU who have ABI between 0.5 and 0.7, modified (reduced) compression may be used [34], pending consultation with a vascular specialist.[7][35]
- For patients with ABI at or below 0.5, ankle pressure <60 mmHg or toe pressure <30 mmHg, compression therapy is not indicated.[2][35]
- ABI and other noninvasive arterial testing may be covered by Medicare if specific criteria are met.[36]
Pressure levels
According to the type of VLU (simple, complex or mixed), and based on the presence of co-morbid conditions, compression therapy for VLU can be delivered using "standard" or "modified" pressure.
- Standard compression: 30-40 mmHg resting pressure measured at the ankle level
- Modified compression: low resting pressure, i.e., 20-30 mmHg, measured at the ankle level
-
For examples of different brands and their intended pressure levels, see "Compression Brands Quick Reference". For more information on compression, see "Compression Therapy".
Table 1 below summarizes pressure levels and management of simple, complex and mixed VLU.
Table 1. Pressure levels and management of simple, complex and mixed VLU
VLU: venous leg ulcer, ABI: ankle brachial index, CHF: congestive heart failure, PAD: peripheral arterial disease. Standard: 30-40 mmHg resting pressure measured at the ankle level, Modified: low resting pressure, i.e., 20-30 mmHg, measured at the ankle level
| Condition |
Pressure
|
Comments |
Follow up |
Referrals |
Healable, simple VLU: ABI 0.8-1.3, can be treated at primary care or community setting
|
| Area < 100 cm2 and onset < 6 months |
Standard |
If low adherence, start with lower level compression and increase gradually (e.g., tubular dressing with 10 mmHg in the first week, then 2 layers of tubular dressing in the second week, and 3 layers in the third week) |
48h |
Wound specialist if:
- VLU has not decreased by 30% in 4 weeks despite adequate treatment
- Edema does not improve in 2 weeks
- Any of the conditions below arise
|
Healable, complex VLU: ABI 0.8-1.3, treated at specialized service/clinic that manages VLU
|
|
Area > 100 cm2 and/or wound onset > 6 months (no other co-morbidities)
|
Standard |
- Reassess, revisit differential diagnoses, consider malignancy
- Review compression and wound management
- Assess adherence
- Consider skin grafting if wound > 25cm2 [2]
|
48h |
As needed, according to reassessment findings |
| Wound area has not decreased by 30% in 4 weeks despite adequate treatment |
Standard |
- Same as above
- Consider adjunctive therapies
|
48h |
As needed, according to reassessment findings
|
| Phlebolymphedema (chronic venous insufficiency and lymphatic insufficiency) See topic "Lymphedema - Introduction and Assessment" |
Standard |
- Specialized bandaging techniques may be required if unusual limb shape
- Skin care due to increased risk of infection
|
48h |
Lymphedema therapist |
| Leg/ulcer infection |
Standard
(or
Modified)
|
- Current infection: manage as appropriate, may apply compression after 24 hours of systemic antibiotics and if afebrile. Consider reducing level of compression if difficult to tolerate. Inspect dressing more frequently to monitor infection
- Recurrent: examine wound regularly and mitigate factors that may contribute to recurrence
|
24h
48h
|
If needed, infectious disease specialist |
| History of non-adherence |
Standard
or
Modified
|
- Reassess, revisit differential diagnoses
- Determine reasons for non-adherence and address modifiable reasons
- Start with lower level compression and increase gradually (e.g., tubular dressing with 10 mmHg in the first week, then 2 layers of tubular dressing in the second week, and 3 layers in the third week)
- Consider use of ulcer gradient compression stocking or intermittent pneumatic compression if within indications
|
48h |
|
| Stable CHF |
Modified
|
- Ensure CHF is stable due to risk of pulmonary edema once leg edema starts to clear
- Diuretics can be increased upon application of compression for the first time
- Monitor closely for sigs of CHF instability, such as peripheral edema, dyspnea, altered mental status, etc.
|
24h |
Cardiologist |
Maintenance or non-healable VLU without PAD
|
| Cause not treatable with conservative interventions due to patient risk factors, comorbidities, lifestyle |
-
|
- May apply low pressure levels (e.g, tubular dressing, with 10 mmHg)
|
|
As needed, depending on findings |
Mixed etiology leg ulcers: ABI < 0.8 or > 1.3, treated at specialized service/clinic or in collaboration with specialist who manages VLU
|
| Non-compressible arteries (ABI > 1.3) |
-
|
- May apply tubular dressing (10 mmHg) while waiting for vascular assessment
|
48h |
Vascular specialist |
| Healable mixed arterial ulcer with mild/moderate PAD (ABI 0.5-0.8) |
Modified
|
- May apply modified compression with low resting pressure (high stiffness), with instructions to contact provider if increased pain, changes in limb color/perfusion, frequent assessment and monitoring for ischemia and pressure damage. Short-stretch bandages of up to 30 mmHg does not adversely affect blood flow and seems well tolerated even by older patients.[37]
|
24h |
Vascular specialist |
| Non-healable mixed arterial ulcer with severe PAD (ABI < 0.5) |
- |
- Ulcer will not heal without prior revascularization to increase blood supply to lower limb
|
- |
Vascular specialist, if critical limb ischemia (patient should be seen in 2-14 days)[38]
Emergency department, if acute limb ischemia (patient should be seen within 4 hours)[38]
|
Modified from Harding et al. 2015 [9]
Choice of compression therapy devices
Compression therapy can be delivered with different types of devices which may have different levels of stiffness. Stiffness of a compression system is the increase of interface pressure measured in the gaiter area when standing up from the supine position.[39] High stiffness systems behave like inelastic systems and usually result in low resting pressures. That is, high stiffness systems do not exert high pressure when calf muscles are relaxed in the supine position and thus may be better tolerated by patients. In order for high stiffness systems to exert pressure, calf muscles need to contract (i.e. walking motion), making these systems beneficial to ambulating patients. For more information on stiffness of different types of compression therapy devices and their indications, see 'Types of Compression Therapy Devices' in "Compression Therapy"
- As for choice of compression therapy device, with the goal of achieving the desired optimal sub-bandage pressure and gradient compression, clinicians may choose the type of compression device according to availability of resources, clinicians' experience and familiarity with the compression device, characteristics of the ulcer, patient preference and support from caregivers.[22][40] Both multicomponent systems with an elastic layer (e.g., 4LB) or short-stretch bandages (SSB) function as high-stiffness systems and are similarly effective choices in treating VLUs.[22][40] Evidence suggests that there is no statistically significant difference in healing rates using elastic compression versus inelastic compression, four layer versus fewer than four‐layer bandage systems, different four‐layer bandage systems, or compression bandages versus compression stockings.[22]
-
2B
As for initial choice of compression therapy to promote VLU healing, we suggest multilayered (multicomponent) compression bandages over single layered compression bandages (Grade 2B)
-
- Rationale: Clinical guidelines and moderate certainty evidence (level B) supported by systematic reviews suggest use of multicomponent systems over single-component systems to promote VLU healing. [7][11][23][24][41] However, if multicomponent systems are not an option or not available, single layered bandages may still be used. After all, some compression is better than none.[7]
- With respect to multicomponent bandages, staff familiarity and experience with the different types of compression systems can greatly influence their effectiveness in treating VLUs.[22] Meta-analyses mostly based on data from countries that routinely use 4-component systems with an elastic component (4LB) have indicated that 4LB was more effective than short-stretch bandages (SSB) in VLU healing.[7] However, in countries where the medical staff is familiar with applying SSB (eg, Netherlands, Austria, Canada), there is no significant difference among VLU healing rates when comparing 4LB and SSB.[2][7][22][42] Two-component bandage systems with an elastic layer appear to perform as well as the four-layer bandage (4LB).[7][22] An industry-sponsored RCT found that more VLUs treated with SSB achieved complete closure in 16 weeks than VLUs treated with 4LB.[43] However, patients in the 4LB group had larger VLUs, which could have skewed results.
- As for insurance coverage, as long as used to treat active VLU, compression bandages and gradient compression stockings (30-40mmHg) are covered by Medicare Part B and most insurance plans in the U.S.. In terms of cost-effectiveness, multilayered compression systems with elastic components have been shown to be more cost-effective than usual care without adequate compression [32][33] and than multicomponent systems with an inelastic component (e.g., short stretch bandages) in studies performed in the U.K. and in Germany where staff had experience with 4LB.[44][45]
- Inelastic compression (e.g., Unna’s Boot) can be useful in the initial phases of edema reduction when frequent dressing changes are needed due to weeping.
-
2B
For selected patients with VLU, two-layer gradient compression stockings (HH) that deliver 40 mmHg pressure at the ankle can be considered an effective alternative to multilayered compression systems (4LB) to promote VLU healing (Grade 2B)
-
- Rationale: Based on results on a large RCT, two-layer compression stockings (HH) are as effective in healing VLU as 4LB. Furthermore, there seems to be additional benefits in reducing VLU recurrence rates and cost-effectiveness. HH is not suitable for all patients - it may be difficult to apply and remove them, and they may not be tolerated by patients with morbid obesity, lipodermatoesclerosis or severe edema. In the trial, more patients in the HH group opted to change to another compression modality mid-treatment.[46][47]
-
2C
For VLU cases that failed to respond to all other compression therapy methods, or for patient who do not tolerate other types of compression devices, intermittent pneumatic compression (IPC) therapy can be used in an attempt to promote VLU healing (Grade 2C)
-
- Rationale: IPC uses an air pump to inflate and deflate an airtight bag wrapped around the leg, but currently there is little evidence to support that addition of IPC to compression therapy offers any benefit. [24][48] Medicare will cover IPC for patients with refractory edema from chronic venous insufficiency with significant ulceration of the lower extremities who have received standard therapy but have failed to heal after 6 months of continuous treatment.[49][50]
- If personnel skilled at applying multicomponent compression bandages is not available, adjustable compression wrap devices (e.g. CircAid JuxtaCure) may be an option to promote VLU healing.[51] VLU treatment with adjustable compression wrap devices may be as effective and cheaper than treatment with multicomponent compression bandages, as the device can be reused multiple times.[51]
Compression therapy for cases that are hard-to-assess/ manage/ heal
Cases that are hard-to-assess, manage and/or heal are cases with factors that increase the likelihood of wound chronicity (chronic wounds are wounds that fail to decrease by 30% in area within 4 weeks of adequate treatment).[38] These factors can be related to [38]:
- The patient (e.g., patients with high body mass index, large limbs, abnormal shaped limb)
- The wound (e.g., VLU in challenging anatomical location)
- The healthcare professional
- The resource/treatment
When such factors are identified, compression therapy pressure levels, devices and application techniques may need to be altered to achieve the desired optimal sub-bandage pressure and gradient compression. See potential solutions for some of these scenarios in the section 'Practice Tips' in topic "Compression Therapy".
Troubleshooting adverse effects
- Compression therapy devices may cause discomfort, pain, trauma to skin, pressure damage and other adverse effects. See more information and measures to mitigate adverse effects in 'Troubleshooting Adverse Effects' in "Compression Therapy".
Transitioning to compression stockings
Compression therapy in the form of gradient compression stockings is a lifelong requirement to prevent new VLU and recurrence in patients with healed VLU. Discharge planning starts at the initial visit. For complex and mixed VLUs, early involvement of an edema management specialist (e.g, physical therapist) is recommended, as patient education and choice of long-term compression that fits patient preference and life-style may take several weeks.
-
2C
To decrease risk of ulcer recurrence in patients with a healed VLU, we suggest compression therapy with gradient compression stockings at the highest pressure patients can tolerate (Grade 2C)
-
- Rationale: VLU treatments generally do not eliminate the underlying venous hypertension that caused VLU in the first place, so a degree of compression seems to be necessary long term. [14] Although clinical guidelines for VLU recommend lifelong use of gradient compression stockings to decrease risk of recurrence [11][14][23][24], current evidence derived from meta-analyses supporting this intervention is considered limited and of low certainty. [41][52] As such, one cannot determine with certainty that compression therapy is effective in reducing risk of recurrence, but one cannot refute this possibility either. As for level of compression, results from one trial suggest that recurrence is lower in high-compression hosiery than in medium-compression hosiery. [52] Patient adherence to treatment can be problematic, but patient education on the importance of long-term compression can help improve adherence. [31] Another complicating factor is the fact that Medicare Part B and most insurance plans do not cover compression therapy (including gradient compression stockings) for prevention of recurrent VLUs once they have healed, which can also impact patient adherence if financial resources are an issue. Thus, the first pair of gradient compression stockings should be ordered when the VLU is healing but still active. If the patient has indications for surgery, underlying vascular pathology should be surgically addressed to reduce risk of recurrence (see ‘Vascular Surgical Interventions’ below).
Physical exercise
-
2C
We suggest supervised exercises to improve calf muscle pump function of patients with active and with healed VLU (Grade 2C)
-
- Rationale: Exercise can increase calf muscle pump and, as a result, decrease venous pressure and edema.[2] Furthermore, exercise has several emotional and physical benefits. Clinical guidelines suggest supervised exercises [2][14][53] supported by results of recent systematic reviews and meta-analysis (level C, low certainty evidence due to imprecision).[54][55] Specifically, simple progressive resistance exercises, such as heel raises, and 30 minutes of walking at least 3 times per week may help heal 1 more patient with VLU for every 4 patients treated with prescribed exercise plus compression than if using compression alone.[55] Exercise might be difficult for patients with low mobility and physical therapist supervision can help increase adherence, but if this resource is not accessible clinicians can still encourage patients to exercise.
Leg elevation
-
2C
Clinicians might opt to recommend leg elevation to decrease edema and prevent recurrence of VLU (Grade 2C)
-
- Rationale: There is lack of evidence that leg elevation can have a direct impact on VLU healing or recurrence. [56] However, studies have shown that leg elevation can help reduce edema and "fatigue" symptoms in patients with chronic venous insufficiency. [57][58] Leg elevation is most effective for edema reduction if performed for 30 minutes three or four times per day, although this regimen might be difficult for patients to follow. [59]
Nutrition
- For patients with malnutrition, nutritional supplementation is recommended
- Rationale: Most patients with VLU are obese or overweight and both are associated with delayed VLU healing. [60] Nutritional assessment and plan should consider all aspects of malnutrition, from overweight to undernutrition. Nutritional support is required if an individual is undernourished. [14][24] Protein intake must be adequate to support the growth of granulation tissue [2][14] and protein deficiency has been shown to be associated with increase in VLU area. [61] Oral zinc does not appear to aid in VLU healing in individuals with no zinc deficiency, however should be supplemented if zinc is deficient. [62]
Oral systemic agents
Pentoxifylline
-
1B
For patients with long standing or large VLU, we recommend use of pentoxifylline 400 mg tablet taken three times a day in addition to compression therapy, to promote ulcer healing (Grade 1B)
-
- Rationale: Current evidence indicates that not only is pentoxifylline an effective adjunct to compression bandaging for treating VLU, but it may be effective in cases where compression cannot be used or tolerated. [2][14][17][63][64] Pentoxifylline is a haemorheological agent known to influence microcirculatory blood flow and oxygenation of ischemic tissues, although the actual mechanism of action is uncertain. [64] It improves the flow properties of blood by decreasing its viscosity. Adverse effects includes gastrointestinal disturbances that are usually tolerated by patients. [46] Pentoxifylline, initially developed to treat peripheral arterial disease, has been used off-label in VLUs.[2] Treatment of VLU with pentoxifylline and compression has been found to be more cost-effective than with compression only. [17][64]
Lymphatic drainage
-
2C We suggest against adjunctive manual lymphatic drainage to promote healing of simple VLU (Grade 2C) [2]
-
- Rationale: There is disagreement among clinical guidelines in regards to the benefits of adjunctive lymphatic drainage for healing of VLU. [65] While the Society for Vascular Surgery discourages its use for ulcers of pure venous etiology [2], the International Consolidated Venous Ulcer Guideline recommends it. [11] Manual lymphatic drainage is a well established, effective therapy in patients with lymphedema [2], and it appears to reduce symptoms of chronic venous insufficiency [66] however, currently there are no data from randomized controlled trials linking manual lymphatic drainage to VLU healing. [2][65][65]
- For patients with phlebolymphedema (chronic venous insufficiency and lymphatic insufficiency), lymphatic drainage is likely to be beneficial. See topic "Lymphedema - Treatment and Emerging Strategies for Prevention"
Early surgical management of incompetent superficial veins
- 1AFor patients with venous reflux due to superficial venous incompetence, as demonstrated by duplex ultrasound and an active healable VLU or a healed VLU, early surgical treatment of incompetent superficial veins in addition to compression therapy is recommended, to promote healing of an active VLU and prevent recurrence (Grade 1A). [18][67][68][69]
- Rationale: high certainty evidence shows that early endovenous ablation of superficial venous reflux as an adjunct to compression therapy is associated with a shorter time to healing of VLU than compression therapy alone, and increases ulcer-free time when compared to endovenous ablation performed after the VLU has healed. In addition, early endovenous ablation of superficial venous reflux is highly likely to be cost-effective over a 3-year horizon compared with deferred intervention. Early intervention accelerates healing of VLU and reduces the overall incidence of ulcer recurrence. [70][71]
- Based on the evidence above, the European Society for Vascular Surgery (ESVS) recommends that a pragmatic approach be adopted in healthcare systems, where assessment and ablation of superficial venous reflux are performed as quickly as possible, and ideally within two weeks of ulcer onset.[18]
- As for insurance coverage in the U.S., CMS considers treatments to eliminate the saphenous vein reflux medically necessary if the patient remains symptomatic after a trial of conservative therapy and has reflux in a saphenous vein. Clinicians should check their respective Medicare Administrative Contractor (MAC) local coverage determination to confirm how long conservative therapy should be in place to meet surgical eligibility. Conservative therapy includes compression therapy, weight reduction, a daily exercise plan, periodic leg elevation.[72] Refer to topic "Medicare Coverage Determinations for Wound Care".
- For additional interventions on surgical management of VLU see section 'Vascular Surgical Interventions' below.
Address Patient Concerns
For all patients with healable, non-healable or maintenance VLUs, pain (related to the ulcer itself and to treatment), odor and exudate are frequently reported as significant events that negatively impact quality of life. These factors pose additional and cumulative effects on sleep, mobility and mood [73] and many patients have strong feelings of powerlessness.[74]
-
For pain management clinicians might opt to follow the World Health Organization (WHO) Pain Ladder for cancer patients, with modifications for wound care. Benefits and harms of each step should be considered. In summary [75]:
-
- Step 1: A non-opiod analgesic (e.g., NSAID) with or without an analgesic adjuvant. Adjuvants include tricyclic antidepressants, anticonvulsants, antihistamines, benzodiazepines, steroids, and phenothiazines.
- Step 2: If pain is not controlled continue the initial medication and add an opioid, such as codeine or tramadol, and an adjuvant
- Step 3: If pain is not controlled discontinue second step medications and initiate a more potent oral narcotic
- For odor clinicians might opt for topical metronidazole and charcoal dressings. Presence of infection should be assessed.
-
-
Topical metronidazole: 0.75% or 0.8% gel can be applied directly on the wound once or twice daily for 7 days to reduce odor. [16] Small size RCTs have shown that topical metronidazole is more effective than placebo [76][77][78] and as effective as PHMB in reducing chronic ulcer malodor. [79] A systematic review [80] concluded that current evidence derived from these small RCTs is of low certainty and thus not strong enough to recommend routine inclusion in guidelines. Topical metronidazole may control odor through its action on anaerobic bacteria that produce volatile acids, without the side effects of oral use as there is little or no systemic absorption. In a large, international survey (n=1444 clinicians) on interventions to control ulcer odor, metronidazole was used by only 43% of the respondents, however among those who used it 49.8% considered it very effective.[81] Reasons behind the relatively low adoption could be lack of availability in some countries, cost and need for prescription. In the U.S., use of topical metronidazol for ulcer malodor is off-label.
-
Charcoal dressings: a systematic review [82] found that activated charcoal dressings applied to fungating wounds significantly controls odor, as long as the dressing is sealed and the wound is dry. Examples include CarboFLEX (ConvaTec, USA), Clinisorb (CliniMed Ltd., UK), Actisorb Silver (KCI, an Acelity company, USA). [16] In a large, international survey (n=1444 clinicians) on interventions to control ulcer odor, charcoal dressings was the most used intervention, however only 48.4% rated this as being very effective. [81] This may be due in part to the wide range of products available, with different modes of action and application methods.
- Acetic acid: the presence of a sweet odor, blue-green drainage, and wet yellow slough may indicate Pseudomonas aeruginosa infection. In such cases, clinicians might opt for gauze moistened with 0.25% acetic acid, applied twice daily, until healthy granulation tissue develops.[83][84]
- Other methods that have been used include silver dressings, PHMB dressings, honey dressings, oral administration of chlorophyll tablets, kitty litter under the bed, commercial deodorizers and peppermint oils on dressings.[16]
-
For exudate management dressings should be tailored to the amount of exudate according to their ability to absorb exudate and promote moisture balance under compression. For information on the main types of dressings and their features see "Dressing Essentials". For decision support on brands, see Dressings Interactive Feature Matrix.
- Other concerns should be addressed on an individualized basis as well.
Local Wound Care
For all patients (healable, non-healable, maintenance), appropriate local wound care should be implemented. Table 2 summarizes interventions for healable and non-healable/maintenance VLUs. [85] Evidence and recommendation for each intervention are listed after the table.
Table 2. Local wound care for venous leg ulcers
|
Healable |
Non-healable/ maintenance |
| Cleansing |
- Gently cleanse with normal saline, sterile water or commercial wound cleanser. Irrigate wound with > 100 mL of room/body temperature solution at low pressure (4-15 psi)
- If infected, consider antiseptic solution
|
| Debridement |
- Topical anesthetics, such as lidocaine-prilocaine cream, if needed to reduce debridement pain
- Surgical, sharp, mechanical, autolytic, enzymatic, or combination of methods
|
- Conservative debridement of non-viable tissue only
- Do not debride if circulation is severely impaired (mixed arterial ulcer)
|
| Infection management |
- Use antimicrobial dressings only in cases of clinical infection (e.g. if increasing pain is observed) or if no healing is seen in 4 weeks despite appropriate care
- Antimicrobial dressings (with cadexomer iodine, silver, etc):
-
- For light exudate: hydrogel or hydrogel colloidal sheet-based
- For moderate, heavy exudate: alginate, hydrofiber, super absorbent
- In confirmed clinical infection, prescribe systemic antibiotics guided by culture
|
- Use antimicrobial dressings and antibiotics only in cases of clinical infection (e.g. if increasing pain is observed)
- Antimicrobial dressings (with cadexomer iodine, silver, etc):
-
- Non-adherent dressing-based
- In confirmed clinical infection, prescribe systemic antibiotics guided by culture
|
| Peri-wound skin care |
Consider barrier products for periwound skin
|
| Moisture balance |
- Fill deep wounds to avoid dead space
- Maintain wound moisture with:
-
- Hydrocolloid, hydrogel, moisture retentive foam
- Manage exudate with:
-
- Alginate, gelling fiber, foam, composite dressing, specialty absorbent
|
- Apply appropriate non-adherent dressing OR
- If minimal or light exudate: paint wound with antiseptics (e.g. povidone iodine)
- If dry or wet gangrene: moisture retentive dressing may cause limb threatening infection
- Avoid conventional dressing products that require daily dressing changes
|
Cleansing
-
2C
We suggest that non-infected VLU wounds be cleansed with a neutral, nonirritating, nontoxic, non-antimicrobial solution such as sterile saline or water initially and at each dressing change, and that routine wound cleansing be accomplished with a minimum of chemical and/or mechanical trauma (4-15 psi of pressure) (Grade 2C)
-
- Rationale: Low certainty evidence (level C) supports routine cleansing to promote faster improvement of VLU compared with no cleaning. Most patients with VLU present with significant wound exudate and other debris in and around the wound area that must be cleansed before dressing application to decrease bacterial load and remove loose material.[2][14][86][87][88] Sterile saline is typically used, but potable tap water, boiled and cooled and bottled water can be used as well.[89] Currently, there is insufficient evidence to demonstrate whether the use of 0.2% polyhexamethylene biguanide (PHMB) solution compared with saline solution; aqueous oxygen peroxide compared with sterile water; propyl betaine and polihexanide compared with a saline solution; or octenidine dihydrochloride/phenoxyethanol (OHP) compared with Ringer's solution makes any difference in the treatment of VLUs.[90] Medicare Part B does not currently cover wound cleansers.
Debridement
-
1C For healable VLUs, we recommend debridement (vs. no debridement) at initial assessment and during subsequent visits as needed (Grade 1C)
-
-
- Rationale: There is consensus in the wound care literature that debridement is necessary to promote wound healing [85][91][23], however current evidence is limited (level C) [2][11][14][86][92][93] and does not allow one to conclude with confidence that debridement improves healing of non-infected VLU, or if wounds that are healing debride themselves. [92] Nevertheless, studies suggest that active debridement removes obvious necrotic tissue and excessive bacterial burden that can slow down the healing process and increase the risk of osteomyelitis and sepsis. [94][95] Furthermore, Medicare Part B and other insurers will only cover surgical dressings for ulcers that have been debrided or surgically created. [15]
-
-
- Current studies do not provide sufficient evidence to conclude with confidence which method of debridement is most effective in promoting VLU healing [2][11][14][86][92], so other factors need to be considered when choosing among debridement methods.
-
2C
When choosing debridement methods for VLU patients, we suggest clinicians consider factors such as status of the wound, health care provider's familiarity with each technique, overall condition of the patient, professional licensing restrictions, patient preference and insurance coverage (Grade 2C)
-
-
Surgical debridement: indicated for VLUs with large necrotic area or associated cellulitis/osteomyelitis, in patients with no significant clotting disorders or active anticoagulant therapy, and with no other contraindications for general/regional anesthesia. Surgical debridement is rapid and highly selective, but is resource intensive (needs operating room and personnel, must be performed by a physician or qualified non-physician practitioner), may cause excessive bleeding, transient bacteremia, damage to tendons and nerves, and may add anesthesia-associated risks. [92]
-
Sharp debridement with scissors, scalpel, and forceps (Figures 1-3): for VLUs with necrotic areas or signs of infection, in patients with no significant clotting disorders or active anticoagulant therapy, patients who are not surgical candidates, or when resources needed for surgical debridement are not available. Sharp debridement must be performed by trained, licensed health care practitioners, and it is often performed at bedside or in a procedure room. It has been termed the “gold standard” of wound debridement [92][96] and is the preferred method recommended by the Society for Vascular Surgery. [2] This method does not have the extra risks and costs associated with general/regional anesthesia. It is less aggressive than surgical debridement, but it is also imprecise and may carry the greatest risk of tissue damage of any of the debridement methods. [94] Debridement is often painful [97] and patients may not tolerate the entire procedure [98], therefore pain needs to be well managed. [2] EMLA (5%) applied for 30 to 45 minutes in a dose of 1 g to 2 g/10 cm² significantly reduces the pain from sharp debridement and decreases post-debridement pain in patients with VLU. [99] However, in certain countries application of EMLA on wounds is not approved by regulatory bodies.
-
Enzymatic debridement (e.g, collagenase): suitable for VLU with no signs of infection, when sharp or surgical debridement is not available or not an option. Topical application on normal tissue can cause irritation of the peri-wound skin. [94]
-
Autolytic debridement (e.g., hydrocolloid dressings): for patients with exudative VLUs [100] with no signs or potential for infection (e.g, ischemia of the limb or digit) [92][100][101], and when sharp or surgical debridement is not available or not an option. Autolytic debridement is done by occluding the wound with a dressing that traps exudate in the wound, allowing endogenous proteolytic enzymes produced by macrophages present in exudate to selectively liquefy necrotic tissue. [92] This method is slow and works only in the presence of exudate but it is highly selective, painless and requires only minimal clinical training. [94]
-
Biological debridement (e.g, maggots): for VLU patients with no personal bias against maggots, when sharp or surgical debridement is not available or not an option. [2] Can be painful. [102]
-
High pressure jet debridement: for VLU patients with same indication and contraindications as sharp debridement, may be an option when sharp/surgical debridement or other forms of debridement are not available. Low-quality RCTs do not show any difference on VLU healing when high pressure jet debridement is compared with sharp debridement. [103][104] Cost of the device may be a limiting factor.
-
Mechanical debridement (e.g, wet-to-dry dressings): for VLU patients with no signs of infection, when sharp or surgical debridement is not available or not an option. This form of debridement is non-selective, slow, and often painful. [100][101] Wet-to-dry dressings can increase potential for infection in large wounds with extensive necrosis. [105]
|
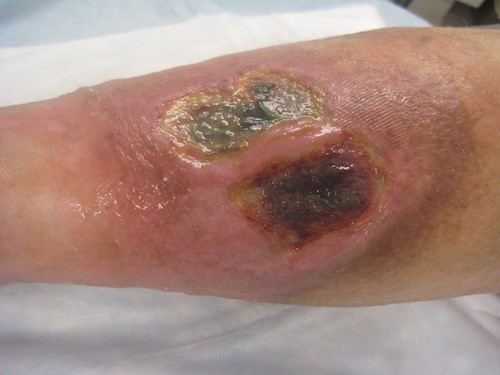
Figure 1. Venous ulcer with fibrin and necrotic tissue pre-debridement
|
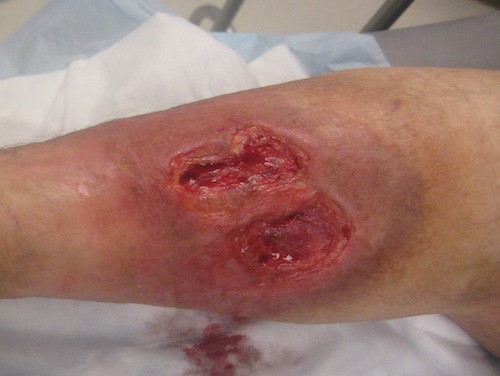
Figure 2. Venous ulcer immediately post sharp debridement
|

Figure 3. Venous ulcer after sharp debridement, primary dressings and compression
|
Infection management
Antimicrobial topical agents
-
2C
Current guidelines suggest use of antimicrobial topical agents (e.g., antimicrobial creams, ointments and impregnated dressings) only on clinically infected VLU or if there is no healing with standard of care for at least 4 weeks (Grade 2C) [2][11][106][107]
-
- Rationale: An antimicrobial is an agent that kills microorganisms or stops their growth without harming the host. Antibiotics are considered antimicrobials as well. In light of the increasing problem of bacterial resistance to antibiotics, topical antibiotic use on non-infected wounds cannot be justified. [2] If, however, patients show signs of VLU infection topic and systemic antibiotic therapy should be promptly initiated. [11]
- As for insurance coverage in the outpatient setting, topical antiseptics and topical antibiotics do not have a special coverage arrangement under the Medicare Part B surgical dressing benefit. [15] Of note, if an antimicrobial impregnated dressing is ordered for a Medicare Part B covered patient, the DME supplier might substitute it for another lower cost product with similar HCPCS, as most antimicrobial-impregnated dressings have similar HCPCS codes as their non-antimicrobial impregnated counterparts even though they are more costly to manufacture. [108] When paid out-of-pocket, antimicrobial impregnated dressings are usually more expensive than their non-antimicrobial impregnated counterparts.
- Effectiveness of antimicrobial topical preparations are supported by different levels of evidence :
-
Cadexomer iodine: for patients with infected VLU, clinicians might opt for cadexomer iodine with compression to promote wound healing. Low-quality evidence (level C) suggests that cadexomer iodine is more effective than standard care in healing VLU [106]. Cadexomer iodine products can also absorb exudate and promote debridement. Cases of hyperthyroidism after treatment of VLU have been reported. [109]
-
Silver-based dressings: a recent network meta-analysis concluded that silver dressings may increase the probability of VLU healing, compared with non-adherent dressings (RR 2.43, 95% CI 1.58-3.74, level B, moderate-certainty evidence in the context of a low-certainty network). [110]
-
Honey-based preparations: current evidence of low quality (evidence level C) does not support use of honey-based products for VLU patients as it is unclear whether honey increases rate of healing of VU,\ compared with no honey. [106][111] One of these trials conducted a rigorous cost-effectiveness analysis in parallel with the RCT and concluded that honey was unlikely to be cost-effective in promoting VLU healing. [112]
-
Povidone-iodine: current evidence of low quality (evidence level C) suggests that there is no difference in VLU healing when povidone-iodine preparations are used compared with other interventions (dextranomer, hydrocolloid dressing, paraffin gauze dressing, foam dressing). [106]
-
Other topical antimicrobials: current evidence is of poor quality and does not allow one to make definite conclusions about the effectiveness of peroxide-based preparations, ethacridine lactate, chloramphenicol, framycetin, mupirocin, ethacridine or chlorhexidine in healing VLU. [113][106]
Systemic antibiotics
- 1CWe recommend use of systemic antibiotics for patients with VLU with clinical signs of infection but not for patients with VLUs that are just colonized by bacteria (Grade 1C)
-
- Rationale: At present, no evidence is available to support the routine use of systemic antibiotics to promote VLU healing (level C, low certainty evidence). [106] Clinical guidelines are in agreement that systemic and topic antimicrobial agents should be used only in cases of clinical infection and not bacterial colonization (< 1x10^6 CFU/g of tissue), due to the increasing problem of bacterial resistance to antibiotics. [2][11] Overuse of antimicrobials is an emergent public health problem, and it is linked to the development of resistant organisms and iatrogenic disease, such as Clostridium difficile colitis, and increased health care costs. [19] The choice of systemic antibiotics should be guided by sensitivities performed on wound culture collected with a validated quantitative swab or biopsy. Oral antibiotics for 2 weeks is preferred as initial treatment. [2][11] combined with mechanical disruption (debridement) of the ulcer.
-
For patients with clinically infected VLU whose bacteriological analysis shows virulent or difficult to eradicate bacteria (such as beta-hemolytic streptococci, pseudomonas, and resistant staphylococcal species), antimicrobial therapy can be initiated even if levels or colony-forming units per gram of tissue are < 1x10^6 CFU/g of tissue (accepted threshold for wound infection). [2][14]
-
Cellulitis surrounding VLUs are usually caused by streptococci or staphylococci and should be treated with systemic gram-positive bactericidal antibiotics, reserving broad coverage for unresponsiveness. [2][14]
-
- For afebrile, healthy patients, may start with flucloxacillin 1g tablets to be taken orally four times a day or clarithromycin 500mg tablets to be taken orally twice a day. Reassess in 5 days. [114]
- If febrile and ill, admit for intravenous antibiotics
Peri-wound care
Skin maceration
-
2C
For patients with VLU whose peri-wound skin is likely to be in contact with exudate, we suggest application of a skin protectant, also known as topical barrier, on the peri-wound skin to prevent skin maceration and enlargement of wound (Grade 2C)
-
- Rationale: Skin maceration is primarily caused by excess exudate, which can enlarge the wound and impede healing [14]. The mainstay of treatment and prevention of skin maceration is management of excess exudate (See 'Moisture Management' below). There is low-quality evidence showing that skin protectant is more effective than no skin barrier on peri-wound skin in promoting healing of VLU. [115] Different types of skin protectants seem to be equally effective in promoting wound healing, but some might be removed/applied more easily than others. [116] Skin protectants are, however, not covered by Medicare and most insurance plans, so cost is an important factor to be considered.
Stasis dermatitis
-
1A
For VLU patients with stasis dermatitis and dry, flakey peri-wound skin, we recommend use of daily moisturizers to alleviate symptoms and dermatitis flares underneath compression (Grade 1A)
-
- Rationale: Patients with VLU often have stasis dermatitis (stasis eczema), which manifests as dry, itchy skin, erythema, hemosiderin deposition, and scaling. [117][118] If left untreated, symptomatic stasis dermatitis can lead to scratching and fissuring which can delay VLU healing and contribute towards development of new ulcers. Stasis dermatitis is a result of venous hypertension [119][120] and thus, compression is considered the main treatment for stasis dermatitis. Topical local treatment is also essential and consists mainly of emollients and steroids. There is high-quality evidence that supports use of moisturizers on eczematous skin to improve symptoms, and decreases the amount of topical corticosteroids needed to achieve similar reductions in eczema severity. [11][121] There is no reliable evidence that one moisturizer is better than another. Moisturizers are however, not covered by Medicare and most insurance plans, so cost is important when choosing moisturizers.
-
2CFor VLU patients with stasis dermatitis and dry, lightly reddened, itchy, inflamed skin, we suggest short courses of topical mid-potency steroids, such as triamcinolone 0.1% or betamethasone valerate 0.12% foam (twice a day for 4 weeks) (Grade 2C).[2][120][122] Clinicians might also opt to change underlying padding to pure cotton cast padding. [6]
-
- Alternative interventions include treatment with oral doxycycline 100 mg with topical tacrolimus 0.1% for four weeks (evidence level C) [123], but tacrolimus is currently FDA-approved only for atopic dermatitis and not for other types of dermatitis.
-
For VLU patients with stasis dermatitis and dry, itching, burning skin, besides interventions listed above, clinicians might also opt to use zinc or calamine impregnated rolled gauze wrap as first layer [6] (e.g, Viscopaste bandage)
Contact Dermatitis
-
2C
Skin moisturizers with no lanolin underneath compression can also help reduce contact dermatitis (Grade 2C) [2]
-
- Rationale: Peri-wound skin of VLU patients seems to be more susceptible to contact dermatitis and care must be taken to avoid irritant agents, sensitizing allergens such as bacitracin, sulfa, lanolin (a component of some moisturizers) [124], and trauma on peri-wound skin due to repetitive removal of dressings. [125]
Moisture management and dressing selection
Selection of primary and secondary dressings should follow established wound bed preparation principles. [10][85] For customized, wound-specific recommendations, use Wound Prep and Dress Tool. For practical information on dressing indications, contraindications, application, Medicare coverage and brands see "Dressing Essentials". General recommendations and evidence regarding dressings for VLUs are listed below.
-
2B
We suggest clinicians apply a topical dressing that will manage excess exudate, protect peri-wound skin and maintain a moist wound bed (Grade 2B)
-
- Rationale: Excess exudate should be contained by absorptive dressings as it has been shown that VLU exudate has high concentrations of proteases and inflammatory cytokines that prohibit wound healing and may damage fragile peri-wound skin. [2][14] Also, several experimental studies support the hypothesis that a moist wound bed increases wound healing when compared to a dry wound bed by facilitating cell migration and matrix formation.
-
2C
Taking into consideration Medicare coverage, cost and level of exudate, we suggest clinicians initiate treatment with the primary dressings below, associated with compression (Grade 2C)
-
- Heavy exudate: specialty absorptive, hydrofiber or alginate dressings
- Moderate exudate: specialty absorptive, foam dressings
- Light exudate: non-adherent dressings or hydrocolloid
- Minimal or no exudate: non-adherent dressings, film or hydrogel
- Rationale: As for choice of primary topical dressing, current evidence (level of evidence C) suggests that when used with adequate compression in patients with VLU, different types of primary dressings are equally effective as long as they are used beneath adequate compression. As a result, other factors such as exudate level, cost, ease of application, clinician and patient preference and dressing availability play a major role in dressing selection. [2][59][113][126][127][128]
-
1CCompared with other primary dressings that can keep wound bed moist, we recommend against use of wet-to-dry dressings to promote wound healing in patients with VLU receiving compression (Grade 1C)
-
- Rationale: Although there is little evidence that one specific type of dressing is superior in promoting VLU healing when associated with compression, wet-to-dry dressings require frequent dressing changes in order to keep wound bed moist, resulting in increased nursing costs. Also, if dry, it can be painful to remove, debride wound unselectively and remove viable tissue, and prevent cells from migrating to the wound bed.
-
2C
As for dressings for pain management, clinicians might opt to use foam dressings containing ibuprofen for painful VLU with at least moderate levels of exudate (Grade 2C)
-
- Rationale: On the basis of two trials, there is some evidence that ibuprofen containing dressings (Biatain-Ibu, Coloplast A/S) provides pain relief for some people with painful venous leg ulcers. The release of ibuprofen into the wound bed is dependent on the presence of wound exudate and it would not have any effect on wounds with no exudate. [99] This dressing is currently not available in the U.S.
Plan Reassessment
- Healable VLUs that fail to reach a 30% decrease in size after 4 weeks of treatment with standard therapy described above should be reassessed.[2][14] Re-evaluation of the patient and wound should be performed before consideration of adjuvant therapies to rule out other differential diagnoses, ensure that compression has achieved edema control, bio-burden and exudate are well managed, and factors impeding healing are not present or under control. See topics "Venous ulcers - Introduction and Assessment" and "How to Determine Healability of a Chronic Wound"
- Based on the reassessment, a revised care plan should be generated.
- Medicare Quality Payment Program, Quality Measure: "Plan of Care for DFU or VLU patients not achieving 30% closure at 4 weeks"
Consider differential diagnoses
- Review VLU differential diagnoses. Referral to other specialists if autoimmune/micro-occlusive disorders, other vascular diseases, dermatologic conditions, metabolic, hematologic disorders are suspected.
-
Wound biopsy: recommended for VLU that do not improve with standard care after 4 weeks of treatment and for all ulcers with atypical features.[5][2]
-
- Preferably, biopsies should be punch or elliptical biopsy specimens, taken from the edge of the ulcer to compare the ulcerated area with the surrounding skin[5], and central provisional matrix.[2]
- H&E is widely used in wound histopathology analysis, but other special stains can be used depending on the differential diagnosis.[5] Additional tissue analysis to include inflammatory cytokines and MMP are still under investigation and does not have routine clinical applicability.[2][129]
- The specimen taken from the center of the wound should also be sent for culture.[5]
Reassess factors that may impede healing
- Uncontrolled diabetes, immunosuppression, smoking, malnutrition
- Inadequate blood supply to the ulcer. Re-check blood supply with non-invasive arterial vascular testing such as ABI, toe brachial index, handheld Doppler waveform analysis, skin perfusion pressure or other.
- Check for thrombophilia, which is associated with recurrent and recalcitrant ulcers.[2] Laboratory evaluation includes:
-
- Inherited hypercoagulable factors (anti- thrombin deficiency, protein C and protein S deficiencies)
- Factor V Leiden (resulting in activated protein C resistance)
- Prothrombin G20210A
- Plasminogen activator inhibitor type 1 mutations
- Hyperhomocysteinemia
- Antiphospholipid antibodies (anticardiolipin and lupus anticoagulant)
- Cryoglobulins and cryoagglutinins
- Factor VIII related antigen, von Willerbrand factor (VWF), D-dimer and factor V Leiden: if indicative of hypercoagulation tendency, pose a risk factor for post-thrombotic syndrome
Check for adequate compression and edema control:
- Evaluate if desired pressure is being delivered through compression method of choice. See topic "How to Select Adequate Compression Therapy Pressure Levels and Products" and topic "Compression Brands Quick Reference"
- Check patient’s adherence. Adherence can be improved by educating patients on the importance of compression therapy.[31] See section 'Patient Education for Clinicians' below.
Check for presence of infection:
-
Wound culture: If infection is suspected in a VLU or if VLU is not healing in 4 weeks, a specimen should be obtained from the wound surface (not the drainage) with a validated quantitative bacteriology swab method and sent for microbiological analysis.[2][14] Infection in VLU can manifest as fever, leukocytosis, worsening pain, cellulitis, purulence, increased exudate, malodor, discolored friable granulation tissue, biofilm, tissue necrosis.[2]
Check for adequate exudate control
- See section 'Moisture management' above, and for ulcer-specific dressings and wound bed preparation suggestions use "Wound Prep and Dress Tool"
Evaluate need for vascular interventions
- Consult with vascular/vein specialist to assess need for vascular intervention. See section 'Vascular Surgical Interventions' below.
Vascular Surgical Interventions
Table 3 summarizes recommendations by the Society for Vascular Surgery and American Vascular Forum (SVS)[2] and the European Society for Vascular Surgery (ESVS) [18] on vascular surgery interventions to treat or prevent VLUs. For surgical interventions for CVD, see section 'Prevention: Surgical Management Strategy' below and topic "Chronic Venous Disease - Surgical Management".
Table 3. Vascular surgery interventions for VLUs - recommendations by the Society for Vascular Surgery and American Vascular Forum (SVS) [2] and the European Society for Vascular Surgery (ESVS) [18]
Findings
|
Suggested surgical procedure (always combined with compression therapy)
|
Desired outcome and strength of recommendation according to the GRADE framework
|
Incompetent superficial veins with axial reflux towards the ulcer
|
Pathologic perforating veins*
|
Deep vein disease
|
|
To promote healing of active VLU (C6)
|
To prevent recurrence of active VLU (C6)
|
To prevent recurrence of healed VLU (C5)
|
To prevent first time VLU in at risk patients (C4b)
|
| Present |
Absent |
Present or absent |
Ablation of superficial incompetent veins
|
1A1A
|
1A1A
|
1A1A
|
2C2C2C2C2C
|
| Present |
Present |
Present or absent
|
Ablation of superficial and perforator *** incompetent veins
|
2C2C2C2C
|
2C2C
|
2C2C
|
2C2C
|
|
Absent
|
Present |
Present or absent
|
Ablation of the “pathologic” perforating veins **
|
2C
|
2C |
2C |
n/a |
|
|
Infrainguinal deep venous obstruction
|
Autogenous venous bypass or endophlebectomy
|
2C
|
2C |
2C |
2C |
|
|
Deep venous reflux
|
Ligation of femoral or popliteal veins
|
2CSuggest against
|
2CSuggest against
|
2CSuggest against
|
2CSuggest against
|
|
|
Deep venous reflux, with axial reflux with structurally preserved deep venous valves
|
Individual valve repair
|
2C |
2C
|
2C
|
2C
|
|
|
Deep venous reflux, with absence of structurally preserved axial deep venous valves when competent outflow venous pathways are anatomically appropriate for surgical anastomosis
|
Valve transposition or transplantation
|
2C |
2C
|
2C
|
2C
|
|
|
Deep venous reflux, no other option available |
Autogenous valve substitutes by surgeons experienced in these techniques
|
2C |
2C |
2C |
2C |
|
|
Proximal chronic total venous obstruction/severe stenosis
|
Endovascular repair: venous angioplasty and stent recanalization
|
1C |
1C |
1C |
1C |
|
|
Proximal chronic total venous obstruction/severe stenosis - bilateral - with recalcitrant VLU and failed endovascular treatment
|
Open surgical bypass with use of an externally supported expanded polytetrafluoroethylene graft
|
2C |
2C |
n/a |
n/a |
|
|
Unilateral iliofemoral venous occlusion/severe stenosis with recalcitrant VLU with failed endovascular reconstruction
|
Open surgical bypass with use of saphenous vein as a cross-pubic bypass (Palma procedure) |
2C |
2C |
n/a |
n/a |
* Pathologic perforating veins (characterized by outward flow of >500 ms duration, with a diameter of >3.5 mm) located beneath or associated with the healed ulcer bed
** If patient undergoes pathologic perforator vein ablation: SVS recommends treatment by percutaneous techniques that include ultrasound-guided sclerotherapy or endovenous thermal ablation (radiofrequency or laser) over open venous perforator surgery to eliminate the need for incisions in areas of compromised skin (Grade 1C).
Adjunctive Therapy
Adjunctive therapies for the healing of VLU should only be considered if VLU does not decrease by 30% after 4 weeks of comprehensive care including compression therapy, debridement, control of bio-burden, wound moisture management, and venoactive agents (e.g. micronised purified flavonoid fraction, hydroxyethylrutosides, pentoxifylline, or sulodexide).[2][19][14][53][18] Comprehensive care should be continued while using adjunctive therapies. Evidence and/or cost-effectiveness supporting these interventions are not sufficiently strong to justify them as primary therapy.[130] Re-evaluation of the patient and wound should be performed prior to initiating adjunctive therapy.[2]
Wound Coverage
Autografts, allografts, cellular and/or tissue products and other methods can be used to achieve wound coverage as an adjunctive intervention, which may result in healing rates of up to 73%.[131]
Autologous Skin Grafts
- Skin grafting should be considered as primary therapy for large VLUs (i.e. >25 cm2), as healing is unlikely without grafting.[2]
-
2CFor patients with VLU that failed to decrease in size by 30% in 4 weeks of standard therapy, we suggest consideration for split-thickness autologous skin graft accompanied with compression, over continuation of standard care alone (Grade 2C).
-
Rationale: Although available literature cannot provide firm evidence of benefit in VLU healing, clinical guidelines suggest autologous skin grafts for recalcitrant VLUs [2][14][132][11], as skin grafting can result in better quality of life in these cases, when compared with standard care alone.[133]
- Application of autologous split-thickness skin graft is performed by surgeons, and requires anesthesia. Split-thickness skin graft is obtained from a healthy area of the patient (e.g., thigh) with a surgical blade or dermatome, and the graft is applied on the wound. As a result, the procedure creates additional wounds (on the area that donated healthy skin), which might increase postoperative discomfort and create cosmetic concerns.
- Prior to any wound coverage procedure, clinical guidelines recommend that the wound bed be prepared adequately, by removing slough, debris, necrotic tissue, reducing bioburden (preferably to 10^5 CFU/g of tissue), and ensuring no beta hemolytic streptococci are present in the ulcer[14]
- It is important to note that surgical coverage of the wound will not address the underlying cause of VLU, and thus continued use of compression therapy is essential.[14] Overall, all patients with VLUs being considered for skin graft should undergo vascular surgery to correct the underlying venous abnormalities causing the VLUs and avoid skin graft breakdown.[2]
- A retrospective study has reported use of the minimally invasive Cellutome system (KCI, an Acelity Company) for autologous skin grafting of venous and mixed venous ulcers.[134]
Free Flaps
- For patients with recalcitrant VLU with severe lipodermatosclerosis, the Wound Healing Society guideline suggests free flap transfer.[14]
-
- Rationale: Wide excision of diseased tissue and replacement with uninjured tissue and venous valves can help in the treatment of VLU. Microsurgical flaps are performed by specialists, require hospitalization and specialized healthcare staff.
Pedicled and perforator flaps
- For patients with recalcitrant VLU with severe lipodermatosclerosis, clinicians might opt to cover VLUs with pedicled or perforator flaps when free flaps are not an option.[135]
-
-
Rationale: Free flaps are still preferred for these cases as they avoid additional scarring in surrounding area and overcome other limitations of pedicled and perforator flaps. Scarring is commonly seen when local flaps are used and primary closure is not possible due to excessive tension. Pedicled flaps, such as the reverse sural flap, sacrifice major vascular supply to the foot. Local transposition flaps can only be used to cover small defects.[135] In general, coverage failure rates of free flaps used to cover defects of lower extremity distal third are comparable to those of local transposition flaps (e.g. propeller flaps).[136]
Cellular and/or tissue products
Cellular and/or Tissue Based Products (CTPs) allow patients with VLUs that have failed to show signs of improvement in 4-6 weeks the chance to receive wound coverage without problems inherent with autografts (e.g., autograft harvesting, hospital stay, anesthesia risks, donor area).[132] Generally, CTPs can be applied in an outpatient setting. The choice of CTP relies heavily on physician/patient preference, costs, availability of resources and accessibility to CTPs. CTPs can be categorized in one of the categories below [15] and may be covered by Medicare and private insurers if all requirements are met – See “Cellular and/or Tissue Based Products”. For decision support on different features of each CTP brand, see CTP Interactive Feature Matrix.
- Medicare Quality Payment Program, Quality Measure: "Appropriate use of Cellular or Tissue Based Products (CTP) for Patients aged 18 Years or Older with DFU or VLU"
-
2C
Human skin allografts: Clinicians might opt to use human skin allografts and compression therapy for patients with non-healing VLU if resources are available (Grade 2C).
-
-
Rationale: Examples of human skin allografts include TheraSkin, Gammagraft, GraftJacket. Studies have shown that treatment with human skin allografts resulted in a higher healing rate of VLUs when compared to standard care, but evidence is considered of low certainty.[132][137][138] An RCT compared a human skin allograft (TheraSkin) and a bioengineered skin graft substitute (Apligraf) to promote healing of VLUs that failed to achieve 30% reduction in size after 4 weeks of standard therapy. Authors found that while both CTPs were equally effective in promoting VLU healing by week 20, treatment with TheraSkin was 42.2% cheaper than with Apligraf (difference was statistically significant, p=0.039) even though the initial wound sizes were not significantly different between groups (evidence level C, due to imprecision and assessor blinding bias).[139]
-
2B
Allogeneic matrix: For VLUs that failed to reduce at least 30% in 4 weeks of adequate therapy, clinicians might opt for dehydrated human amnion/chorion membrane combined with compression therapy to promote healing (Grade 2B).
-
-
Rationale: Dehydrated human amnion/chorion membrane allograft (e.g., EpiFix, AmnioBand), a type of allogeneic matrix, has been shown to promote faster VLU healing when associated with compression therapy than compression therapy alone. Evidence supporting use of dehydrated human amnion/chorion membrane for VLU healing is currently considered of moderate certainty (level B).[132][138][140][141][142][143][144][145][146]
- There is moderate certainty evidence (level B) that another type of allogeneic matrix - human fibroblast-derived dermal substitute (Dermagraft) - when combined with compression therapy promotes faster healing of VLUs with less than 12 months in duration compared to compression therapy alone.[147] Despite successful use of Dermagraft in the treatment of VLUs in clinical trials, [147] currently in the U.S. Dermagraft is only FDA-approved for treatment of diabetic foot ulcers.
- There is low certainty evidence (level C) that cryopreserved placental tissue using human viable wound matrix (e.g., Grafix) associated with compression therapy is beneficial in achieving complete VLU closure in refractory cases. [148]
-
2B
Composite matrix: For non-healing VLUs, clinicians might consider bilayered bioengineered living cellular construct and compression over standard therapy (Grade 2B).
-
-
Rationale: Clinical guidelines [2][11][14] suggest use of composite matrix, bilayered skin equivalent (e.g., Apligraf) with compression to promote healing of VLUs that failed to decrease more than 30% in 4 weeks of standard treatment. Evidence for this recommendation is of moderate certainty (level B), based on 2 trials: one that compared a bilayered skin equivalent (e.g., Apligraf) with foam dressings and another that compared with a custom-made dressing that looked like Apligraf. [132][149][150] Bilayered bioengineered skin has been shown to be more cost-effective than Unna boot, but the study was partly funded by the manufacturer.[150] Despite satisfactory outcomes reported by literature, in practice composite matrices may not be easily accessible, shelf life can be relatively short (e.g.,15 days for Apligraf). Also, treatment of VLUs with bilayered skin equivalent may be as effective but more costly than with human skin allograft. [139]
-
2C
Acellular matrix: Non-healing VLU can also be treated with acellular collagen matrix derived from porcine intestinal mucosa and compression therapy (Grade 2C).
-
-
Rationale: Low certainty evidence supports use of acellular porcine matrix (e.g, Oasis Wound Matrix) to treat recalcitrant VLU.[126][132][138][151][152] As for cost-effectiveness, an industry-sponsored study concluded that acellular porcine matrix is more cost-effective than bilayered bioengineered skin (e.g., Apligraf) or single-layered dermal replacement (e.g.,Dermagraft).[153] When compared to standard care alone, acellular porcine matrix was shown to be more effective but more costly.[153] Effectiveness across different types of acellular matrix may vary. For instance, hyaluronic matrix, another type of acellular matrix, does not seem to perform better than other types of dressings in promoting VLU healing.[154][113]
Other biologics
- 2BCultured epithelial allografts or growth arrested human cells: We suggest against use of cultured epithelial allografts or growth-arrested human keratinocytes and fibroblasts (HP802-247) as adjunctive therapy for VLU (Grade 2B).
-
-
Rationale: Studies published so far have shown no statistically significant difference in VLU healing outcomes between experimental groups (evidence level B). [14][132]
-
2C
Autologous bioengineered skin or cultured epidermal autografts (CEA): We suggest against use of autologous bioengineered skin or cultured epidermal autografts (CEA) for coverage of non-healing VLU (Grade 2C).
-
-
Rationale: Use of CEA or split-thickness cultured autografts to promote healing of recalcitrant VLUs is supported by low certainty evidence. [155][156][157] [158][159] Nevertheless, the Wound Healing Society clinical guideline [14] suggests against use of CEA given the disadvantages of this method, such as the need for the patient to undergo a biopsy to retrieve skin, followed by a wait period of weeks for the cells to be cultivated.[132] The FDA has approved some of these products as humanitarian-use devices (HUD). Medicare and private insurers in the U.S. do not routinely cover these products.
-
2CPlatelet-rich Plasma (PRP) and Growth factors: We suggest against use of PRP, growth factors and cytokines as a primary therapy or to promote healing of VLU that fail to decrease in size after 4 weeks of treatment with first line therapies (Grade 2C).
-
- Rationale: Chronic wounds are deficient in growth factors and cytokines and have high levels of proteases that break down these proteins that are essential for wound healing.[160] Administration of exogenous growth factors and cytokines such as granulocyte-macrophage colony stimulating factor (GM-CSF) and platelet-derived growth factor (PDGF, manufactured or in autologous platelet-rich plasma) has shown promise in improving VLU healing.[160][161][162][163] However, these findings are based on low certainty evidence derived from small trials with methodological limitations.[160][164][165][166][167][168] Clinical guidelines also do not support routine use of growth factors and cytokines in VLU.[14][53] Furthermore, use of these interventions can be costly, most are considered off-label (use not approved by the FDA) and are not covered by Medicare or other private insurance plans, except when part of clinical trials to treat chronic wounds.[169]
Systemic Pharmaceutical Agents
-
1BMicronized purified flavonoid fraction (diosminhesperidin): For patients with long standing or large VLU, we recommend use of micronized purified flavonoid fraction (diosminhesperidin) in addition to compression therapy, to promote ulcer healing (Grade 1B).
-
- Rationale: Micronized purified flavonoid fraction (MPFF, also known as diosminhesperidin, diosmiplex, Daflon 500mg ) is an antioxidant flavonoid not approved by the FDA for treatment of VLU in the U.S (it is approved as a dietary supplement though). However, clinical guidelines and experts [2][11][170]recommend use of adjunctive MPFF, based on RCTs and a meta-analysis that showed increased VLU healing rate in patients treated with MPFF, compression therapy and adequate wound care, compared to compression therapy and wound care alone (at 6 months, relative risk reduction was 32%; 95% CI, 3%-70%)[171] MPFF is taken orally and might cause eczema and diarrhea.[172] MPFF is not covered by Medicare or most private insurance plans. Another flavonoid, hydroxyethylrutoside (HR, also known as troxuretin) has been shown to help in VLU healing when associated with compression, however studies were evidence is of low certainty (Evidence level C).[173] HR is currently not approved by the FDA for treatment of VLU.
-
2CAspirin: We suggest against use of aspirin to aid in healing or prevent recurrence of VLU (Grade 2C).
-
- Rationale: Current evidence is not strong enough to support routine use of aspirin as an adjunctive therapy to treat or prevent recurrence of VLU.[2][174][175][176] Aspirin is known to cause adverse effects, such as gastric ulceration and other gastrointestinal effects, as well as hepatotoxicity, exacerbation of asthma, skin rashes and renal toxicity.[174][177][178]
-
2COral zinc: For patients who do not have a deficit of total body zinc, we suggest against use of oral zinc to aid in healing or prevent recurrence of VLU in patients that do not have a deficit of total body zinc (Grade 2C).
- Rationale: Current evidence, albeit of low quality, suggests that oral zinc does not improve VLU healing in patients without a deficient total body zinc reservoir.[14][62]
-
2CSulodexide: We suggest against use of sulodexide as an adjunctive therapy to treat VLU in association with compression therapy (Grade 2C).
-
- Rationale: Sulodexide resembles heparin, with antithrombotic and pro-fibrinolytic properties.[179] There is some evidence that it can help in healing of VLU, but adverse effects and cost-effectiveness have not been well evaluated.[179] In most trials that investigated this compound, patients had to receive daily intramuscular injections for up to 30 days, which might result in higher nursing costs. This drug is currently not available in the U.S.
Biophysical Interventions
-
2CElectrical stimulation therapy: For recalcitrant VLUs, clinicians might opt for adjunctive electrical stimulation therapy, combined with standard care to promote ulcer healing if wound coverage with skin graft or cellular and/or tissue products is not an option (Grade 2C).
-
- Rationale: In electrical stimulation therapy/electroceutical treatment, microcurrents of electrical energy are applied to the ulcer area, in an attempt to replace the skin current, which is disrupted in chronic wounds.[180] Various studies have reported that electrical stimulation alleviates ulcer pain and may improve wound healing.[181][182] However, clinical guidelines do not fully agree that electrical stimulation should be recommended as an adjunctive therapy for non-healing VLUs [2][14][11] highlights that current evidence is not strong enough to support a recommendation to use electrical stimulation in VLU treatment.[2] Indeed, few studies focus solely on VLUs, and there is controversy regarding type of electrical stimulation, dosage, and duration of treatment.[14]
-
2CElectromagnetic therapy: We suggest against use of electromagnetic therapy in the treatment of VLU until further research can provide higher level of evidence (Grade 2C).
-
- Rationale: Electromagnetic therapy induces an electromagnetic field effect that is thought to attract electrically-charged cells that are important for wound healing. A Cochrane systematic review concluded that based on conflicting RCT results, at present it is not clear that electromagnetic therapy can aid in VLU healing.[183]
-
Ultrasound therapy: 2BWe suggest against use of ultrasound therapy as a primary therapy to promote healing of simple or complex VLU (Grade 2B) 2CClinicians may consider ultrasound therapy with compression to treat recalcitrant VLUs if wound coverage with skin graft or cellular and/or tissue products is not an option (Grade 2C).
-
- Rationale: There is at least moderate quality evidence that low dose, high frequency ultrasound is not more effective than standard care in treating VLU, and low quality evidence that low dose ultrasound is not more effective than standard care in treating VLU.[184] A few recent studies indicated that these modalities could be effective, however these studies had potential biases or were underpowered to detect statistical significance.[185][186] Ultrasound therapy has been delivered in different formats and duration, and standardized parameters have not been established. Other types of ultrasound therapy and dosing might prove to be effective in the future.
-
2CHyperbaric oxygen therapy: We suggest against use of hyperbaric oxygen therapy in the treatment of VLU (Grade 2C).
-
- Rationale: Currently, there is insufficient evidence to recommend hyperbaric oxygen therapy (HBOT) as an adjunctive therapy to promote healing of recalcitrant VLUs.[187] One small RCT showed that VLUs of the HBOT group healed significantly faster than ulcers of the control group by week 9, however this effect did not persist to 18 weeks.[188] Another RCT did demonstrate significantly faster healing rates in the HBOT group compared to the placebo group at 12 weeks however, it did not follow patients past that.[189] In addition, VLU is not an indication covered by Medicare or most insurance carriers.
-
Negative pressure wound therapy: 2CWe suggest against use of NPWT as primary therapy for VLU (Grade 2C). 2CClinicians might opt to use NPWT prior to and after skin grafting of non-healing VLU (Grade 2C).
-
- Rationale: NPWT collects excessive wound exudate and is thought to reduce the frequency of dressing changes and to promote healing by drawing together of wound edges, increasing perfusion, and removing infectious material and exudate.[190] However, currently there is not enough evidence to recommend NPWT as a primary treatment of VLU.[2][190] There is low certainty evidence that NPWT (compared with no NPWT) can help healing of VLU if used prior to and right after skin grafting. Medicare will cover NPWT for treatment of VLU if compression therapy and other types of standard care fail. Some adverse effects include wound maceration, retention of dressings, wound infection and other injuries. The device needs to be worn continuously by patients, and can interfere with mobility.[190]
- 2CClinical guidelines do not support use of other therapies such as whirlpool, laser therapy, phototherapy with UV light, or warming to treat VLU (Grade 2C).
- Rationale: Current evidence is not strong enough to support these interventions to promote healing or prevent recurrence of VLU.[2][11][14]
PREVENTION
Background
An active VLU is an advanced manifestation of chronic venous disease (CVD). According to the 2020 revised CEAP Classification, an active VLU is a C6 (see Table 4 below and section 'CEAP Classification System' in topic " Venous ulcers - Introduction and Assessment"). A VLU can be prevented by focusing on managing symptoms and/or signs of CVD in earlier stages.
A comprehensive assessment of the patient including history, risk factors and physical examination is crucial for development of a customized, patient-centric prevention plan. For an introduction and assessment of patients with CVD and venous VLU including epidemiology, risk factors, etiology, pathophysiology, history, physical examination, diagnosis, differential diagnoses, documentation and ICD-10 coding, see "Venous Ulcers - Introduction and Assessment". For clinical guidelines and quality measures specific to VLU, see "Venous Ulcers - Overview".
- The term CVD has been defined as “(any) morphological and functional abnormalities of the venous system of long duration manifest either by symptoms and/or signs indicating the need for investigation and/or care”.[191][18]
- The term “chronic venous insufficiency” (CVI) is reserved for advanced CVD, which is applied to functional abnormalities of the venous system, producing edema, skin changes, or venous ulcers, corresponding with C3 to C6 of the CEAP classification
Table 4. The 2020 revision of CEAP: Abbreviations and Descriptions of the Clinical Component of CEAP [192]
| CEAP Clinical Description |
- C0 No visible or palpable signs of venous disease
- C1 Telangiectasias or reticular veins
- C2 Varicose veins
- C2r Recurrent varicose veins
- C3 Edema
- C4 Changes in skin and subcutaneous tissue secondary to chronic venous disease
- C4a Pigmentation or eczema
- C4b Lipodermatosclerosis or atrophie blanche
- C4c Corona phlebectatica
- C5 Healed ulcer
- C6 Active venous ulcer
- C6r Recurrent active venous ulcer
|
Primary prevention - preventing first time VLU in patients with chronic venous disease
Conservative Management Strategy
The conservative management strategy of CVD is summarized below. Patients with symptomatic CVD with or without an active VLU may benefit from conservative treatment, whether they are not undergoing interventional treatment, awaiting intervention, or experiencing persistent symptoms after intervention.
- Compression therapy remains the cornerstone of conservative treatment for CVD. It has proven effective in controlling symptoms and reducing edema. It can also help reduce skin induration in limbs affected by lipodermatosclerosis and/or atrophie blanche (clinical CEAP class C4b). For details on compression therapy, see 'Transitioning to compression stockings' above, and topics "Compression Therapy" and "Gradient Compression Stockings for Patients with Venous Leg Ulcers".
- Emerging evidence highlights the significance of physical exercise and weight reduction in obese patients for reducing symptoms and preventing the progression of CVD.[18]
- An alternative conservative approach involves the use of venoactive drugs (e.g. pentoxifylline) to reduce symptoms and edema. When prescribing venoactive drugs, the specific therapeutic actions of the different available drugs should be considered. See section 'Oral systemic agents' and 'Systemic Pharmaceutical Agents' above.
- In clinical practice, conservative measures may be used alone or in combination, either temporarily or for a longer period, as part of a shared decision between practitioner and patient. This individualized approach will also depend on local circumstances and available healthcare resources.
Surgical management strategy
- For symptomatic patients with CVD, comprehensive duplex ultrasound is recommended to assess venous reflux and/or obstruction.[193][18]
- For symptomatic patients with CVD due to venous reflux and/or obstruction, consultation with a vascular surgeon is recommended.
- A summary of surgical management for CVD for prevention of VLU is provided below. For surgical management of patients with VLU, see section 'Vascular Surgical Interventions' above. For details on surgical indications, techniques, and post-operative care, see topic "Chronic Venous Disease - Surgical Management". For a review on venous anatomy, refer to the topic "Applied Anatomy and Physiology in Wound Care".
Interventions for superficial venous incompetence
Superficial venous incompetence is defined as reflux of superficial veins lasting > 0.5 seconds on duplex ultrasound. Clinical practice guidelines recommendations for CVD patients with incompetent superficial veins are listed below.[18]
For patients with superficial venous incompetence [18]:
- 1BPresenting with symptomatic varicose veins (CEAP clinical class C2S), interventional treatment is recommended (Grade 1B)
- 2CPresenting with edema (CEAP clinical class C3), other non-venous causes of edema should be considered before planning interventional treatment (Grade 2C).
- 1CPresenting with skin changes as a result of chronic venous disease (CEAP clinical class C4 - C6), interventional treatment of venous incompetence is recommended (Grade 1C).
- 1CUndergoing treatment using endovenous techniques with or without phlebectomies, procedures should be performed in the outpatient setting where possible (Grade 1C).
Interventions for deep venous pathology
Deep vein reflux and/or obstruction poses a significant burden on both patients and healthcare systems. Initial therapy should typically begin with non-invasive treatments, with compression therapy as the cornerstone. If there is no clinical improvement, invasive treatment options may be considered. An appropriate assessment, confirming clinically significant deep vein reflux and/or obstruction, is essential before initiating any treatment.[18]
The largest group of patients eligible for deep venous interventions are those suffering from post-thrombotic syndrome, which presents as a set of symptoms and signs of CVD, caused by impaired venous outflow due to deep venous obstruction and/or reflux following a DVT.[18]
Clinical practice guidelines are summarized below [18]:
Iliac vein outflow obstruction (proximal chronic total venous obstruction/severe stenosis)
- For patients with iliac vein outflow obstruction, management by a multidisciplinary team is recommended.
- 2CFor patients with iliac vein outflow obstruction without severe symptoms, neither endovascular nor surgical interventions are recommended (Grade 2C).
- 1CFor patients with iliac vein outflow obstruction and severe symptoms/signs, endovascular treatment should be considered, as the first choice treatment (Grade 1C). [18]
- 2CFor patients with iliac vein outflow obstruction suffering from a recalcitrant venous ulcer, severe post-thrombotic syndrome, or disabling venous claudication, surgical or hybrid deep venous reconstruction may be considered when endovascular options alone are not appropriate (Grade 2C).[18]
Deep venous incompetence of the lower limbs
- 2BFor patients with extensive axial deep venous incompetence and severe persistent symptoms and signs, where previous management has failed, surgical repair of valvular incompetence may be considered in specialized centers (Grade 2B).
Combined superficial and deep venous pathology
- 2CIn patients with chronic venous disease, caused by combined superficial and deep venous incompetence, treatment of incompetent superficial veins should be considered (Grade 2C).
Patient education
- Best practices related to education measures to prevent VLUs include regular exercise, leg elevation when at rest, careful skin care, weight control, and appropriately fitting foot wear.[2][11]
Prevention strategy by CEAP Classification
FOR PATIENTS WITH CEAP C1-C2
For patients with CEAP C3-C4
Patients with C3-4 disease may progress to VLU (C6), depending on their co-morbidities and lifestyle habits.[2]
- Exercise
- 2BFor patients with symptomatic chronic venous disease, exercise should be considered to reduce venous symptoms (Grade 2B).[18][194]
- Compression
- For patients with chronic venous disease who have never developed a VLU (C3-4), clinical guidelines suggest 20 to 30 mm Hg compression with knee high or thigh high gradient compression stockings. If the chronic venous disease was due to prior deep venous thrombosis, 30-40 mmHg compression with gradient compression stocking is recommended.[18]
- 1BFor patients with chronic venous disease and edema (CEAP clinical class C3), compression treatment, using below knee elastic compression stockings, inelastic bandages or adjustable compression garments, exerting a pressure of 20 – 40 mmHg at the ankle, is recommended to reduce edema (Grade 1B). [18]
- 1BFor patients with chronic venous disease and lipodermatosclerosis and/or atrophie blanche (CEAP clinical class C4b), using below knee gradient compression stockings, exerting a pressure of 20-40 mmHg at the ankle, is recommended to reduce skin induration (Grade 1B). [18]
- 2BFor patients with post-thrombotic syndrome, below knee gradient compression stockings, exerting a pressure of 20 – 40 mmHg at the ankle, should be considered to reduce severity (Grade 2B).[18]
- 2BFor patients with post-thrombotic syndrome, adjuvant intermittent pneumatic compression may be considered to reduce its severity, in addition to compression therapy 20-40 mm Hg (Grade 2B).[198]
- See topic "Gradient Compression Stockings for Patients with Venous Leg Ulcers".
- Oral, Systemic Pharmaceutical Agents
- 2BFor patients with symptomatic chronic venous disease, who are not undergoing interventional treatment, are awaiting intervention, or have persisting symptoms and/or edema after intervention, medical treatment with venoactive drugs (e.g. pentoxifylline) should be considered to reduce venous symptoms and edema, based on the available evidence for each individual drug (Grade 2B).[18][18][195][196][197]
- Surgical interventions
- For patients with with CEAP C3-C4 with superficial venous incompetence [18]:
- 2CPresenting with edema (CEAP clinical class C3), other non-venous causes of edema should be considered before planning interventional treatment (Grade 2C).
- 1CPresenting with skin changes as a result of chronic venous disease (CEAP clinical class C4 - C6), interventional treatment of venous incompetence is recommended (Grade 1C).
- For patients with CEAP C4 with deep venous pathology, see Table 3 'Vascular Surgical Interventions' above, which summarizes recommendations by the Society for Vascular Surgery and American Vascular Forum (SVS) and the European Society for Vascular Surgery (ESVS).[2][18]
FOR PATIENTS WITH CEAP C5 (healed VLU)
- See section 'Prevention of VLU recurrence' below.
Prevention of VLU recurrence
For the majority of VLU patients, surgical correction of superficial venous incompetence and/or underlying deep venous pathology are essential tools to prevent VLU recurrence (see section 'Vascular Surgical Interventions' above).[18] Alternatively, a conservative approach, consisting of compression therapy using below knee gradient compression stockings may be useful to prevent ulcer recurrence.[18]
Conservative Management Strategy
- Recommendations include: compression therapy with gradient compression stockings at the highest pressure patients can tolerate [2][5][11][27], moisturizer to prevent skin breakdown [11], supervised calf muscle exercise [2][11][56] and smoking cessation.[11] Current evidence shows that compared with usual care, gradient compression stockings are effective in preventing venous leg ulcer recurrence and likely to be cost-effective. In people with a healed VLU, wearing compression stockings helps to reduce the risk of recurrence by about half.[199] For simple VLUs, gradient compression stockings can be ordered while ulcer is active but complete healing is expected soon (Medicare covers gradient compression stockings only when VLU is active). For complex VLUs, consult edema management specialist. See section 'Transitioning to Stockings' in "Compression Therapy" above.
- Leg elevation can be suggested, although current evidence to support this intervention in prevention of VLU recurrence is of low quality.[56]
- Community-based clinics to promote adherence to compression therapy or prevent recurrence can be suggested to patients, although evidence on the effectiveness of such clinics is still unclear.[27]
Surgical Management Strategy
- Consultation with a vascular surgeon for consideration of surgical vascular procedures to prevent recurrence of an active (C6) or healed VLU (C5) is recommended (e.g. ablation of superficial incompetent veins for superficial reflux).[2] See vascular interventions in section 'Vascular Surgical Interventions' above.
INTERVENTIONS FOR SUPERFICIAL VENOUS INCOMPETENCE
INTERVENTIONS FOR DEEP VENOUS PATHOLOGY
1CFor patients with active (C6) or healed venous leg ulceration (C5) and iliac vein outflow obstruction, venous stenting should be considered (Grade 1C).[18]
PATIENT EDUCATION - FOR CLINICIANS
VLU is a chronic, long-term problem, with recurrence rate are as high as 70%. Therefore, long-term maintenance must be addressed even for healed ulcers.[14] Patient adherence to treatment can be problematic, but patient education on the importance of long-term compression and lifestyle changes can help improve adherence.[31] We recommend sharing our patient education materials with VLU patients.
-
Gradient Compression Stockings for Patients
- Patient Education - Venous Leg Ulcer
- Other relevant handouts will be available soon
Patient education to prevent VLU for the first time
- For patients with chronic venous disease who have never developed a VLU (C1-4), clinical guidelines suggest regular exercise, leg elevation when at rest, careful skin care, weight control, and appropriately fitting foot wear[2][11].
- For patients with chronic venous disease who have never developed a VLU and have varicose veins and swelling or venous stasis skin changes (C3-4) due to primary valvular reflux, guidelines suggest compression compression, 20 to 30 mm Hg, knee or thigh high[2].
Patient education for VLU healing
- Compression, supervised calf muscle exercise, smoking cessation and adequate nutrition are recommended
Patient education to prevent VLU recurrence
- Compression therapy with gradient compression stockings at the highest pressure patients can tolerate [5][2][11][27], moisturizer to prevent skin breakdown [11], supervised calf muscle exercise [2][11][56], smoking cessation [11]
- Leg elevation can be suggested, although current evidence to support this intervention in prevention of VLU recurrence is of low quality [56].
- Community-based clinics to promote adherence to compression therapy or prevent recurrence can be suggested to patients, although evidence on the effectiveness of such clinics is still unclear [27].
APPENDIX
Summary of Evidence (SOE)
SOE – Cleansers
(Back to text)
- A 2021 Cochrane systematic review evaluated four studies with a total of 254 participants. All studies included comparisons between different types of cleansing solutions. Authors concluded that currently, there is insufficient evidence to demonstrate whether the use of 0.2% polyhexamethylene biguanide (PHMB) solution compared with saline solution; aqueous oxygen peroxide compared with sterile water; propyl betaine and polihexanide compared with a saline solution; or octenidine dihydrochloride/phenoxyethanol (OHP) compared with Ringer's solution makes any difference in the treatment of VLUs.[90]
- The 2006 Wound Healing Society guideline, updated in 2015, suggested that wounds be cleansed with a neutral, nonirritating, nontoxic solution such as sterile saline or water initially and at each dressing change, and that routine wound cleansing be accomplished with a minimum of chemical and/or mechanical trauma. Their suggestion was based on low-quality evidence (Evidence level C) from on 1 literature review, 1 clinical case series, and 2 experimental studies, and based on principles that irrigating and cleansing the wound removes loose impediments to wound healing [14][86]
- The 2010 AAWC guideline recommends that VU be cleansed gently at low pressure (4-15psi) with safe, non-antimicrobial cleanser, based on 1 RCT at high risk of bias/imprecision, 1 literature review, and 1 study that validated the clinical efficacy and cost effectiveness of multidisciplinary guidelines for the diagnosis and treatment of venous leg ulcer, and was not specific to cleansers in wound care. Although authors classified evidence as high-quality (evidence level A), according to the GRADE methodology, we would classify evidence as low-quality (evidence level C) due to high risk of bias/ imprecision. [11]
- The 2014 American Society for Vascular Surgery guideline suggested that venous leg ulcers be cleansed initially and at each dressing change with a neutral, nonirritating, nontoxic solution, performed with a minimum of chemical or mechanical trauma (Grade 2C). Their suggestion was based on low-quality evidence (Evidence level C), from on 1 literature review, 1 RCT with high risk of bias and imprecision, and 1 clinical case series, and assuming that most patients with VLUs present with significant wound exudate and other debris in and around the wound area that must be cleansed routinely before dressing application.[2]
SOE – Debridement
(Back to text)
- A 2015 Cochrane systematic review that evaluated 10 RCT (715 participants) recognized that there is consensus in the wound care literature that debridement is necessary to promote wound healing [85] [91] [23], however authors found only minimal evidence to support active debridement of VLU to promote wound healing. Most studies had unclear risk of bias or were at high risk of bias. The smallness of the evidence base means that one cannot conclude with confidence that debridement improves healing, or which method of debridement, or duration of debridement confers most benefit in the healing of venous ulcers. RCTs that met inclusion criteria in this review evaluated only autolytic and enzymatic debridement methods, and none of the included RCTs evaluated surgical, sharp or mechanical methods of debridement, or debridement versus no debridement [92].
- The 2014 Society for Vascular Surgery[2], the 2015 Wound Healing Society [14][86], and the 2010 Association for Advancement of Wound Care (AAWC) [11] guidelines support VLU debridement of VLU to promote wound healing, although the levels of evidence assigned by these societies differed from each other, possibly due to different evidence grading methodologies and different studies from which evidence was drawn (SVS uses GRADE framework).
- A 2011 analysis of 14 guidelines for VLU found that debridement was suggested in 86% of the guidelines, but only one-third suggested at the strong level [23].
|
Intervention for VLU
|
Society for Vascular Surgery, 2014
|
AAWC, 2010
|
Wound Healing Society, 2015
|
|
VLU debridement at initial evaluation vs. no debridement
|
Grade 1B (strong recommendation, moderate quality evidence)
|
A (high quality evidence)
|
Level II (moderate quality evidence)
|
|
Additional maintenance debridement of VLU
|
Grade 2B (weak recommendation, moderate quality evidence)
|
n/a
|
Level II (moderate quality evidence)
|
|
Surgical debridement for VLU with slough, nonviable tissue, or eschar.
|
Grade 1B (strong recommendation, moderate quality evidence)
|
C2
|
n/a
|
|
Hydrosurgical debridement
|
Grade 2B (weak suggestion, moderate quality evidence, as an alternative to surgical debridement)
|
n/a
|
n/a
|
|
Ultrasonic debridement
|
Grade 2C (weak suggestion not to use intervention vs. surgical debridement, low quality evidence)
|
A (high quality evidence) for high-frequency
|
n/a
|
|
Enzymatic Debridement
|
Grade 2C (weak suggestion, low quality evidence, as an alternative to surgical debridement)
|
A (high quality evidence)
|
n/a
|
|
Biological debridement
|
Grade 2B ((weak suggestion, moderate quality evidence, as an alternative to surgical debridement)
|
A (high quality evidence)
|
n/a
|
|
Mechanical debridement
|
n/a
|
C (low quality evidence)
|
n/a
|
|
Autolytic
|
n/a
|
A (high quality evidence)
|
n/a
|
- We reviewed the Cochrane systematic review and the studies that the three guidelines used to grade quality of evidence and strength of recommendation (when applicable) for each of intervention above. Applying the GRADE framework to the combined body of evidence, we found that:
-
- Evidence that support the recommendation to perform debridement at initial evaluation and for maintenance to promote VU healing compared to no debridement was of low quality (evidence level C), provided mainly by observational studies. Conclusions of other studies cited by guidelines were not directly relevant in supporting this intervention in VU, as they primarily compared effectiveness of different debridement methods, or assessed effectiveness of debridement in other types of wounds (e.g., pressure injuries, diabetic foot ulcers), which may have a different response to debridement compared to VU. Other studies were not directly relevant, as they did not evaluate effect of debridement as an intervention in VU healing as an outcome.
- Current studies do not provide sufficient evidence to conclude with confidence which method of debridement confers most benefit in the healing of venous ulcers. Studies that concluded that one method was superior than other had important methodological limitations (level C). Other studies included in the guidelines were not directly relevant, as they compared effectiveness of methods of debridement in other types of wounds (e.g, pressure ulcers), or did not evaluate VU healing as an outcome.
SOE – Exudate management, moist wound bed
(Back to Text)
- The 2014 Society for Vascular Surgery (SVS)[2], the 2015 Wound Healing Society (WHS)[14][86], and the 2010 Association for Advancement of Wound Care (AAWC)[87] guidelines support managing VLU exudate and maintaining a moist wound bed, although the levels of evidence assigned by these societies differed from each other, possibly due to different evidence grading methodologies and different studies from which evidence was drawn (SVS uses GRADE framework).
|
Intervention
for VLU
|
Society for Vascular Surgery, 2014
|
Wound Healing Society, 2015
|
AAWC, 2010
|
|
Manage venous leg ulcer exudate and protect peri-wound skin
|
Grade 2B (weak recommendation, moderate quality evidence)
|
Level I (high quality evidence)
|
A (high quality evidence)
|
|
Maintain a moist wound bed
|
Grade 2C (weak recommendation, low quality evidence)
|
Level I (high quality evidence)
|
A (high quality evidence)
|
- We reviewed the studies that the three guidelines used to grade quality of evidence and strength of recommendation for each of intervention above and found that:
-
- The recommendation to maintain a moist wound bed is supported by moderate quality evidence (evidence level B), provided mainly by experimental studies that demonstrated that moist environments promote faster wound healing. Conclusions of other studies cited by guidelines were not directly relevant in supporting this intervention, as they primarily compared effectiveness of different dressings in VU healing, and did not necessarily evaluate effect of moist vs. non-moist environment in wound healing.
- The recommendation to manage venous leg ulcer exudate and protect peri-wound skin is supported by moderate quality evidence (evidence level B), provided mainly by experimental studies that demonstrated that exudate has higher levels of MMP and suggested that the presence of excessive levels of MMP at the wound surface of pressure ulcers may impede the healing of these wounds. Conclusions of other studies cited by guidelines were not directly relevant in supporting this intervention, as they primarily compared effectiveness of different dressings in VU healing, and did not necessarily evaluate effect of VLU exudate and MMP in wound healing and peri-wound skin integrity.
SOE - Topical Dressings
(Back to text)
Alginate: Low-quality evidence (Evidence level C due to high risk of bias) suggests that when associated with compression, alginate dressings, hydrocolloid and plain non-adherent dressings are equally effective in healing venous ulcers
- A 2016 systematic review evaluated the same RCTs (295 participants) and arrived at the same conclusion as the authors of the Cochrane review. [200]
- A 2015 Cochrane systematic review that evaluated 5 RCTs (295 participants). All studies were industry funded. 1 compared different brands of alginate, 3 compared alginate with hydrocolloid, 1 with plain non-adherent dressing. Most participants received the dressing and four-layer bandage. No statistically significant between-group differences were detected for any comparison, for any healing outcome. Meta-analysis was feasible for one comparison (alginate and hydrocolloid dressings) with data from two RCTs (84 participants) pooled for complete healing at six weeks: risk ratio 0.42 (95% confidence interval 0.14 to 1.21). Adverse event profiles were generally similar between groups (not assessed for alginate versus plain non-adherent dressings. Authors concluded that the current evidence base is of low quality, mainly due to mostly uncertain risk of bias. Evidence does not suggest that alginate dressings are more or less effective in the healing of venous leg ulcers than hydrocolloid or plain non-adherent dressings, and there is no evidence to indicate a difference between different proprietary alginate dressings. It is possible that dressing performance, in terms of ease of removal, is better for hydrocolloid dressings than alginate. [201]
Hydrogel: Low-quality evidence (Evidence level C due to high risk of bias) suggests that when associated with compression, low-adherent dressings, different brands of hydrogel or miscellaneous dressings are equally effective and present no difference in adverse effects in healing venous ulcers
- 2 systematic reviews identified 5 RCTs (379 participants) that compared hydrogel dressings with either low adherent dressings (2 RCTs), another brand of hydrogel (2 RCTs) and miscellaneous dressings (1 RCT). Evidence from RCTs was of low or uncertain quality due to risk of bias, but in none of the comparisons was there evidence that any one dressing type was better than others in terms of number of ulcers healed. [154]
Foam: Low-quality evidence (Evidence level C, due to high risk of bias) suggests that when associated with compression, foam dressings, hydrocolloid, paraffin gauze, hydrocapillary, knitted viscose, and protease modulating matrix dressings are equally effective and present no difference in adverse effects in healing venous ulcers
- A 2016 systematic review concluded that the venous leg ulcer pairwise comparisons of alginate vs. low-adherent dressings, alginate vs. hydrocolloid, alginate vs. hydrofiber, foam vs. low-adherent dressings, foam vs. hydrocolloid, and hydrocolloid vs. low-adherent dressings included in our meta-analysis revealed moderate-quality level B evidence that there were no statistically significant differences in ulcer healing efficacies between the two interventions examined in each comparison [200]
- A 2014 AHRQ systematic review also concluded that based on the low quality evidence available, there are no data to support superiority of specific dressings in venous ulcer healing or adverse effects [19]
- A 2013 Cochrane systematic review that evaluated 12 RCTs (1023 participants) that compared foam to another dressing found no statistically significant difference in wound healing or adverse events for any comparison. [202]Studies: 3 RCTs compared hydrocellular foam dressings and polyurethane foam dressings. 5 RCTs compared foam dressings and hydrocolloid dressings. 2 compared foam dressings with paraffin gauze, 1 with hydrocapillary dressing, 1 with knitted viscose dressing, and 1 with protease modulating matrix. Most participants received the dressing and four-layer bandage. Most studies were industry funded. Authors concluded that at present there is no evidence to suggest that foam dressings are better or worse than any other primary wound contact dressing for the healing of venous leg ulcers when applied beneath compression devices. However, the current evidence base is mainly of low quality, mainly due to high/uncertain risk of bias.
Hydrocolloid: Low-quality evidence (Evidence level C, due to high risk of bias) suggests that when associated with compression and compared to non-adherent dressings, foam, alginate, different brands of hydrocolloid, gauze, lyophilized collagen dressing and magnesium sulfate paste and gauze, hydrocolloid dressings showed equivalent dressing effectiveness in promoting complete ulcer healing.
- A 2011 review of systematic reviews analyzed 3 systematic reviews (search date 1997, 16 RCTs; search date 2003, 15 RCTs; and search date 2006, 27 RCTs). The first systematic review identified 9 RCTs, the second review identified 8 RCTs, and the third review identified 9 RCTs comparing hydrocolloid dressings versus simple dressings in the presence of compression. Conclusion: Compared with simple dressings Hydrocolloid dressings are no more effective than simple low-adherent dressings at increasing ulcer healing rates in people receiving compression (author classifies evidence of high-quality evidence). However underlying systematic reviews describe quality of evidence as uncertain or low. [113]
- 2016 systematic review: 14 RCTs. 5 RCTs vs. foam (417 participants, 95% CI 1.00 (0.81, 1.22), 8 RCTs vs. low-adherent dressings (746 participants, 95% CI 1.15(1.00, 1.33). 1 RCT vs. alginate (40 participants, 95% CI 0.61, 0.13-2.96) There were no statistically significant differences in ulcer healing efficacies between the two interventions examined in each comparison. Evidence of moderate quality due to high risk of bias. [200]
- 2007 systematic review: 22 RCT - 8 RCT vs non-adherent dressing (792 participants, RR=0.98, 0.85 to 1.12), 4 RCT vs foam (311 participants, RR=0.98 (0.79 to 1.22), 2 RCT vs. alginate (80 participants, RR0.72 (0.48 to 1.69), 3 RCTs vs hydrocolloid (98 participants, RR=1.56, 0.67 to 3.63), 2 RCTs vs hydrogel (237 participant, no meta-analysis), 1 RCT vs gauze (28 participants, no meta-analysis), 1 RCT vs lyophilized collagen dressing (93 participants), 1 RCT vs magnesium sulfate paste and gauze (110 participants). No statistically significant difference in ulcer healing and adverse effects. Evidence was of low or uncertain quality due to risk of bias mainly. [154]
- A 2015 systematic review of cost-effectiveness analyses identified 1 study (lower-quality, Drummond score < 8) that compared hydrocolloid and Vaseline gauze dressing associated with compression in treatment of venous ulcers and found that hydrocolloid was more cost-effective than Vaseline gauze.
SOE – Wet-to-dry
(Back to text)
WHS recommends against using wet-to-dry dressings to promote wound healing (Level I) [14][86]. Among the studies analyzed, there were 2 meta-analyses that showed no significant differences in terms of the proportion of ulcers healed or reduction in wound size in VLU treated with different types of dressings, including continuously moist saline gauze dressings. Authors of the meta-analyses highlighted the fact that studies had important methodological limitations. In reviewing the studies included in the meta-analyses, we confirmed methodological limitations, and in light of the GRADE framework would consider it of low/uncertain quality (evidence level C).
SOE – Topic antimicrobial agents
(Back to text)
- Cadexomer iodine: A systematic meta-analysis of 4 trials that compared cadexomer iodine with standard care in VU patients receiving compression indicated cadexomer iodine is more effective in promoting VU healing (RR 2.17, 95% CI 1.30 to 3.60) [106]. However, trials had a small sample size, were at high risk or bias or had unclear risk of bias with limitations that might affect the magnitude of effect of the intervention (evidence level C). No difference in VU healing was observed when cadexomer iodine was compared with hydrocolloid dressing, paraffin gauze dressing, dextranomer or silver-impregnated dressings.
- Silver-based preparations: A systematic review of 12 RCTs, found no statistically significant difference in VU healing in patients treated with silver preparations compared with usual care, placebo, growth factor, non-adherent dressing, and other antimicrobial dressings [106]. One RCT was large and at low risk of bias, others were small and at unclear risk of bias.
- Honey: A meta-analysis of 2 RCT found no statistically significant difference in VU healing in VU treated with honey-based preparations and hydrogel or other non-honey dressings of clinicians’ choice [106]. One RCT was large and at low risk of bias, the other was small and at high risk of bias. The same RCTs were evaluated by another meta-analysis that yielded same results. Evidence was considered of low quality (evidence level C), mainly due to high risk of bias that can affect results of the meta-analysis and imprecision [111].
SOE – Systemic Antibiotics
(Back to text)
- A 2014 Cochrane systematic review that evaluated 5 RCTs (233 participants) that reported 8 comparisons of systemic antibiotics (co-trimoxazole, gentamicin, amikacin, ciprofloxacin, trimethoprim, amoxicillin and levamisole) versus placebo, usual care or another antibiotic. In 1 RCT, more participants healed on levamisole compared to placebo (RR 1.31, 95% CI 1.06 to 1.62). However, the trial had an unclear risk of bias - did not state whether VU were infected or not at baseline, and had a small sample size – results could have happened by chance (evidence level C). Levamisole is unlicensed in the UK, and is only available from ’special order’ suppliers for use in treating roundworm infection, and it was withdrawn from the US market in 1999. Authors of the review concluded that at present, no evidence is available to support the routine use of systemic antibiotics in promoting healing of clinically infected venous leg ulcers. However, the studies had an unclear risk of bias with potential limitations that can affect the results of the studies and sample size was small (evidence level C). Therefore, authors could not recommend the discontinuation of any of the agents reviewed. In light of the increasing problem of bacterial resistance to antibiotics, current prescribing guidelines recommend that antibacterial preparations should be used only in cases of clinical infection, not for bacterial colonization [106].
- Clinical guidelines agree that routine culture of the ulcer should not be done. Culture (preferably quantitative) should be employed only when there was evidence of active infection [23]
- The 2014 Society for Vascular Surgery [2], the 2015 Wound Healing Society (28,35), and the 2010 Association for Advancement of Wound Care [87] guidelines support treatment of VU with clinical signs of infection and with more than 1 x 10^6 CFU/g of tissue with antimicrobial therapy, although the levels of evidence assigned by these societies differed from each other, possibly due to different evidence grading methodologies and different studies from which evidence was drawn (SVS uses GRADE framework).
|
Intervention for VLU
|
Society for Vascular Surgery, 2014
|
AAWC, 2010
|
Wound healing Society, 2015
|
|
Treatment of VU with >1 x 10^6 CFU/g of tissue and clinical evidence of infection with systemic antimicrobial therapy
|
Grade 2C (weak suggestion, low quality evidence)
|
A (high quality evidence)
|
Level II (moderate quality evidence)
|
- We reviewed the Cochrane systematic review and the studies that the three guidelines used to grade quality of evidence and strength of recommendation (when applicable) for each of intervention above. Applying the GRADE framework to the combined body of evidence, we found that:
-
- Evidence that support the recommendation to treat VU with >1 x 10^6 CFU/g of tissue and clinical evidence of infection with systemic antimicrobial therapy compared with no systemic antimicrobial therapy was of low quality (evidence level C). Authors of the 2014 Cochrane systematic review concluded that at present, no evidence is available to support the routine use of systemic antibiotics in promoting healing of clinically infected venous leg ulcers. However, the studies had an unclear risk of bias with potential limitations that can affect the results of the studies and sample size was small (evidence level C). Conclusions of other studies cited by guidelines were not directly relevant in supporting this intervention in infected VU, as they compared effectiveness of topical antimicrobial agents or did not evaluate effectiveness of systemic antibiotics in VU healing
SOE – Stasis dermatitis
(Back to text)
- A 2017 Cochrane systematic review on eczema (not stasis dermatitis specifically, but stasis dermatitis is a form of eczema) evaluated 77 RCT (6603 participants) and concluded that most moisturizers showed some beneficial effects, producing better results when used with active treatment, prolonging time to flare, and reducing the number of flares and amount of topical corticosteroids needed to achieve similar reductions in eczema severity. Authors not find reliable evidence that one moisturizer is better than another [121].
SOE – Compression
(back to text)
- Authors of a 2021 Cochrane systematic review found that there is moderate‐certainty evidence (downgraded once for risk of bias) that [25]:
- 1. There is a shorter time to complete healing of venous leg ulcers in people wearing compression bandages or stockings compared with those not wearing compression (pooled hazard ratio for time‐to‐complete healing 2.17, 95% confidence interval (CI) 1.52 to 3.10; I2 = 59%; 5 studies, 733 participants);
- 2. People treated using compression bandages or stockings are more likely to experience complete ulcer healing within 12 months compared with people with no compression (10 studies, 1215 participants);
- 3. People using compression bandages or stockings probably have a lower mean pain score than those not using compression (four studies with 859 participants and another study with 69 ulcers): pooled mean difference −1.39, 95% CI −1.79 to −0.98; I2 = 65%
- 4. Compression bandages or stockings versus no compression may improve disease‐specific quality of life, but not all aspects of general health status during the follow‐up of 12 weeks to 12 months (four studies with 859 participants; low‐certainty evidence).
- Authors of a 2012 Cochrane systematic review concluded that compression increases ulcer healing rates compared with no compression, on the basis of 8 RCT at unclear or high risk of bias [7]. Authors of the review believe overall potential limitations of the studies were likely to lower confidence in the estimate of effect mainly due to uncertainty in whether outcome assessment was blinded (evidence level B).
- A more recent 2014 systematic review also found moderate level evidence that compression is better than no compression in promoting VU healing [41].
- No robust RCTs with results published after 2014 that compared compression vs. no compression for patients with VU have been found.
- Guidelines: All 14 guidelines for venous ulcer evaluated by O’Donnell in 2011 recommend compression for VU healing, and most recommend it at the strongest recommended level [23].
- The 2014 Society for Vascular Surgery (SVS)[2], the 2015 Wound Healing Society (WHS)[14][86], and the 2010 Association for Advancement of Wound Care (AAWC)[87] guidelines recommend compression therapy over no compression to promote VU healing.
SOE – Types of Compression
(Back to text)
Systematic Reviews and Meta-analyses
- According to the 2012 Cochrane systematic review on the topic, there is evidence that multi-component compression systems are more effective than single-component systems. Most trials that evaluated this comparison were small and at unclear or high risk of bias and did not find a statistically significant difference in outcome, but one was large and at unclear risk of bias, and found that multi-component system was more effective than single-component [7]. Authors of the review believe that overall potential limitations of the large study are somewhat likely to lower confidence in the estimate of effect mainly due to lack of blinding of outcome assessment (evidence level B). The review also concluded that multi-component systems containing an elastic bandage appear to be more effective than those composed mainly of inelastic constituents, based on 5RCTs pooled data. 4 RCTs were small and at high or unclear risk of bias due to non-blinded outcome assessment, however 1 RCT was large and at low risk of bias. The pooled data of individual patient data or IPD (which allows a more sensitive analysis compared to aggregate data) showed at a statistically significant level that multi-component systems (4 layers bandages, or 4LB) with elastic bandage are more effective than multi-component systems that include an inelastic bandage (short stretch bandage, or SSB). However, authors recognize that staff familiarity with certain compression systems can greatly influence their effectiveness. The difference between 4LB and short-stretch bandages (SSB) in the IPD meta-analysis could be partly explained by skill and experience of bandagers; 3 out of 5 trials (75% of participants) were conducted in the UK, where 4LB is standard [7].
- A more recent 2014 systematic review and meta-analysis [203] performed with aggregate data was unable to identify superiority in VLU healing of one approach in the three comparisons of interest: compression stockings vs compression bandages, 4LB systems vs systems that contain less than four layers, and SSBs vs. long-stretch bandages (LSBs - include 4LB and other elastic systems). It is important to note that the categories of compression compared by this study are different than the ones in the Cochrane review [7] and thus the group of trials representing each comparison differed as well. For instance, even though trials comparing SSB and 4LB are included in the assessment of SSB vs. LSB, the overall result of the meta-analysis is different because other studies that compare other types of LSB and SSB are included in the group of trials. Also, this study included 2 more recent trials that did not identify difference in VLU healing between 4LB and SSB. One of these trials [204] is a large RCT with low risk of bias. Ultimately, authors of this study agreed that: there is at least moderate-quality evidence that compression is superior to no compression and should be the mainstay of treatment for venous ulcers of the lower extremities, that multicomponent systems are superior to single component systems, and that an elastic component is needed [41].
- Another systematic review and meta-analysis published in 2014 (Nelson et al) compared 4LB and SSB and found no difference in their effectiveness in VLU healing. This study included a recent trial conducted by the Canadian Bandage Trial team to the body of evidence analyzed by O’Meara in the Cochrane meta-analysis on compression therapy [7]. This RCT had 428 participants and was considered of low risk.
Randomized Controlled Trials
- A 2019 RCT (n=66 patients) compared use of adjustable compression wrap (CircAid JuxtaCure) (n=33) and inelastic bandage (Coban 2 Layer ) ( n=33) on VLU healing.[51] Study duration was 12 weeks. Authors concluded that adjustable compression wraps are significantly cheaper and more effective (not significantly) in achieving VLU healing and that self-applicable, adjustable compression wraps are therefore a powerful, cost-effective alternative to inelastic bandages in treating VLU. Evidence can be considered of low certainty (level C), as assessors/patients were not blinded and sample size was small. Manufacturer of the adjustable compression wrap provided all compression material, dressings, and measuring devices.
Clinical guidelines
- Out of 14 clinical guidelines, four guidelines advocated four-layer bandages and three short stretch garments [23].
- Interventions related to compression therapy for VLU recommended by The 2014 Society for Vascular Surgery (SVS)[2], the 2015 Wound Healing Society (WHS)(28,35), and the 2010 Association for Advancement of Wound Care (AAWC)[87] are summarized below.
|
Intervention for VLU
|
Society for Vascular Surgery
|
AAWC
|
Wound healing Society
|
|
Use compression therapy to promote healing in patients with venous ulcers
|
1A
|
A
|
I
|
|
Use high compression levels (e.g. 40 mmHg) at the ankle level to promote ulcer healing. High strength compression can be applied successfully using many methods including multilayered elastic compression, Unna's boot, compression stockings, and other
|
n/a
|
A
|
I
|
|
Choose multicomponent compression bandage over single-component bandages for the treatment of venous leg ulcers.
|
Grade 2B
|
A
|
n/a
|
|
Choose elastic compression bandage as it heals more than inelastic compression
|
n/a
|
A
|
n/a
|
|
Two-layer compression improves comfort or quality of life more than 4-layer or short stretch
|
n/a
|
A
|
n/a
|
|
Elastic compression stockings with moisture retaining dressing improve VU healing, pain and application time compared to short-stretch compression bandages or Unna’s Boot
|
n/a
|
A
|
n/a
|
|
intermittent pneumatic compression when other compression options are not available, cannot be used, or have failed to aid in venous leg ulcer healing after prolonged compression therapy.
|
Grade 2C
|
n/a
|
n/a
|
SOE – Compression to prevent recurrence
(Back to Text)
- A 2024 Cochrane systematic review summarized 8 RCTs (1995 participants) that evaluated compression in patients with healed ulcers. There is some evidence that compression hosiery might prevent ulcers, but the evidence is of low quality, due to risk of bias (lack of blinding), imprecision, and indirectness. Compression with EU class 3 compression stockings may reduce reulceration compared with no compression over six months. Use of EU class 1 compression stockings compared with EU class 2 compression stockings may result in little or no difference in reulceration and noncompliance over 12 months. UK class 3 compression hosiery may reduce reulceration compared with UK class 2 compression hosiery; however, higher compression may lead to lower compliance. There may be little to no difference between Scholl and Medi UK class 2 compression stockings in terms of reulceration and noncompliance. There is insufficient evidence to aid selection of different types, brands, or lengths of compression hosiery.[52]
- A 2014 systematic review concluded that currently low-quality evidence (based on imprecision and heterogeneity) supports the effect of compression on ulcer recurrence [41]
- All but one of the 14 clinical guidelines for VLU analyzed by O’Donnell in 2011 recommended below-knee 20/30-mm Hg compression stockings to prevent ulcer recurrence with a strong grade of recommendation in seven [23]
- The 2014 Society for Vascular Surgery (SVS)[2], the 2015 Wound Healing Society (WHS)[14][86], and the 2010 Association for Advancement of Wound Care (AAWC)[87] guidelines recommend compression therapy over no compression to reduce risk of VLU recurrence after VLU has healed.
|
Intervention for VLU
|
Society for Vascular Surgery, 2014
|
AAWC, 2012
|
Wound healing Society, 2015
|
|
In a patient with a healed venous leg ulcer, we suggest compression therapy to decrease the risk of ulcer recurrence.
|
Grade 2B
|
A
|
Level I
|
- New RCTs since 2014 evaluating use of compression to prevent VLU recurrence were found
SOE – Exercise
(Back to Text)
- A 2015 review on the effect of supervised exercise on healing and quality of life of VLU patients found 10 articles (RCTs, cohort studies with small sample sizes) that used physical therapy or exercise for patients with open or healed VLUs. They found that although there is evidence that exercise strengthens the calf muscle pump and improves ankle ROM, few studies have investigated the effect of these interventions on QOL and healing, and few involved the supervision of a physical therapist. Authors concluded that there is currently a lack of evidence that physical therapy-oriented exercise has an effect on wound healing, QOL and ulcer recurrence. [54]
- A 2014 review on VLU treatment has found low level of evidence (level C) that support a structured program of calf muscle exercise may improve hemodynamic performance and prevent ulcer recurrence [65]. A physical therapist can be instrumental in helping the patient adhere to this care plan.
- Clinical guidelines support exercises to increase calf muscle pump function [2][14][53]. The Wound Healing Society guideline states that this intervention has been demonstrated to be helpful in VLU prevention (level III of evidence) and the Society for Vascular Surgery (SVS) suggests supervised exercise to reduce pain and edema in patients with active VLU (Grade 2B). Of note, studies cited by SVS evaluated effect of exercise on strengthening of calf muscle pump in patients with VLU but did not correlate it with VLU healing or prevention of recurrence [205] [206].
- A small RCT (Meagher, 2012) evaluated VLU healing in 40 participants with newly diagnosed VLU in randomized in control group and supervised exercise group. Participants who took more steps per day showed faster venous ulcer healing times when compared with those who took fewer steps (p<0.005) Trial is small and underpowered to detect statistical differences, unclear if outcome assessment was blinded (level C) [207].
SOE – Leg elevation
(back to text)
- There are no published clinical trials on effects of leg elevation on VLU healing or recurrence.
- In 1994, Abu-Own et al conducted a small study and concluded that limb elevation enhanced the microcirculatory flow velocity in liposclerotic skin of patients with chronic venous insufficiency [57]
- In 2004, Xia et al evaluated 10 patients with an echographic image analysis and concluded that Leg elevation is extremely effective in reducing edema, even if only for three to four hours [58]
- In a 2010 review, Collins & Seraj assigned level C to leg elevation to minimize edema in patients with chronic venous insufficiency and as adjunctive therapy for VLU and cited Abu-Own et al. as a reference [59]
- The 2012 Association for Advancement of Wound Care guideline assigned level C1 of evidence to leg elevation for VLU healing and prevention of recurrence, and cited the references above [53]
SOE – Pentoxifylline
(Back to Text)
- A 2012 Cochrane systematic review included 12 RCTs (864 participants) comparing pentoxifylline and compression with placebo or no treatment in people with VLU. Authors concluded that pentoxifylline 400 mg tablet taken three times a day, is an effective adjunct to compression bandaging for treating venous ulcers and may be effective in the absence of compression. The majority of adverse effects were gastrointestinal disturbances, and were tolerated by participants. Quality of trials was variable (Evidence level B) [64]
- A 2021 meta-analysis with 13 RCTs (n=921 individuals) found that compared with placebo, pentoxifylline significantly improved the ulcer healing rate (RR = 1.59, 95%CI 1.22 to 2.07, P < .001) while increased the incidence of gastrointestinal disturbances (RR = 2.29, 95%CI 1.04 to 5.03, P = .04) at the same time. Moreover, pentoxifylline also shortened the mean duration of complete wound healing (P = .007) and shrank ulcer size (P = .02). Currently available evidence suggests that pentoxifylline could help venous leg ulcers heal more quickly and effectively.[17]
SOE - Vascular Surgical Interventions
(Back to Text)
Systematic reviews and meta-analyses
- A 2023 Cochrane systematic review included 2 RCTs to determine the effects of superficial endovenous ablation on the healing and recurrence of venous leg ulcers and the quality of life of people with venous ulcer disease. Authors found that both trials randomised participants to endovenous treatment and compression or compression alone, however the compression alone group in the EVRA trial received deferred endovenous treatment (after ulcer healing or from six months). There is high-certainty evidence that combined endovenous ablation and compression compared with compression therapy alone, or compression with deferred endovenous treatment, improves time to complete ulcer healing (pooled hazard ratio (HR) 1.41, 95% CI 1.36 to 1.47; I2 = 0%; 2 studies, 466 participants). Authors concluded that endovenous ablation of superficial venous incompetence in combination with compression improves leg ulcer healing when compared with compression alone. This conclusion is based on high-certainty evidence. There is moderate-certainty evidence to suggest that it is probably cost-effective at one year and low certainty evidence of unclear effects on recurrence and complications. [69]
Randomized controlled trials
- In 2020, Gohel et al. reported that one-year outcomes from the Early Venous Reflux Ablation (EVRA) randomized trial showed accelerated venous leg ulcer healing and greater ulcer-free time for participants who are treated with early endovenous ablation of lower extremity superficial reflux. Authors found that ulcers recurred at a lower rate of 0.11 per person-year in the early-intervention group compared with 0.16 per person-year in the deferred-intervention group (incidence rate ratio, 0.658; 95% CI, 0.480-0.898; P = .003). Time to ulcer healing was shorter in the early-intervention group for primary ulcers (hazard ratio, 1.36; 95% CI, 1.12-1.64; P = .002). At 3 years, early intervention was 91.6% likely to be cost-effective at a willingness to pay of £20 000 ($26 283) per quality-adjusted life year and 90.8% likely at a threshold of £35 000 ($45 995) per quality-adjusted life year. Authors concluded that early endovenous ablation of superficial venous reflux was highly likely to be cost-effective over a 3-year horizon compared with deferred intervention. Early intervention accelerated the healing of venous leg ulcers and reduced the overall incidence of ulcer recurrence. [68]
- A 2018 RCT (EVRA study, n=450 patients) showed that early endovenous ablation of superficial venous reflux as an adjunct to compression therapy is associated with a shorter time to healing of VLU than compression therapy alone, and increases ulcer-free time when compared to endovenous ablation performed after the VLU has healed. Patient and clinical characteristics at baseline were similar in the two treatment groups, with VLUs that were 6 weeks to 6 months old.[70][71]
SOE – PRP and Growth Factor
(Back to Text)
- A 2016 Cochrane systematic meta-analysis on use of autologous platelet-rich plasma (PRP) for chronic wounds evaluated 10 RCTs, 3 of which focused only on VLU. Authors concluded that it is unclear if autologous PRP has an effect on VLU and other types of chronic wounds other than diabetic foot ulcers. Studies showed no difference in the risk of adverse events in people treated with PRP or standard care. These findings are based on low quality evidence due to the small number of studies and patients included, and their poor methodological quality. Authors mentioned that the very few RCTs evaluating PRP are underpowered to detect treatment effects, if they exist, and are generally at high or unclear risk of bias [164]
- As for clinical guidelines, AAWC did not find evidence to support use of PDGF in VLU, and WHS does not support use of growth factors in VLU, although isolated reports suggest potential usefulness (28,99)
- Reviews of systematic analyses and meta-analyses included a small trial with 60 participants that compared patients who received GM-CSF 200 mcg or 400 mcg (once a week for 4 weeks). Healing rates of the intervention group was higher at a statistically significant level compared to placebo at 13 weeks [113] [208] [209]. However, the trial was small and underpowered to detect treatment effects.
- A few recent studies have evaluated the efficacy and safety of PRGF or epidermal growth factors (EGF) as local treatment for venous ulcers. One small trial with 58 patients compared application of PRGF and standard care and found that the average percentage healed area of the PRGF group was significantly higher than that of the control group (p=0.001). However, the trial was small and at unclear risk of bias [161] . Another study was observational and evaluated efficacy, safety of wound dressing containing epidermal growth factor (EGF) in a collagen-gel matrix on hard-to-heal venous leg ulcers, but there was no control group [162].
- In 2014, Barrientos et al published a review on growth factors for chronic wounds and concluded that the four growth factors that have shown the greatest potential in randomized controlled trials include GM-CSF, PDGF, bFGF and VEGF. Current studies to date are small, and have disparate endpoints and modes of growth factor and cytokine administration. Larger randomized controlled trials are needed to support efficacy, side effect profiles, and long term outcomes. All data should be interpreted with caution and any off-label use of these products for the management of wounds should be used in conjunction with the standard of care for non-healing wounds. Areas for future study include optimal delivery methods for growth factors and use of different combinations of growth factors and other adjuvant therapies in addition to debridement [160].
SOE – Split-thickness skin graft
(Back to Text)
A 2013 Cochrane systematic meta-analysis evaluated two trials that compared split-thickness autografts with a hydrocolloid dressing in 102 participants [210] [211]. In Warburg 1994, both groups had vein surgery and authors found no evidence of a significant benefit for autografts over a dressing in this small trial (102 participants) (risk ratio (RR) of healing with autografts 0.89, 95%confidence interval (CI) 0.34 to 2.31). Jankunas 2007 reported a large difference in healing rates (RR of healing 42.93, 95% CI 2.72 to 677.16). Authors of the meta-analysis concluded that these trials provide no firm evidence of benefit rather than firm evidence of lack of benefit. The different results could be due to the fact that at baseline, the Warburg 1994 trial had larger ulcers in the skin graft group, which may have biased the results against grafting, or it may indicate that allocation was subverted as there was no indication whether allocation was concealed [132]
Clinical guidelines: The 2014 Society for Vascular Surgery (SVS)[2] and the 2010 Association for Advancement of Wound Care (AAWC)[87] guidelines included different interventions related to autologous skin grafting.
|
Intervention for VLU
|
Society for Vascular Surgery
|
AAWC
|
Wound healing Society
|
|
Split-thickness skin grafting with continued compression for selected large venous leg ulcers that have failed to show signs of healing with standard care for 4 to 6 weeks.
|
Grade 2B
|
n/a
|
n/a
|
|
Pinch grafts
|
n/a
|
C2
|
n/a
|
We reviewed the Cochrane systematic review and the studies that the two guidelines used to grade quality of evidence and strength of recommendation (when applicable) for each of intervention above. Applying the GRADE framework to the combined body of evidence, we found that:
- Evidence that support use of autologous split-thickness skin grafting with continued compression for VLU that failed to decrease in size by 30% after 4 weeks compared with standard care is of low quality evidence (evidence level C). Authors of the 2013 Cochrane systematic review concluded that the trials studied provide no firm evidence of benefit rather than firm evidence of lack of benefit. Trials were small and/or at high and unclear risk of bias due to lack of outcome assessment blinding, and had inconsistency of results, probably due to discrepancies of wound size in different groups at baseline in of one of the trials. Studies on autologous skin graft cited by guidelines were not directly related to the use vs. no use of autologous split-thickness grafts to promote healing of VLU, as they compared split-thickness grafting with or without low molecular weight heparin or were retrospective studies on pinch grafts for VLU (28,99).
SOE – Cultured epithelial autografts
(Back to Text)
Recommendations related to this intervention given by the 2014 Society for Vascular Surgery (SVS)[2], the 2010 Association for Advancement of Wound Care (AAWC)[87] and the 2015 Wound Healing Society (WHS)[14][86] are listed below:
|
Intervention for VLU
|
Society for Vascular Surgery
|
AAWC
|
Wound healing Society
|
|
Cultured epithelial autografts or allografts have not been demonstrated to improve stable healing of venous ulcers
|
n/a
|
n/a
|
I - against use of these interventions
|
|
Split-thickness cultured autografts
|
n/a
|
C2
|
n/a
|
|
Cultured epidermal autografts (autologous keratinocytes)
|
n/a
|
B
|
n/a
|
We reviewed the citations that the two guidelines used to grade quality of evidence and strength of recommendation (when applicable) for each of intervention above. Applying the GRADE framework to the combined body of evidence, we found that:
- Use of cultured epidermal autografts to treat recalcitrant VLU is based on low quality evidence (evidence level C). One large RCT cited by the AAWC guideline concluded that cultured autologous keratinocytes in a fibrin vehicle were more effective in promoting VLU healing than standard care [155]. However, this study may be at high risk of bias due to attrition bias (no information on which group patients lost to follow up belonged to, which might affect outcomes), and lack of blinding in outcome assessment. Other observational studies also provide low quality evidence that support this intervention [156][157]. Case series evaluated use of split-thickness cultured autografts and provide low quality evidence to support their use in VLU [158] [159].
SOE – Skin allograft
(Back to Text)
- A 2013 Cochrane meta-analysis evaluated 5 small trials comparing fresh or frozen allografts for VLU. Authors found a higher healing rate with allografts than standard care, but these small trials were of poor quality and hence they recommended this result be treated with caution (evidence level C). Most of the trials provided insufficient evidence of benefit, rather than firm evidence of lack of benefit - Cochrane
- The 2012 clinical guideline for VLU published by AAWC categorized use of allografts for VLU as low quality evidence (level C)
Randomized controlled trials
- In 2019, Cazzell S published a small pilot RCT (n=18) that compared human acellular dermal matrix (DermACELL AWM) with conventional care as an adjunctive treatment of VLUs with more than 60 days of duration. At 24 weeks, 1 application of D-ADM demonstrated an increase in the healing rate over conventional care (44.4% vs. 33.3%, respectively), and decrease in wound size over conventional care at 24 weeks (with an average reduction of 59.6%, in contrast to the conventional care arm with an average reduction of 8.1%), though these differences were not statistically significant. Sample numbers of this manufacturer-sponsored trial were too low to determine statistical significance as expected (evidence level C, due to imprecision, lack of healthcare personnel and patient blinding, unclear randomization process). [137]
- In 2018, Towler et al. published an RCT that compared a human skin allograft (TheraSkin) and a bioengineered skin graft substitute (Apligraf) to promote healing of VLUs that failed to achieve 30% reduction in size after 4 weeks of standard therapy. Authors found that while both CTPs were equally effective in promoting VLU healing by week 20, treatment with TheraSkin was 42.2% cheaper than with Apligraf (difference was statistically significant, p=0.039) even though the initial wound sizes were not significantly different between groups (evidence level C, due to imprecision and assessor blinding bias).[139]
- No new RCTs related to use of human skin allografts published since 2012 have been found. There have been some other non-RCT studies since then, but these do not confer the degree of quality needed for the evidence to be considered relevant. Austin Pourmoussa
SOE - Allogeneic Matrix
(Back to Text)
Dehydrated human amnion/chorion membrane allograft
Systematic reviews and/or meta-analysis
- A 2024 systematic review and meta-analysis by Alomairi et al [146] included 10 RCTs (n=633 participants) that aimed to assess the efficacy of human amniotic membranes (HAM) in treating venous and diabetic ulcers. Findings revealed that HAM treatment significantly accelerated ulcer closure compared to groups treated with standard of care alone (p<0.0001), demonstrating over 90% complete healing compared to standard care. Despite moderate heterogeneity among studies, the results strongly suggested the effectiveness and safety of HAM therapy for venous and diabetic leg ulcers. The review was not industry sponsored, however, it did not stratify studies between VLU and DFU and instead analyzed results together.
Randomized controlled trials
- In 2022, Serena et al [145] conducted an RCT with 60 patients that evaluated safety and effectiveness of weekly and biweekly applications of dehydrated human amnion and chorion allograft (dHACA, AmnioBand Membrane; MTF Biologics) plus standard of care compared to standard of care alone on VLUs. At 12 weeks, significantly more venous leg ulcers healed in the two dHACA-treated groups (75 percent) than in the standard-of-care group (30 percent) ( p = 0.001) even after adjustment for wound area (p = 0.002), with an odds ratio of 8.7 (95 percent CI, 2.2 to 33.6). There were no significant differences in the proportion of wounds with percentage area reduction greater than or equal to 40 percent at 4 weeks among all groups. Authors concluded that dHACA and standard of care, either applied weekly or biweekly, significantly healed more venous leg ulcers than standard of care alone. An important finding in this study demonstrated that there were no differences in outcomes in terms of weekly (12 applications in 12 weeks) versus biweekly (6 applications in 12 weeks) application of dHACA. This trial was industry sponsored. The main limitation of this study was that blinding of patients and investigators was not possible because of the treatment regimens used in both groups (moderate certainty, evidence level B)
- In 2018, Bianchi et al [144] conducted an RCT with 109 subjects who received dehydrated human amnion/chorion membrane (EpiFix) and multilayer compression (n = 52) or dressings and multilayer compression therapy alone ( n = 57). Results showed that participants receiving weekly application of EpiFix and compression were significantly more likely to experience complete wound healing than those receiving standard wound care and compression at 12 and 16 weeks (60% versus 35% at 12 weeks, P = 0.0128, and 71% versus 44% at 16 weeks, P = 0.0065). This trial was industry sponsored, outcome assessors were not blinded (although a group of wound specialists blinded to the study and patients evaluated all images from both groups) (moderate certainty, evidence level B)
- In 2014, Serena et al [140] published results of a RCT with 84 participants that evaluated the safety and efficacy of one or two applications of dehydrated human amnion/chorion membrane allograft and multilayer compression therapy vs. multilayer compression therapy alone in the treatment of VLU. At 4 weeks, significantly more VLUs treated with the allograft showed a reduction in size greater than 40% compared with the control group (p=0.005). This trial was industry sponsored, outcome assessment was not blinded and the study end point was a surrogate measure of wound healing (low quality, evidence level C)
- Other studies are small, retrospective or did not report outcomes of use of membrane products on VLU separately.[141][142][143]
Human fibroblast-derived dermal substitute
- A 2013 Cochrane systematic review analyzed 2 trials (71 participants) that compared a single-layered dermal replacement (Krishnamoorthy 2003; Omar 2004) with standard care of four-layer compression and a low-adherent dressing. Both trials evaluated Dermagraft (Smith and Nephew), described as a human fibroblast-derived dermal replacement; the origin of the cells being newborn foreskin. There was no evidence of benefit associated with dermal skin replacement compared with standard care at 8 weeks. Authors concluded that there was therefore insufficient evidence, based on 2 these small studies, that single-layered dermal skin replacement heals venous ulcers more quickly than a simple low-adherent dressing [132].
- A study published in 2013 that was not included in the Cochrane review above (Harding et al)[147] published results of a large RCT with 366 patients that evaluated the efficacy and safety of human fibroblast-derived dermal substitute (Dermagraft) plus four-layer compression therapy compared with compression therapy alone in the treatment of VLU. Authors found that overall, the proportion of patients with VLUs that healed completely by treatment week 12 was not statistically significantly different between the interventional and control groups. However, when VLUs were stratified and only VLUs with less than 12 months in duration were considered, the proportion of VLUs completely healed by week 12 was significantly higher in the group treated with Dermagraft and compression than in the group treated with compression alone (P = 0·029). This study was industry sponsored, and there was no mention of blinded outcome assessment (moderate quality, evidence level B).
SOE – Composite Matrix
(Back to Text)
A 2013 Cochrane systematic meta-analysis concluded that bilayer tissue-engineered skin replacement, used with compression, increases the rate of healing of VLU compared with simple dressings used with compression. This conclusion was based on 2 trials (345 participants) that compared a bilayered skin equivalent with standard care of foam or a dressing made to look like the skin equivalent [132][149][150]. Meta-analysis shows that the artificial skin was significantly better in promoting VLU healing compared with simple dressings - 1.51 (95% CI 1.22 to 1.88). Authors of the meta-analysis mentioned that these 2 trials provide reasonable evidence that a greater proportion of venous ulcers heal with artificial skin than a simple dressing, but that the lack of an intention-to-treat analysis for the Falanga 1998 trial [150] reduces their certainty of the results [132]. Although the trial performed by Falanga et al was large, it was considered by the review’s authors at high risk of bias for attrition and performance biases (loss of patients to follow up and lack of blinding in outcome assessment). The trial performed by Brown-Etris [149] included only 36 participants and was underpowered to detect differences between groups. (level B)
Listed below are recommendations regarding this intervention given by the 2014 Society for Vascular Surgery (SVS)[2], the 2010 Association for Advancement of Wound Care (AAWC)[87] and the 2015 Wound Healing Society (WHS)(28,35):
|
Intervention for VLU
|
Society for Vascular Surgery
|
AAWC
|
Wound healing Society
|
|
Cultured allogeneic bilayer skin replacements - Apligraf (with both epidermal and dermal layers) to increase the chances for healing in patients with difficult to heal venous leg ulcers in addition to compression therapy in patients who have failed to show signs of healing after standard therapy for 4 to 6 weeks.
|
Grade 2A
|
A
|
I
|
No new RCTs on use of bilayered bioengineered skin for VLU since 2012 have been found. One retrospective study with data from 1489 patients found that treatment with bilayered skin increased the probability of VLU healing by 29% compared with porcine SIS dressing (hazard ratio = 1.29 [95% confidence interval 1.06, 1.56], p = 0.01) [212].
SOE – Acellular Matrix
(Back to Text)
- Studies on acellular matrices were not included in the 2013 Cochrane review [132] on skin grafts for VLU, as acellular matrices were considered as dressings. However, we grouped acellular matrices under CTPs, according to Medicare categorization of these products.
- In 2005, Mostow et al [151] published results of a RCT with 120 participants that compared treatment of VLU with acellular porcine matrix and compression vs. standard care. VLU in the interventional group healed more than in the control group at a statistically significant level. There was no significant difference in wound recurrence rates between the two groups. Potential biases of this industry-sponsored study include lack of blinding in outcome assessment, and sample size not large enough to reach statistical power (low quality evidence, level C). The Agency for Healthcare Research and Quality (AHRQ) and authors of a systematic review (Valle et al) considered evidence provided by this trial as low quality [152].
- Other non-RCT small studies evaluated use of acellular matrices in VLU, and reported positive results [213][214].
- Hyaluronic acid dressings can also be considered acellular matrices. Evidence on its effectiveness in VLU healing compared to standard care with non-adherent gauze or other types of dressing is of low quality:
- One RCT (n=170 patients) investigated the efficacy and safety of an hyaluronic acid (HA)-impregnated gauze pad compared with a hydrocolloid (HC) dressing, in patients with leg ulcer of venous or mixed etiology [215]. The primary endpoint was the difference between the groups regarding the percentages of patients achieving a reduction of at least 40% of the initial wound surface after 56 days of treatment. There was no statistically significant difference in healing rates between the interventional and control groups. Another trial (n=89 patients) investigated the efficacy and safety of a gauze pad containing HA in local treatment of venous leg ulcers, compared with its neutral vehicle. The primary endpoint was the percentage of wound size reduction after 45 days. The number of healed ulcers was significantly higher in the HA group at day 45 and 60 (p<0.05) [216]. One small cohort study reported positive results for another type of hyaluronic acid dressing [217] and another study compared hyaluronic acid based dressing and compression therapy versus non-adherent gauze and compression therapy, but this study did not report baseline comparability, inclusion/exclusion criteria, randomization, blinding, allocation concealment or sample size calculation [154][218].
Listed below are recommendations regarding acellular matrix given by the 2014 Society for Vascular Surgery (SVS)[2], the 2010 Association for Advancement of Wound Care (AAWC)[87] and the 2015 Wound Healing Society (WHS)[14][86]:
|
Intervention for VLU
|
Society for Vascular Surgery
|
AAWC
|
Wound healing Society
|
|
Porcine small intestinal sub-mucosa tissue construct in addition to compression therapy for the treatment of venous leg ulcers that have failed to show signs of healing after standard therapy for 4 to 6 weeks
|
Grade 2B
|
n/a
|
II
|
- We reviewed the citations that the two guidelines used to grade quality of evidence and strength of recommendation (when applicable) for each of intervention above. Applying the GRADE framework to the combined body of evidence, we found that:
-
- Use of porcine small intestinal submucosa tissue construct to treat recalcitrant VLU is based on low quality evidence (evidence level C). The guidelines also used Mostow’s study [151] as a reference to their suggestion. As mentioned above, potential biases of this industry-sponsored study include lack of blinding in outcome assessment, and sample size not large enough to reach statistical power (low quality evidence, level C). The Agency for Healthcare Research and Quality (AHRQ) and authors of a systematic review [126] considered evidence provided by this trial as low quality [152].
SOE – Drugs/biologics
(Back to Text)
The 2013 Cochrane systematic review described one trial (Goedkoop 2010) that compared growth-arrested human keratinocytes and fibroblasts (HP802-247) with placebo equivalent in 110 participants. For the purposes of the analysis authors of the review combined the results of the six intervention groups and compared them with the placebo group; there was no statistically significant difference between the two groups at either 12 weeks or 24 weeks (Cochrane, 2013)
Listed below are recommendations regarding this intervention given by the 2014 Society for Vascular Surgery (SVS)[2], the 2010 Association for Advancement of Wound Care (AAWC)[87] and the 2015 Wound Healing Society (WHS)[14][86]:
|
Intervention for VLU
|
Society for Vascular Surgery
|
AAWC
|
Wound healing Society
|
|
Cultured epithelial allografts have not been demonstrated to improve stable healing of venous ulcers
|
n/a
|
n/a
|
I – suggest against
|
|
Cultured allogeneic keratinocyte lysate
|
n/a
|
C1
|
n/a
|
SOE – Phlebotonics
(Back to Text)
- A 2017 systematic review [170] concluded that there is moderate quality evidence supporting the use of MPFF in VLU healing, however it excluded the largest trial that assessed use of MPFF on VLU healing, which was unpublished and showed no difference in VLU healing between control and interventional groups[219]. This trial was included in the 2013 Cochrane systematic meta-analysis [173] described below.
- A 2016 Cochrane systematic meta-analysis [172] evaluated 6 RCTs (461 participants) that compared efficacy of phlebotonics like aminaftone, diosmine, or rutoside versus placebo on ulcer healing. Pooled results showed no statistically significant differences between phlebotonics and placebo (RR 0.94, 95% CI 0.79 to 1.13; I2 = 5%; 461 participants). Quality of evidence was downgraded due to selective outcome and incomplete outcome data reporting (evidence level C). Authors also evaluated other outcomes related to chronic venous insufficiency and found that moderate-quality evidence shows that phlebotonics may have beneficial effects on edema and on some signs and symptoms related to CVI such as trophic disorders, cramps, restless legs, swelling and paraesthesia when compared with placebo but can produce more adverse effects.
- A 2013 Cochrane systematic meta-analysis [173] evaluated 9 RCTs (1075 participants) that compared efficacy of flavonoids and compression on VLU healing. Five of these trials investigated Micronised Purified Flavonoid Fraction (MPFF), and 4 evaluated hydroxyethylrutosides (HR). Meta-analyses showed that more venous leg ulcers were healed in the MPFF groups than in the control groups (RR 1.36; 95% CI 1.07 to 1.74), and in the HR groups than in the control groups (RR 1.70; 95% CI 1.24 to 2.34). However, most of these trials were poorly reported, and so had an unclear risk of bias for randomization, allocation concealment, blinding and methods for addressing incomplete outcome data. There was also a possibility of publication bias (level of evidence C). The only trial with a low risk of bias, with the largest sample size showed no difference in VLU healing outcomes between patients in the MPFF experimental group and control group[219], and it was unpublished, raising the possibility of publication bias, as its result was not favorable for flavonoids. There is evidence to show that studies that are inadequately concealed and not blinded can lead to overestimation of treatment effects by up to 41% and 17% respectively [173].
SOE – Aspirin
(Back to Text)
- A 2016 Cochrane systematic review included two RCTs of oral aspirin (300 mg/daily) given to patients with VLU in addition to compression and compared with compression and placebo, or compression alone. Results of the small trials indicated that aspirin was effective in increasing healing rate, preventing recurrence and healing more VLUs compared with the control groups. However, authors of the review downgraded the evidence due to potential selection bias and imprecision due to the small sample size and concluded that low quality evidence from these two trials indicate that there is currently insufficient evidence for them to draw definitive conclusions about the benefits and harms of oral aspirin on the healing and recurrence of venous leg ulcers[174].
Randomized Controlled Trials
- A 2018 phase 2 study comparing 300 mg of aspirin with placebo for the treatment of VLUs (AVURT) trial conducted in the UK found no effect on VLU healing with aspirin as an adjunct to compression. The quality of evidence is low due to small sample size.[176]
SOE - Zinc
(Back to Text)
- A 2014 Cochrane systematic review included 6 small RCTs comparing oral zinc sulphate with placebo or no treatment in people with arterial or VLU. Pooling of these trials indicated no statistically significant difference between the two groups for healing (RR 1.22, 95%CI 0.88 to 1.68). Overall, there is no evidence that oral zinc increases the healing of arterial or venous leg ulcers, however evidence is of low quality due to small size of trials and unclear risk of bias (poor reporting of outcomes) [62].
SOE - Sulodexide
(Back to Text)
- A 2016 Cochrane systematic review included 4 RCTs (463 participants) comparing sulodexide as an adjuvant to local treatment (including wound care and compression therapy) compared with local treatment alone. Meta-analysis of three RCTs suggests an increase in the proportion of ulcers completely healed with sulodexide as an adjuvant to local treatment compared with local treatment alone (rate of complete healing with sulodexide 49.4% compared with 29.8% with local treatment alone; RR 1.66; 95% CI 1.30 to 2.12). This evidence for sulodexide increasing the rate of complete healing is of low quality due to risk of bias (evidence level C). It is unclear whether sulodexide is associated with any increase in adverse events (4.4% with sulodexide versus 3.1% with no sulodexide; RR 1.44; 95% CI 0.48 to 4.34) (evidence level C) [179]
SOE – Electrical Stimulation
(Back to Text)
- Several studies show that electrical stimulation may be useful in treating VLU, however most studies have a small sample size and may be underpowered to detect statistical significance [182] [220]included other types of ulcers and did not report results for each type separately ) [221] [222] [223], or are observational in nature [224].Treatment parameters and type of electrical stimulation among these studies are controversial.
SOE – Electrical magnetic therapy
(Back to Text)
- A 2015 Cochrane systematic review [183] included 3 small RCTs (94 participants) that evaluated the use of electromagnetic therapy (EMT) and sham EMT in patients with VLU. Two studies that reported healing rates had conflicting results regarding effectiveness of EMT, and the other study reported significantly greater reductions in ulcer size in the EMT group however this result may have been influenced by differences in the baseline characteristics of the wounds/patients. Authors of the review concluded that at present, there is no high-quality evidence (it is not clear) that EMT speeds the healing of venous leg ulcers. Further research would be needed to answer these questions (evidence level C).
- Trials cited by the AAWC venous ulcer guideline [11] were the same as the ones evaluated by the Cochrane review described above.
SOE – Ultrasound therapy
(Back to Text)
- A 2010 Cochrane systematic review included 8 RCTs that evaluated high frequency ultrasound (HFU) and low frequency ultrasound (LFU) for treatment of VLU. All trials were small, of poor-quality and heterogeneous. Authors of the review concluded that there is no reliable evidence that US hastens healing of venous ulcers. There is a small amount of weak evidence of increased healing with US, but this requires confirmation in larger, high-quality RCTs. There is no evidence of a benefit associated with low frequency US [184].
- A meta-analysis conducted by Voigt et al in 2011 [225] included 8 RCTs. Results demonstrated that early healing (at =5 months) in patients with venous stasis and diabetic foot ulcers was favorably influenced by both high- and low-intensity ultrasound delivered at a low frequency—either via contact or noncontact techniques. However, the quality of the data may be suspect, especially for low-frequency low-intensity noncontact ultrasound because of significant biases. In patients presenting with either venous stasis or diabetic foot ulcers (Wagner classification 1-3), early healing appears to be facilitated by either low-frequency low-intensity noncontact ultrasound or low-frequency high-intensity contact ultrasound.
The 2014 Society for Vascular Surgery [2], the 2015 Wound Healing Society [14][86], and the 2010 Association for Advancement of Wound Care [87] guidelines do not support use of ultrasound therapy as primary therapy in the treatment of VLU, although the levels of evidence assigned by these societies differed from each other, possibly due to different evidence grading methodologies and different studies from which evidence was drawn (SVS uses GRADE framework).
|
Intervention for VLU
|
Society for Vascular Surgery
|
AAWC
|
Wound healing Society
|
|
Ultrasound therapy for wounds that fail to progress after 4 weeks of standard care
|
2B, suggest against ultrasound as primary therapy
|
A
|
III
|
- A few recent RCTs evaluated the use of ultrasound in the treatment of VLU. Beheshti et al [185] and Olyaie et al [186] published results of a RCT (90 participants) that compared standard care with HFU and MIST ultrasound therapy on VLU that failed to respond to SC for 4 weeks. Size of ulcer, mean degree of pain and edema in ultrasound therapy was decreased after the 4-month visit in comparison to the standard-treatment group (p = 0.01, p < 0.0001 and p < 0.0001, respectively). However, authors report no blinding in outcome assessment and there is no description of sample size calculation (may be underpowered to detect statistical differences)
- Watson et al [226] conducted a large RCT with 337 patients with at least one venous leg ulcer of >6 months' duration or >5 cm [3] area and an ankle brachial pressure index of = 0.8, that aimed at assessing the clinical effectiveness of weekly delivery of low dose, high frequency therapeutic ultrasound in conjunction with standard care for hard to heal VLU. Authors concluded that low dose, high frequency ultrasound administered weekly for 12 weeks during dressing changes in addition to standard care did not increase ulcer healing rates, affect quality of life, or reduce ulcer recurrence. Authors concluded that therapeutic ultrasound does not confer any benefit on the healing of hard to heal venous leg ulcers (that is, >5 cm2 in area or >6 months old, or both). The trial can be considered of good quality (evidence level A)
SOE – Hyperbaric oxygen therapy
(Back to Text)
- A 2015 Cochrane review was able to find only one small RCT that evaluated use of HBOT on VLU healing [187][188]. The trial (16 participants with VLU for more than a year) compared treatment with 2.4 ATA for 90 minutes to a total of 30 sessions over six weeks with an air-breathing sham treatment on the same schedule. There was a significant reduction in wound area at six weeks following the administration of HBOT (33%mean difference in area ulcerated, 95%CI 19 to 47), but this effect did not persist to 18 weeks and there was no significant increase in the proportion of ulcers healed at any time (RR 5.00, 95%CI 0.28 to 90.18, P = 0.28). The trial did not report undertaking a sample size calculation and may have been underpowered to detect any statistically significant effect of treatment. Also, it was considered at unclear risk for selection and reporting bias [187].
Randomized controlled trial
- A 2018 RCT (n=31) evaluated effect of adjunct HBOT on healing of patients with VLU that failed to show an area decrease of > 50% in 4 weeks, whose transcutaneous oxygen measurement were indicative of a hypoxic wound responsive to oxygen challenge, and who did not have contraindications for HBOT. Authors found that the HBOT intervention group had a mean of 95% reduction in ulcer size compared to 54% for the placebo group (t=-2.24, p=0.042, mean difference -40.8, SE 18.2) at 12 weeks. They concluded that HBOT may improve refractory healing in venous leg ulcers, however patient selection is important. In this study, HBOT as an adjunct treatment for nonhealing patients returned indolent ulcers to a healing trajectory.[189]
SOE – Negative Pressure Wound Therapy
(Back to Text)
- A 2015 Cochrane review on the use of negative pressure wound therapy (NPWT) for hard to heal leg ulcers (all types) [190] was able to identify only 1 RCT. The trial was small (n=60) (Vuerstaek 2006) [227], and compared NPWT followed by a punch skin-graft transplant and further NPWT treatment with standard wound care followed by a punch skin-graft transplant and subsequent standard care. The study found that the median time to healing in the NPWT group was 28 days (95% CI 25.5 to 32.5) compared with 45 days (95% CI 36.2 to 53.8) in the standard care group. The study reported an adjusted hazard ratio of 3.2, 95% CI 1.7 to 6.2 suggesting a higher chance of healing with NPWT. However, this evidence was considered of low quality due to imprecision and indirectness. Authors of the review concluded that there is no RCT evidence on the effectiveness of NPWT as a primary treatment for leg ulcers, and that there is limited evidence regarding the use of negative pressure wound therapy (NPWT) for the treatment of hard-to-heal leg ulcers. Given the current uncertainties, practitioners may elect to consider various characteristics such as costs and symptom management properties when choosing between alternative treatment options for leg ulcers. Cochrane. A recent RCT [228] that compared 2 types of NPWT has not been included in the review as review authors are awaiting clarifications from the study authors. The paper suggests that people with VLUs were randomized along with people with foot ulcers. However, the paper only reported data for those with VLU - which is a sub-group of the overall population. This might impact on the randomized nature of the study Cochrane.
|
Intervention for VLU
|
Society for Vascular Surgery
|
AAWC
|
Wound healing Society
|
|
NPWT prior to skin graft/flap
|
n/a
|
C1
|
II
|
|
NPWT as primary therapy for VLU
|
2C, suggest against
|
n/a
|
n/a
|
REVISION UPDATES
| Date | Description |
| 5/30/24 | Expanded sections Surgical Vascular Interventions and Prevention |
| 6/19/19 | Added section on Compression Therapy for Cases that are Hard-to-Assess/ Manage/ Heal, updated references |
| 10/24/19 | Updated sections on Compression Therapy and Adjunctive Therapies |