NPWT Documentation Checklist |
CLINICAL
Overview
This topic provides an evidence-based review on the use of Negative Pressure Wound Therapy (NPWT) with a focus on chronic wounds. For guidance on selection of NPWT device, device application, and assessment of NPWT effectiveness, see "How to Select and Apply Negative Pressure Wound Therapy Devices". For decision support on different brands of disposable and durable NPWT devices and dressings, see NPWT Interactive Matrices for "NPWT Disposable Pumps", "NPWT Durable Pumps" and "NPWT Dressings". To assess if NPWT is indicated for a specific wound type, use the Wound Prep & Dress Tool. For a case series of circumferential NPWT, see "Case: Circumferential Negative Pressure Wound Therapy"
Background
NPWT is an advanced wound care modality that utilizes subatmospheric (negative) pressure to assist with wound healing via delayed primary intention. Various systems are in use today and are generally composed of the main unit which provides negative pressure, a collection canister for exudate, connection tubing to carry exudate to the collection canister, a foam or gauze dressing medium to conduct negative pressure to the wound bed, and a semipermeable adhesive tape or film to form a seal. While NPWT systems have different features and indications (e.g, varied dressing interfaces, vacuum settings, single versus multiple use), the principle of applying a negative pressure to the wound in a closed environment is the same for all products.[1]
The intervention was developed in the 1990s, and the uptake of NPWT in the healthcare systems of developed countries has been dramatic.[2] A US Department of Health report estimated that between 2001 and 2007, Medicare payments for NPWT pumps and associated equipment increased from USD 24 million to USD 164 million (an increase of almost 600%).[1][3] Initially only one NPWT manufacturer supplied NPWT machines (the VAC system: KCI, An Acelity Company, San Antonio, Texas), however, as the NPWT market has grown, a number of different commercial NPWT systems have been developed, with machines becoming smaller and more portable. Single use, disposable devices are recent additions to the different types of devices currently used. “Homegrown” devices, assembled locally ad-hoc by clinicians are also seen, and can be particularly prevalent in areas with limited resources. These systems usually consist of a wound dressing interface, transparent occlusive films, and negative pressure generated by vacuum suction pumps or traditional wall suction.[1]
A wide spectrum of clinicians with various backgrounds and training prescribe and apply NPWT, and it is now used both in secondary and primary (community) care, particularly following the introduction of ambulatory systems, and prophylactically, to prevent surgical site infection.
How the intervention works
The NPWT dressing interface is applied directly to the wound bed, which expedites wound healing through several processes [2][4] (Figure 1):
- promotes tissue perfusion and proliferation via tissue strain (macro and microstrain)
- encourages wound contraction, physically drawing wound edges together
- provides exudate management and evacuates infections material
- decreases interstitial tissue edema, serving to decrease pain
- provides a moist wound healing environment
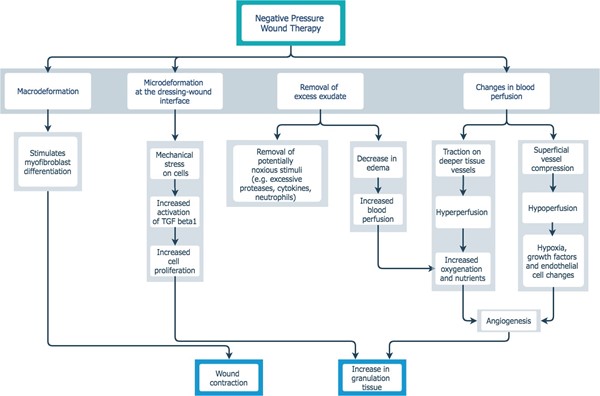
Figure 1. NPWT - proposed mechanism of action[2][4]
General Indications and Contraindications
Indications
NPWT
- Most manufacturers endorse application of NPWT for patients with chronic, acute, traumatic, sub-acute and dehisced wounds, burns (partial thickness or full thickness post debridement), orthopedic injuries/open fractures, ulcers (such as diabetic, pressure and venous insufficiency), flaps, and grafts.
- Practically, management of highly exudative wounds is a very popular indication for many clinicians in the acute and post-acute settings. Also, closed management of wounds with NPWT can be extremely beneficial in addressing patient-specific risk factors of poor hygiene, poor dressing change adherence related to caregiver availability, physical and psychological barriers, or knowledge deficits.
INCISIONAL NPT
- Used for management and protection of closed surgical incisions, typically for 7-14 days, in specific patient populations.[5] See more details in the section 'Evidence and Recommendations' below.
- The WHO Global Guidelines for the Prevention of Surgical Site Infections (SSI) 2016 [6] recommends incisional NPT to reduce surgical site infections for high risk adult patients (obese, smokers, history of delayed healing, immunosuppressants, etc), taking facility and patient resources into account.
- The American College of Surgeons and Surgical Infection Society (SIS) Surgical Site Infection Guidelines [7] recommend Incisional NPT application to decrease SSI rate in open colorectal and vascular procedures over incisions in which the skin is closed with staples. [8]
- Incisional NPT for complex surgical procedures is gaining popularity, in part due to preliminary data demonstrating reduction in SSI.[9]
OPEN ABDOMEN NPT
- Indicated for temporary closure of abdominal wall when open abdomen management is implemented following peritonitis or abdominal trauma, to treat or prevent intra-abdominal hypertension, and to manage abdominal compartment syndrome. Open abdomen NPT appears to be associated with better outcomes in terms of mortality compared to modified Barker Vacuum Pack (mBVP) technique, which is another form of temporary closure [10], or to no NPWT. [11]
NPWT WITH APPLICATION OF CLEANSING SOLUTION
- NPWT with application of cleansing solution (also known as NPWTi) is appropriate for select patients, including those with substantial comorbidities that are known to impair wound healing and/or response to infection (e.g., complex hosts), or those who have a clinically complex wound.[12] NPWT with application of cleansing solution should not be used routinely to treat uncomplicated wounds or hosts without clinically significant comorbidities.[12]
- Complex hosts can be defined as patients who are [12]:
- An American Society of Anesthesiologists physical status classification of ≥ 2, OR
- Have two or more clinically relevant comorbidities, such as coronary artery disease, peripheral vascular disease, active cancer, renal failure or diabetes mellitus.
- Complex wounds include:
- Wounds that contain non-viable tissue, such as severe traumatic wounds, diabetic foot infections, and wounds complicated by invasive infection or extensive biofilm.[12] An increasingly common indication for NPWT with application of cleansing solution is necrotizing soft tissue infections.[13]
- NPWT with application of cleansing solution is a method that is particularly useful when one of the primary goals of wound management is to remove infectious material and other wound debris, as well as to promote granulation (classical NPWT effects).[14] NPWT with topical wound solution instillation allows the combination of sterile wound coverage with active moist wound therapy, resulting in softening of devitalized tissue and infectious material, followed by aspiration of debris through negative pressure.[14]
- Wounds that still have > 60% of wound area covered with devitalized tissue despite standard treatment with adequate debridement on patients for whom surgical debridement is contraindicated.[14]
- Wounds for which multiple sessions of surgical debridement are anticipated. NPWT with application of cleansing solution may decrease need for repeated surgical debridements.[14]
- See more details on NPWT with application of cleansing solution in the section 'Evidence and Recommendations' below.
Disposable NPWT
- Typical indications include [15]:
- Patient-related indications: patients who are mobile, patients who work, as a step down from a larger standard NPWT device, people who do not have access to electricity.
- Wound-related indications: smaller wounds (1-18 cm2), minimal to moderate exudate (<=80ml/week), depth < 3cm.
Contraindications
NPWT
- Osteomyelitis without current antibiotic therapy, wound malignancy, exposed organs, bowel, or blood vessels, use in thoracic or abdominal cavities, necrotic tissue with eschar present, unstable structures (e.g. flaps or grafts), patients at increased risk of bleeding, and non-enteric or unexplored fistulas (to clarify, NPWT can be applied around or in the vicinity of a fistula, but not applied to directly evacuate fistula contents).
INCISIONAL NPT
- Not for use on open wounds, and with some systems being silver impregnated, it is contraindicated or for patients with allergy or sensitivity to silver.
NPWT WITH APPLICATION OF CLEANSING SOLUTION
- Same contraindications as traditional NPWT listed above.[12]
- Some manufacturers (e.g., V.A.C Veraflo) mention that NPWT with application of cleansing solution may be used with caution in patients at an increased risk of bleeding complications, with signs of systemic infection or advancing infection at the wound site, with exposed structures, with explored tunnels or with explored undermining.
DISPOSABLE NPWT
- Typical contraindications include [15]:
- Patient-related contraindications: patients with adherence issues, with dementia, color-blind (cannot see visual alarms well).
- Wound-related contraindications: larger wounds (>18 cm2), large quantity of exudate (>80ml/week), depth > 3cm, wounds with >20% coverage with eschar and/or devitalized tissue after debridement
NPWT Types and Features
In general, NPWT systems are comprised of a vacuum source, dressing, and drainage tubing. Nevertheless, the several NPWT devices commercially available may differ in several manners:
Power supply
NPWT systems can be divided into electrically-powered and mechanically non-powered devices:
- Electrically-powered NPWT: operates on various power sources, including disposable or rechargeable batteries, or outlet power in order to provide negative pressure. The electronic mechanism can translate to a larger overall footprint and a louder machine.
- Mechanically-powered NPWT: uses mechanical energy from a spring mechanism to create a vacuum for evacuation of exudate, allowing for a more compact and stealthy device.
Durability
- Non-disposable: durable medical equipment maintained after each use, and re-used for multiple patients (e.g., ActiV.A.C.™ or Renasys™)
- Disposable: non-durable device, discarded after use on one patient, and not re-used (e.g, SNAP™ Therapy System, Pico™ Single Use NPWT)
Dressing Interfaces
Dressing interfaces vary by manufacturer. Most systems offer more than one interface material, as well as antimicrobial options. The most common dressing mediums include:
- Foams (polyurethane, polyvinyl alcohol, etc),
- Hydrocolloids
- Gauze
Suction (Subatmospheric or Negative Pressure Setting)
- Continuous: NPWT device produces vacuum at a set pressure continuously without interruption. Continuous setting is helpful in cases in which a dressing seal is difficult to achieve, as it limits the risk of leakage; this is crucial when using NPWT as a bolster over a skin graft, as graft would become detached from wound bed at each return to atmospheric pressure if a varied pressure setting was utilized. [16]
- Varied: NPWT device produces cycled negative pressure in one of two ways, known as cyclic and intermittent. It may be associated with greater stimulation of granulation tissue formation. [16] This setting can also be associated with increased pain. Most systems recommend initiating NPWT at continuous suction to establish a seal and confirm therapy viability before changing to any varied settings. Examples of varied settings are:
-
- Intermittent: e.g. -150mmHg for 5 minutes, followed by 2 minutes at -50mmHg,
- Cyclic: e.g. -100mmHg to -25mmHg in decreasing increments of 10 mmHg every 30 seconds, then increasing back to -100mmHg in the same manner.
Clinical application
- Incisional Negative Pressure Therapy (NPT): For management of primarily closed surgical incisions, there are special systems available. Incisional negative pressure systems help approximate edges of the incision, removes fluids and infectious materials, and acts as a barrier to external contamination. [5] This therapy is utilized for its ability to decrease patient pain postoperatively, increase perfusion to the incision site, evacuate local subcutaneous edema, encourage lymphatic flow, and decrease contamination. Most systems provide continuous negative pressure at a maximum -125mmg for up to 7 days (not all systems allow selection of pressure setting, and some systems are able to remain intact up to 14 days). Incisional negative pressure is recommended by both the World Health Organization (WHO)[6] and the American College of Surgeons (ACS)[7] for select patient populations, in order to prevent surgical site infections (SSI).
- Open Abdomen Negative Pressure Therapy (NPT) (e.g, AbThera, Acelity) [17] : For some surgical procedures, it is necessary to leave the abdominal cavity open for re-exploration; complex hernia repair, gross contamination and inflammation necessitating staged or delayed bowel resection or re-anastamosis, among others. The open abdomen management system provides temporary closure of the abdominal cavity. Therapy serves to:
- Evacuate ascities/infectious materials and reduce edema
- Provide tension on the layers of the abdominal wall to reduce loss of fascial domain
- Act as a barrier between the viscera and the external environment
- NPWT with application of cleansing solution (e.g. Acelity, Cardinal): For complex wounds and/or patients with multiple co-morbidities.[12] In recent years, the addition of closed administration of a topical solution into the wound bed without necessitating removal and reapplication of NPWT has broadened the available therapeutic modalities for complex wound care. Some systems offer specialized foam dressings that are more hydrophobic in order to disperse solutions more effectively. Other systems rely on gravity or constant negative pressure to move solution through the dressing system.
- The dynamics of cleansing solution delivery fall into two primary categories: simultaneous and intermittent.[18]
-
- Simultaneous delivery (e.g., Cardinal SVED) administers irrigation solution and NPWT continuously with varying flow rates that are adjusted independent of NPWT pressure.
- Intermittent delivery (e.g., V.A.C Veraflo Instillation Therapy/Dwell) involves pausing NPWT while a prescribed volume of solution fills the dressing and is delivered to the wound bed, followed by resumption of negative pressure and subsequent evacuation of the solution, along with exudate and debris.
- Types of cleansing solutions: various topical solutions are utilized, including wound cleansers/antiseptics, topical antibiotics, local anesthetics, etc. Currently, the standard of care is normal saline; however other solutions are utilized in clinical practice. The basis for selection of agent mostly includes clinician preference and literature evidence related to effectiveness as a topical cleansing agent in the absence of simultaneous use with NPWT (i.e. evidence describes appropriateness when used as a topical cleanser with traditional dressing regiment versus use with NPWT cleansing system). In the original studies for KCI VeraFlo, Prontosan (polyhexanide 0.01%) and 0.9% normal saline were validated for use with their system. Very little validated evidence exists for any solution other than normal saline. In fact, many studies demonstrate no clinical value of using antiseptics over normal saline in acutely infected wounds. [12][19] The only caveat is in the case of infected implanted hardware, where surfactant containing solutions are recommended for disruption of biofilms.
Evidence and Recommendations
Diabetic Foot Ulcers
-
2CFor patients with non-infected, nonischemic DFUs that have failed to respond to 4 weeks of adequate standard care or for patients with wounds from a diabetic foot amputation (postoperative wound) clinicians might opt to use adjunctive NPWT instead of standard dressings to promote wound healing (Grade 2C).
- Rationale: Clinical guidelines suggest use of NPWT as an adjunctive therapy to promote healing of post-surgical wounds in patients with DFU (post amputation and/or skin grafting), or after debridement of non-surgical DFUs.[20][21][22][23] Evidence supporting use of NPWT in these contexts is considered of low certainty (evidence level C), as outlined by recent systematic reviews and meta-analyses.[24][25][26] For more details, see topic "Diabetic Foot Ulcer - Treatment".
Pressure Ulcers/Injuries
- 2CFor patients with healable Stage 3 or 4 pressure ulcers/injuries (PUs/PIs) that have failed to respond to 4 weeks of adequate standard care, clinicians might opt to use adjunctive NPWT to promote wound healing (Grade 2C)
- Rationale: The application of continuous or intermittent negative pressure to wounds [27] may be used as an adjunctive therapy for deep, Stage 3 or 4 PU/PI [28][29], even before consideration for definitive flap coverage as long as ulcer is clean and there are no contraindications to NPWT, and as long as a NPWT system is necessary to reduce the number of dressing changes (e.g., wound or ulcer with large amount of exudate).[30] NPWT helps promote PU/PI healing through increased blood perfusion and granulation tissue, and decreased bacterial load.[31] Evidence of NPWT used in this context is considered of low certainty (evidence level C).[32][1] There is currently no rigorous evidence available favoring use of NPWT over other alternatives in the treatment of pressure ulcers.[1] For more details, see topic "Pressure Ulcers/Injuries - Treatment"
- For patients with healable Stage 3 or 4 pressure ulcers/injuries that have failed to decrease in area by 40% after 4 weeks of adequate standard care, expert consensus suggests use of NPWT with application of cleansing solution, if resources are available (Expert opinion).[33]
- Rationale: The expert group believes that patients with a healable Stage 3 or 4 PU/PI should receive proactive, ‘aggressive’ care. The group supports adoption of NPWT with application of cleansing solution at earlier stages of the wound healing continuum, provided resources and skilled personnel are available.[33]
Surgical wounds
- For treatment of surgical wounds healing by secondary intention (e.g., open, infected groin wounds, excised pilonidal sinus), clinicians might opt to use NPWT to promote wound healing if NPWT is needed to reduce the number of dressing changes or is expected to help manage symptoms better than other alternatives.
- Rationale: There is currently no rigorous evidence available regarding the clinical effectiveness of NPWT in the treatment of surgical wounds healing by secondary intention. More randomized controlled clinical trials are needed to evaluate NPWT for this condition.[34][35]
- As for surgical wound healing by primary intention (i.e., NPWT over primarily closed surgical incisions), clinical guidelines recommend utilization of incisional NPT to decrease surgical site infections in specific high-risk patient populations (i.e., obese, smokers, history of delayed healing, immunosuppressants, patients undergoing open colorectal and vascular procedures resulting in incisions where the skin is closed with staples). [6][7]
- Rationale: Incisional NPT is used to remove exudate from the wound, with the intention to promote faster healing and prevent SSI. Clinical guidelines recommend use of incisional NPT for high-risk patient populations. Utilization of Incisional NPT for complex surgical procedures is increasing, in part due to preliminary data demonstrating reduction in SSI. For instance, a study comparing use of incisional NPT and traditional closure following open pancreatoduodenectomies (Whipple procedure) showed that incidence of postoperative superficial SSI was significantly lower in the incisional NPT group when compared to the traditional closure group (9.7% and 31.1% respectively).[9] Many randomized clinical trials have been published in the past decade, however due to the lack of information on the study designs, incomplete reporting or small sample size, evidence supporting use of incisional NPT to prevent SSI or other undesired outcomes compared to other dressings is still of low certainty. A systematic review (n=2957 patients) found that evidence supporting positive clinical effects of NPWT for reducing SSI, wound dehiscence, and decreasing time to healing continues to be of low certainty (evidence level C), due to the lack of high-quality studies evaluating this intervention to date.[36] An industry-prepared meta-analysis concluded that incisional NPT performed significantly better at reducing the incidence of SSIs than traditional dressings, however the analysis was based on low certainty studies.[37] The high incidence of blisters occurring when NPWT is used following orthopedic surgery suggests that the therapy should be limited until safety in this population is established.[8]
Skin grafts
- 2CFor patients treated with split-thickness skin grafts, clinicians might opt to apply NPWT over the skin graft to decrease graft loss (Grade 2C).
- Rationale: There is some evidence that NPWT increases graft take compared to conventional therapy.[8][38] Conventional therapy causes pain during dressing changes. NPWT is an alternative method to cover the wound bed. There appears to be clear cost benefits when non-commercial systems are used to create the negative pressure required for wound therapy, with no evidence of worsening of clinical outcome.[8]
Cellular and/or tissue based products (CTP) or skin substitutes
- 2CFor patients with wounds treated with CTPs, clinicians might opt to apply NPWT over the CTP to promote wound healing (Grade 2C)
- Rationale: Application of NPWT over dermal substitutes (e.g. Integra®, Pelnac®, Matriderm®, and Alloderm®, etc) may expedite the wound healing process by reducing the time required for vascular ingrowth into the dermal substitute, improving dermal graft take, and allowing for earlier autograph application over the dermal substitute.[4][39][40]The addition of NPWT to a CTP, particularly in difficult areas of slow granulation (exposed bone, scalp injuries etc), appears to speed up the process of granulation tissue formation in these ‘hard-to-heal’ areas, and could be an alternative to free microvascular tissue transfer coverage.[4][41] NPWT enables dermal substitutes to come into closer contact with the wound bed, and may reduce risk of infection by effectively removing wound secretions and inflammatory factors.[40] An experimental study demonstrated that NPWT in combination with CTP resulted in higher collagen formation and neovascularization when compared to control groups.[42]
- As for NPWT pressure levels and operations mode, a negative pressure level up to 125 mmHg, and intermittent vacuum suction have been suggested.[40] Intermittent vaccuum suction may accelerate the migration of endothelial cells in dermal substitutes and stimulates a stronger vasodilator response.[40][43]
Complex patients and/or complex wounds
- For select patients (with several comorbidities and/or complex wounds), clinicians might opt for NPWT with application of cleansing solution to promote wound healing.[12][14][44]
- Rationale: Use of NPWT with application of cleansing solution (e.g. Cardinal SVED, Vera Flo) may help remove debris and decrease bacterial load on infected or non-healing wounds.[14] Despite increasing popularity, evidence supporting its use to promote wound healing or treat infection is of low certainty, with a lack of randomized controlled clinical trials on the intervention.[12][14][44] Thus, expert committees recommend reserving NPWT with application of cleansing solution to select cases, such as complex patients or complex wounds. See section on 'Indications' above.
- Coverage: NPWT with application of cleansing solution (intermittent or continuous) is considered experimental by most health insurers, and is thus non-covered.[45]
- See application techniques in the topic "How to Select and Apply Negative Pressure Wound Therapy Devices"
Risks and Complications
Potential negative consequences of NPWT include:
- Bleeding
- Pain, due to excessive ingrowth of foam into tissue. Some manufacturers recommend not exceeding 72 hours between dressing changes to decrease risk.[46] Some devices such as those with gauze interfaces or non-adherent layers built in to the dressing may be left in place longer.
- Peri-wound maceration (softening due to exposure to prolonged exposure to moisture or exudate)
- Retention of dressing materials that may contribute to wound infection/abscess
- Other injuries as cited by the FDA [47]
NPWT devices are usually worn continually by patients during treatment. They can interfere with mobility, and, anecdotally, can be noisy, which may cause sleep disturbances in some patients. [8]
Prescribing/Ordering
Prescription is required. Information needed for ordering:
- Patient demographics, insurance information
- History of any NPWT use in any wound (usually last 60 days)
- Wound etiology and location with narrative of patient history
-
- History of present illness/previous treatment history/failed therapies including products used
- Past medical history/comorbidities and treatment plan
- Presence of infection and current treatment/any current antibiotic therapy
- Nutritional status and plan if compromised
- Consequences or alternatives for patient if unable to obtain NPWT
- Duration of therapy (weeks/months)
- Frequency of dressing changes (for dressing and canister needs)
- Frequency depends on manufacturer recommendations for device and specific dressing type utilized, patient condition, wound severity, exudate levels, need for re-evaluation of wound bed, etc, but most NPWT dressings will be changed 2-3 times weekly. Incisional NPT or other smaller disposable systems may be left in place for 7-14 days. For specific information on a product, go to the product page using "Product Navigator" and find the frequency of replacement in the "Essentials Table", tab "Product Reimbursement".
- Follow-up for patient and plan of care for dressing changes (Home health, wound clinic, self change, caregiver, etc)
- Therapy start date
- Goal for therapy
- Prescriber information and signature
Documentation Requirements
Documentation of thorough wound assessment and dressing change procedure for NPWT dressing changes should be recorded at each visit for progress, reimbursement capture, and submission to insurance or durable medical equipment (DME), if NPWT product is durable (versus disposable).
- See NPWT Documentation Checklist
Minimum recommended documentation elements:
- Basic wound assessment including wound bed/surrounding periwound appearance, exudate, wound measurements/progress, associated pain, patient adherence as well as changes to plan of care including nutritional augmentation, pressure offloading, casting/orthotic use, and need for debridement (should be documented separately in procedural note).
- If ulcer is in lower extremity, blood supply to the ulcer should be checked upon physical examination (e.g., pulses palpation) and with non-invasive arterial tests such as ankle-brachial index (ABI), handheld audible Doppler, toe pressure, transcutaneous oximetry.
- Presence of any anatomical structures, sutures or other devices visible, and if this has changed from last assessment. If wound is on the abdomen, has wound odor changed or is effluent/succus present in dressing? Keep a vigilant eye for fistulization. Also note any skin reactions to dressing tape during subsequent changes.
- Wound changes or progress as a result of NPWT such as contraction, epithelialization, or increased granulation. Also note signs of non-healing such as rolled wound edges or excessive maceration, which necessitates re-assessment of therapy plan
- What was removed from the wound during the dressing change, how many pieces of foam, gauze, and any additional contact layers or products such as fistula isolation devices
- Device settings
- Utilization of products/dressings during reapplication including skin barriers, adjunctive products such as stoma rings or paste, and also document what is applied to wound bed including number of foam pieces or other physical barriers such as contact layers over exposed tissue structures
- Time needed for removal, assessment, and reapplication of therapy
- Pain management strategies used and tolerance of therapy
- If any assistance is required during change
This may seem cumbersome for the initial application of therapy, but will assist with evaluating ongoing effectiveness of therapy and will improve continuity of care.
PATIENT EDUCATION FOR CLINICIANS
Educating and providing resources for the patient and any caregivers on NPWT dressing application while you are applying the therapy can address overall knowledge deficit, and increase patient adherence to plan of care. It will also empower the patient and caregiver to troubleshoot the system at home if any issues arise. Education in the form of visual instructions, patient specific literature, manufacturer contact information, and clinician contact information will ensure your patient has the tools necessary to successfully continue therapy.
Clinicians should have the contact information for each manufacturer representative at hand in order to maintain competency in application of the various NPWT devices. Representatives often have written and virtual resources made specifically for patients who are prescribed therapy with units from their company.
In summary, clinicians can follow the steps below:
- Ensure patient can hear NPWT device alarm in case it goes off
- Instruct patient to call manufacturer or clinician if alarm goes off
- Provide patient with explicit instructions in the event therapy is interrupted. Common instructions include instructing patient to remove NPWT dressing if device stops working and therapy cannot be resumed within 2 hours. If NPWT system is removed, patient may apply gauze moistened with saline or other dressing as prescribed by provider during off hours or weekends, while waiting for follow up appointment.
CODING, COVERAGE AND REIMBURSEMENT
There are two sets of codes used for coding and billing of application of NPWT systems:
- Current Procedural Terminology (CPT) codes, used for provider and facility fees, and
- Healthcare Common Procedure Coding System (HCPCS) Level II, used to report cost of devices (these will be used either by the DME provider who is billing the payor directly, or by facility applying the device)
Typical Medicare reimbursement for NPWT is as follows:
- Medicare covers durable traditional durable NPWT devices/supplies, and provider/facility fees for patients in both outpatient and inpatient settings, provided criteria are met.
- Medicare does not separately reimburse disposable NPWT device/supplies. In inpatient settings, the disposable NPWT device/supplies and provider/facility fees are included in the MS-DRG payment. In outpatient settings (home health agencies, hospital-based outpatient wound care departments, physicians or qualified health professional offices), the American Medical Association established 2 CPT codes for disposable NPWT, which include reimbursement for provider/facility fees and device/supplies (97607 and 97608). Disposable NPWT device/supplies may be initiated upon presentation of the wound, whereas insurance coverage for standard NPWT requires the wound to be present for at least 30 days, during which wound is treated with standard care. [15]
- Incisional negative pressure systems are typically disposable NPWT device/supplies, and as such may be subject to the same coding and coverage guidelines. In inpatient settings, they are not separately reimbursed and are included in DRG payment. They are also not covered in ambulatory surgery (ASC) settings. The reimbursement is mainly limited to Hospital Based Outpatient Departments (HOPDs) and provider offices, where it is appropriate to utilize the two CPT codes for disposable NPWT which already include device and supplies.
CPT Codes
Medicare coverage of provider and facility fees related to application of NPWT systems is managed by Medicare Administrative Contractors (MAC), under Medicare Part A or Part B. Each jurisdiction may have its own specific local coverage determination and policies, as follows:
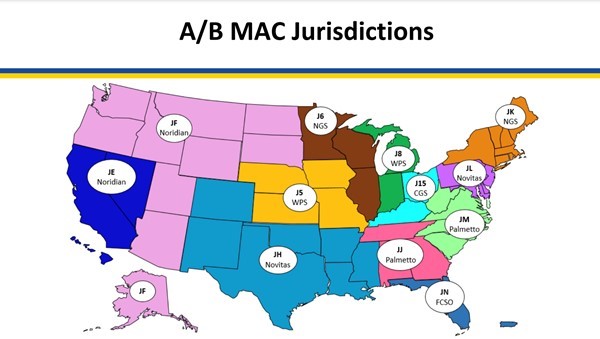
Medicare Administrative Contractors (MAC) as of 3/28/23
- Noridian:
- Negative Pressure Wound Therapy Pumps (L33821), updated on 01/01/2024 [48]
- Negative Pressure Wound Therapy Pumps - Policy Article (A52511) [49]
- Billing and Coding: Wound and Ulcer Care (A58565) - revision effective date 1/1/2024 [50]
- Billing and Coding: Wound and Ulcer Care (A58567) - revision effective date 1/1/2024 [51]
- Wound and Ulcer Care (L38902) [52]
- Wound and Ulcer Care (L38904) [53]
- National Government Services, Inc. (NGS)
- Outpatient Physical and Occupational Therapy Services (L33631) [54]
- Billing and Coding: Outpatient Physical and Occupational Therapy Services (A56566) [55]
- Wisconsin Physicians Service Insurance Corporation (WPS)
- Billing and Coding: Wound Care (A55909) [56]
- Response to Comments: DL37228 Wound Care (A57846) [57]
- Response to Comments: Wound Care (DL37228) (A55910) [58]
- Wound Care (L37228) [58]
- Novitas Solutions, Inc.
- Billing and Coding: Wound Care (A53001) [59]
- Wound Care (L35125) [60]
- CGS Administrators, LLC
- Negative Pressure Wound Therapy Pumps (L33821) [61]
- Negative Pressure Wound Therapy Pumps - Policy Article (A52511) [62]
- Billing and Coding: Outpatient Physical and Occupational Therapy Services (A57067) [63]
- Low frequency, non-contact, non-thermal ultrasound (CPT code 97610) (A56175) [64]
- Outpatient Physical and Occupational Therapy Services (L34049) [65]
- Palmetto
- Billing and Coding: Spiracur SNaP® Wound Care System (A53781) [66]
- Outpatient Occupational Therapy (L34427) [67]
- Outpatient Physical Therapy (L34428) [68]
- First Coast Service Options, Inc. (FCSO)
- Billing and Coding: Wound Care (A55818) [69]
- Wound Care (L37166) [70]
- Wound care (L37166): Medicare Part A/B local coverage determination (LCD) comment summary (A55757) [71]
CPT codes used for application of NPWT systems are listed below:
| CPT Codes | Description |
|
97605
|
Negative pressure wound therapy (e.g., vacuum assisted drainage collection), utilizing durable medical equipment (DME), including topical application(s), wound assessment, and instruction(s) for ongoing care, per session, total wound(s) surface area less than or equal to 50 square centimeters
|
|
97606
|
Negative pressure wound therapy (e.g., vacuum assisted drainage collection), utilizing durable medical equipment (DME), including topical application(s), wound assessment, and instruction(s) for ongoing care, per session, total wound(s) surface area greater than 50 square centimeters
|
|
97607*
|
Negative pressure wound therapy, (e.g., vacuum assisted drainage collection), utilizing disposable, non-durable medical equipment including provision of exudate management collection system, topical application(s), wound assessment, and instructions for ongoing care, per session; total wound(s) surface area less than or equal to 50 square centimeters
|
|
97608*
|
Negative pressure wound therapy, (e.g., vacuum assisted drainage collection), utilizing disposable, non-durable medical equipment including provision of exudate management collection system, topical application(s), wound assessment, and instructions for ongoing care, per session; total wounds(s) surface area greater than 50 square centimeters
|
*97607 and 97608 replace the previous G0456 and G0457 therapy codes. The 97607 and 97608 codes include the pump, dressing and canister, and can only be billed when the entire system is provided to the patient. These two codes should not be used if only a dressing change is performed for a disposable NPWT system.
HCPCS Level II
Medicare coverage of NPWT devices is managed by Durable Medical Equipment Medicare Administrative Contractors (DME MAC). As it relates to NPWT devices, DME MAC Jurisdictions (A,B,C and D) follow the same local coverage determination (LCD) and policy:
-
L33821: Negative Pressure Wound Therapy Pumps
-
A52511: Negative Pressure Wound Therapy Pumps
The LCD and Policy articles are summarized below:
Level II HCPCS Code
| Description
| Coverage Criteria
|
|
E2402
|
Negative pressure wound therapy electrical pump, stationary or portable
|
A. For Ulcers and Wounds(*) in the Home Setting: - A complete wound therapy program described by criterion 1 and criteria 2, 3, or 4(**), as applicable depending on the type of wound, must have been tried or considered and ruled out prior to application of NPWT.
B. For Ulcers and Wounds(*) Encountered in an Inpatient Setting: - After wound treatments described under A-1 through A-4 have been tried or considered and ruled out, NPWT is initiated because it is considered in the judgment of the treating physician, the best available treatment option, OR
- The beneficiary has complications of a surgically created wound (for example, dehiscence) or a traumatic wound (for example, pre-operative flap or graft)
|
|
A6550
|
Wound care set for negative pressure wound therapy electrical pump, includes all supplies and accessories
|
|
A7000
|
Canister disposable, used with suction pump, each
|
|
A9272
|
Suction, disposable, includes dressing, all accessories and components, any type, each (products reported with this code are not covered by Medicare)
|
Not covered under DME benefit
|
* The beneficiary has a chronic Stage 3 or 4 pressure ulcer, neuropathic (for example, diabetic) ulcer, venous or arterial insufficiency ulcer, or a chronic (being present for at least 30 days) ulcer of mixed etiology.
** Criteria 1-4:
- For all ulcers or wounds, the following components of a wound therapy program must include a minimum of all of the following general measures, which should either be addressed, applied, or considered and ruled out prior to application of NPWT:
- Documentation in the beneficiary’s medical record of evaluation, care, and wound measurements by a licensed medical professional, and
- Application of dressings to maintain a moist wound environment, and
- Debridement of necrotic tissue if present, and
- Evaluation of and provision for adequate nutritional status
- For Stage 3 or 4 pressure ulcers:
- The beneficiary has been appropriately turned and positioned, and
- The beneficiary has used a group 2 or 3 support surface for pressure ulcers on the posterior trunk or pelvis (see LCD on support surfaces), and
- The beneficiary’s moisture and incontinence have been appropriately managed
- For neuropathic (for example, diabetic) ulcers:
- The beneficiary has been on a comprehensive diabetic management program, and
- Reduction in pressure on a foot ulcer has been accomplished with appropriate modalities
- For venous insufficiency ulcers:
- Compression bandages and/or garments have been consistently applied, and
- Leg elevation and ambulation have been encouraged
NPWT pumps and supplies will be denied if one (1) or more of the following are present [72]:
- Presence of necrotic tissue with eschar, if debridement is not attempted; or
- Osteomyelitis that is not concurrently being treated with intent to cure; or
- Cancer in the wound; or
- Presence of an open fistula to an organ or body cavity within vicinity of wound
Continued Coverage [72]
For wounds and ulcers described under A or B above, once placed on an NPWT pump and supplies, for coverage to continue, a licensed medical professional must do the following:
- On a regular basis:
- Directly assess the wound(s); and
- Supervise or perform NPWT dressing changes; and
- At least monthly, document changes in the ulcer’s dimensions and characteristics
When Coverage Ends [72]
For wounds and ulcers described under A or B above, an NPWT pump and supplies will be denied as not reasonable and necessary with any of the following, whichever occurs earliest:
- Criteria C1-C2 above cease to occur; or
- Per the treating physician, wound healing has occurred to the degree that NPWT may be discontinued; or
- Any measurable degree of wound healing has failed to occur over the prior month; or
- Four (4) months have elapsed using an NPWT pump in the treatment of the most recent wound; or
- Once equipment or supplies are no longer being used
For non-disposable NPWT
- The two CPT® codes of 97605 and 97606 can only be used when DME NPWT (e.g., KCI ACTIV.A.C.Therapy System) is being used (HCPCS II E2402, A6550, A7000). Do not use these two codes for application of disposable NPWT systems.
For disposable NPWT
- Disposable NPWTs (e.g., SNAP) are not considered DME under Medicare coverage policies.
- Use the two codes 97607 and 97608 after performing all of the services described in the code descriptors (including provision of exudate management collection system). For Medicare, the disposable NPWT may be coded as A9272 and is not separately reimbursed. Dressing changes without removal and replacement with a new device do not fall under the two aforementioned codes.
- For home health agencies, effective January 1, 2017, Medicare makes a separate payment amount for a disposable NPWT device for a patient under a home health plan of care.[73] Payment is equal to the amount of the payment that would otherwise be made under the Outpatient Prospective Payment System (OPPS).[73] Disposable NPWT devices are billed using the same codes 97607 and 97608 and include both performing the service and the device. The CPT code is reported with one of the 3 revenue codes, depending on the practitioner that provided the service: Skilled nurse – 0559, Physical therapist – 042x, Occupational therapy – 043x.
- When using revenue codes 042x or 043x, the HHA should not use the therapy plan of care modifiers (GO or GP) for NPWT services
REVISION UPDATES
| Date | Description |
| 7/10/19 | Expanded section on 'Indications'. Updated section 'Evidence and Recommendations' and 'Coding, Coverage and Reimbursement' |
| 4/22/19 | Updated section 'Coding, Coverage and Reimbursement' with new links to LCD and articles. Updated section 'Evidence and Recommendations' with new references |