ABSTRACT
Arterial or ischemic leg ulcers (AU) are leg ulcers that develop due to inadequate blood supply to the skin (arterial insufficiency). The decrease in blood supply may be caused by underlying peripheral arterial disease (PAD) that results from narrowing of the arteries to the legs (atherosclerosis), or may be caused by other non-atherosclerotic diseases. See sections 'Epidemiology', 'Risk Factors', 'Etiology' and 'Pathophysiology' below.
History: Patients with AU need a comprehensive assessment and history including risk factors for PAD, comorbidities, history of non-atherosclerotic conditions that involve the arterial system and may lead to AU, medication, nutritional, family and social history, and psychosocial status. A common presentation of AU to the wound clinician is a patient with asymptomatic PAD who undergoes a trigger (e.g. local minor trauma or infection) resulting in a non-healing ulcer. For a framework to conduct a history of a patient with AUs, see section 'Assessment'.
Physical Examination: A bilateral lower-extremity examination should be performed, with focus on signs and symptoms of ischemia and infection.[1][2][3] Evaluation includes inspection, palpation, lower extremity vascular assessment, peripheral neuropathy assessment, and ulcer examination. Special attention should be given to ulcer complications such as soft tissue infection, osteomyelitis, and gangrene.
Diagnosis: Objective noninvasive arterial testing is recommended for all patients with a lower extremity ulcer. The diagnosis of AU requires identification of arterial insufficiency as a significant or primary etiology of the ulcer, which is achieved through history and physical exam, and objective diagnostic evidence of impaired lower limb perfusion. [4][5][2]
- For all patients with suspected chronic limb-threatening ischemia (CLTI)/PAD, guidelines recommend ankle brachial index (ABI) and ankle pressure (AP) and as the first-line screening tests to detect PAD.[5][3] However, due to the high prevalence of arterial calcification among patients with AU, ischemia should also be documented by at least one of the following non-invasive arterial test modalities: toe pressure, toe brachial index, continuous doppler wave ultrasound, transcutaneous oximetry (TcPO2) or skin pressure perfusion.[3][6][5][7] See section 'Noninvasive Arterial Tests'.
- Other vascular imaging modalities may be ordered. See section 'Vascular Imaging'.
Differential diagnoses: Mixed ulcers (i.e. ulcer with mixed etiology, for instance, arterial/venous, arterial/neuropathic, etc), diabetic foot ulcer, venous ulcers, pressure ulcers/injuries, skin manifestations of COVID-19 (e.g. chilblains-like lesions, acute limb ischemia due to hypercoagulability), and others.
Classification systems and characterization of AUs: For all patients with AU and CLTI, clinical guidelines recommend using the Society for Vascular Surgery Lower Extremity Threatened Limb Wound/Ischemia/Foot Infection (WIfI) classification system.[5][6]
Ulcer prognosis (healability) assessment: Categorization of wound healability (i.e., healable, maintenance, or non-healable) is of particular importance.[8] This designation defines for the clinician, patient, and family an expected course of action, plan of care, and healing rate. A framework is summarized in this topic.
Documentation: To support the diagnosis of AU, signs and symptoms must be attributable to objectively proven arterial insufficiency by noninvasive arterial tests. Proper documentation includes number and position of ulcers on the leg, wound measurements including area, perimeter, and depth, description of wound edge (pre and post debridement), peri-wound area, wound base quality, amount and type of drainage, any signs of infection, and previous history of debridement. Signs of improvement need to be documented to support medical necessity (as per Medicare requirements). Specific tools that facilitate standardized assessment and documentation of patients with AU should be used whenever possible. As part of a patient-centered approach, it is recommended that clinicians adopt patient-reported outcome measures to assess patient’s Quality of Life (QOL), pain, and depression. See documentation tools.
Treatment: See topics "Arterial Ulcer - Treatment", "Arterial ulcer - Surgical Treatment"
Prevention and Patient Education: See topic "Patient Education - Arterial Ulcer"
When to refer to specialists:
-
Vascular specialist: urgent referral if acute limb ischemia, manifested by six ‘Ps’ (onset<2 weeks): pain, pallor, pulselessness, poikilothermia (cold), paresthesias, and paralysis. Chronic limb-threatening ischemia (onset >2 weeks): ischemic rest pain, nonhealing wound/ulcers, or gangrene attributable to objectively proven arterial occlusive disease.[9]
- See other specialists in section 'Referrals to other specialists'
ICD-10 Coding: See 'Appendix - ICD-10 Coding'
Clinical Guidelines and Quality Measures: See topic "Arterial Ulcers - Overview"
Background
- Definitions:
-
- Arterial or ischemic leg ulcers (AUs): AUs are leg ulcers that develop due to inadequate blood supply to the skin (arterial insufficiency). The decrease in blood supply may be caused by underlying peripheral arterial disease (PAD) that results from narrowing of the arteries to the legs (atherosclerosis), or may be caused by other non-atherosclerotic diseases.
-
- The majority (around 70%) of leg ulcers are caused by venous disease. However, approximately 22% of people with leg ulcers have arterial insufficiency.[10][11]
- Ulcers that are caused solely by arterial insufficiency (i.e. pure AUs) are less common than ulcers of mixed etiology (i.e., mixed arterial/venous and mixed arterial/neuropathic).[12] Only 10% of people with leg ulcers have pure arterial insufficiency.[13]
- It is essential to differentiate AUs from venous leg ulcers (VLUs). VLUs are treated primarily with compression therapy, but this intervention may lead to necrosis if applied to AUs.[10]
- A common presentation of AU to the wound clinician is a patient with asymptomatic PAD who undergoes a trigger (e.g. local minor trauma or infection) that results in a non-healing ulcer.
- Peripheral arterial disease (PAD): also known as peripheral vascular disease, peripheral arterial occlusive disease, or arteriosclerosis obliterans, PAD is a chronic condition characterized by decreased blood supply to the upper or lower limbs, due to partial or complete arterial occlusion. In this topic, PAD refers to atherosclerotic disease of the lower limb.
-
- Patients with PAD may be asymptomatic or present with symptoms such as impaired walking, classic intermittent claudication, chronic limb-threatening ischemia or acute limb ischemia.[14]
- PAD is usually associated with higher cardiovascular morbidity and mortality.[14]
-
Chronic limb-threatening ischemia (CLTI): also known as critical limb ischemia, CLTI is an advanced manifestation of PAD. The term “chronic limb-threatening ischemia” is preferred over “critical limb ischemia”, as the latter implies threshold values of impaired perfusion rather than a continuum.[5]
-
- Around 3-10% of all patients with PAD have CLTI.[15][16] One of the clinical manifestations of CLTI is AU.
- CLTI is a clinical syndrome defined by “chronic ( ≥ 2 weeks) ischemic rest pain, non healing wound/ulcers, or gangrene in 1 or both legs attributable to objectively proven arterial occlusive disease.”[14] Venous, traumatic, embolic, and nonatherosclerotic etiologies (e.g. vasculitides, collagen vascular disease, Buerger's disease, neoplastic disease, dermatoses, and radiation arteritis) are excluded from etiologies leading to CLTI. [5]
- All patients with suspected CLTI should be referred to a vascular specialist.[5]
- Acute limb ischemia (ALI): ALI is a sudden reduction in arterial supply to the limb, which could threaten its survival. ALI often occurs due to thromboembolic events such as an embolic event related to atrial fibrillation, trauma, or occlusion of a stent or bypass graft, etc.
-
- ALI is considered an emergency, and the delay in its management could yield permanent disability, amputation, or death; 4-6 hours without management could result in excessive tissue necrosis and damage.
- Typically, the onset is < 2 weeks (in contrast with CLTI, whose onset is ≥ 2 weeks).[17]
- Relevance:
-
- Financial burden:
-
-
AUs: among chronic ulcers, AUs pose the largest financial burden to Medicare in the U.S. (the mean Medicare spending per wound in 2014 was $9418).[18]
-
CLTI: it is estimated that the total Medicare cost for treating cases of CLTI (including rest pain and tissue loss such as AU or gangrene) is $12 billion per year, and the cost per patient per year is estimated to range from $49,700 to $55,700.[1][19]
- Amputation: in addition to the financial burden, CLTI patients have an increased risk of limb amputation. Amputation affects quality of life, impairs function and productivity, has negative psychological impacts, and increases the risk for other comorbidities and mortality.
-
- About 29% of patients with CLTI die or undergo major amputation in the first year.[19][20] The median survival of patients with CLTI is only 3.5 years, shorter than that for patients with cancer, stroke, or heart failure.[19][20]
-
The 5-year mortality rate after major amputation due to CLTI is about 70%, that is, twice the 5-year cancer mortality rate in the United States. As many as 50% of CLTI patients proceed directly to amputation without undergoing vascular assessment or revascularization.[12]
Epidemiology
Prevalence
-
AUs: In the United States, the prevalence of AUs among Medicare beneficiaries in 2014 was estimated to be 0.4%.[18]
-
CLTI: the estimated prevalence of CLTI among Americans is 1.3%.[1]
-
- Around 3-10% of all patients with PAD have CLTI.[15][16]
- Nearly 5-10% of patients with asymptomatic PAD or intermittent claudication develop CLTI after 5 years. [1]
-
PAD: PAD affects approximately 8.5 million people above the age of 40 years.[1][21] Prevalence of PAD dramatically increases with age, representing about 20% in octogenarians. Of note, these rates are nearly twice as high in African Americans in comparison to other races, at any given age.[14]
Incidence
-
AUs: The monthly incidence rate of AU is estimated to be 1.8 cases per 100 patients with advanced illness in Canada. [22]
-
CLTI: The estimated incidence of CLTI in the United States is about 3500 cases per one million.[1]
-
PAD: In the last decade, incidence of PAD in Europe has decreased, whereas in the U.S incidence has also decreased for females but has increased for males.[23]
-
- In 2017, the incidence rate of PAD in the United States was 202.3 and 171.1 per 100,000 among males and females, respectively.[23]
Mortality rates
-
PAD: In the last decade, despite the decreased incidence of PAD in Europe and in the United States, mortality has increased (by 140-158% in the United Kingdom, and by 135-131% in the United States). [23]
-
- The most common causes of death in patients with PAD are coronary and cerebrovascular disease, which account for 40%-60% and for 10%-20% of occurrences, respectively. [14]
Amputation rates
-
PAD: Major lower limb amputations in patients with PAD and intermittent claudication have been reported in 1% to 3% of patients over a 5-year period. [14]
-
CLTI: In the presence of CLTI however, the risk of a major amputation within 1 year is of approximately 50%. [14]
Risk Factors
- Most AUs develop due to atherosclerosis, but AUs can also be a result of non-atherosclerotic causes.[1]
- The American College of Cardiology (ACC) and American Heart Association (AHA) guidelines have identified four groups of patients who are at increased risk of PAD [1][2][9]:
- Patients 65 years of age or older. [24]
- Patients 50 to 64 years of age with risk factors for atherosclerosis (i.e., smoking, diabetes mellitus, hypertension, hyperlipidemia, chronic kidney disease) or family history of PAD.
-
Smoking: increases the risk of PAD by 2- to 4-fold.[14] Second hand, passive exposure to cigarette smoke appears to increase vascular endothelial inflammation to a similar extent as active cigarette smoking, thus increasing the risk for atherosclerotic plaque development in children and adults. [25]
-
Diabetes: increases the risk of PAD by 2- to 4-fold, similar to smoking.[14]
-
Hypertension: increases the risk of PAD by 0.5- to 1.2-fold.[14]
-
Dyslipidemia: high cholesterol/low-density lipoprotein (LDL) increases the risk for PAD; however, high-density lipoprotein (HDL) reduces the risk.[14]
- Patients younger than 50 years with diabetes mellitus and one or more additional risk factors for atherosclerosis (see above).
- Patients with known atherosclerotic disease in another vascular bed (coronary, carotid, subclavian, renal, mesenteric artery stenosis, or abdominal aortic aneurysm)
- Each cardiovascular risk factor seems to be associated with specific patterns of lower limb atherosclerosis disease (Figure 1).[5][26]
-
As previously mentioned, AUs may also result from non-atherosclerotic etiologies. Risk factors for development of AUs in those cases include the presence of pathological conditions that interfere with adequate blood supply and wound healing (See section ‘Etiology’ below).
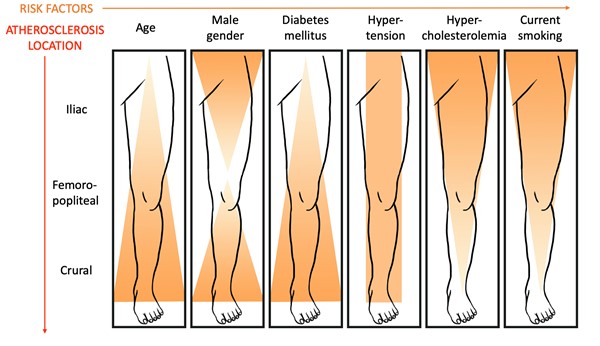
Fig 1. Patterns of atherosclerotic disease and associated risk factors [5][26]
Etiology
Inadequate tissue perfusion is the primary etiology in the pathogenesis of AUs.[15] Inadequate tissue perfusion most frequently occurs due to atherosclerosis and PAD, however similar clinical manifestations may result from non-PAD conditions.[1] Non-PAD conditions should be considered when an AU is present without the classic atherosclerotic risk factors and without objective evidence of atherosclerosis in vascular beds other than those in the lower extremities [27]:
-
PAD: PAD is the most prevalent etiology of AUs.[27] PAD leads to narrowing of the limb arteries due to atherosclerosis of medium/large-sized arteries.
-
Non-PAD etiologies include [2][27]:
-
- Popliteal artery entrapment syndrome
- Cystic adventitial disease
- Endofibrosis of the iliac artery
- Chronic exertional compartment syndrome
- Fibromuscular dysplasia
- Idiopathic mid-aortic syndrome
- Arterial manifestations of pseudoxanthoma elasticum.
- Vasculitis: thromboangiitis obliterans (also known as Buerger’s disease)
-
- Thromboangiitis obliterans is an inflammatory disease of the small and medium vessels of the hands and feet. The disease may cause claudication, rest pain, ulceration, or gangrene. It is associated with smokers and these patients are typically younger than patients with claudication due to PAD. [2]
- Various hematologic conditions (e.g., thalassaemia, and sickle cell disease, factor V Leiden thrombophilia)
- Various environmental conditions (e.g. pernio and frostbite).
- COVID-19 with thrombosis [28][29]
Pathophysiology
Inadequate tissue perfusion plays a major role in the pathogenesis of AUs. The pathophysiology of AUs resulting from PAD is often illustrated through a model of atherosclerosis (Figure 2). [14]
- In the normal artery, the healthy endothelium maintains arterial homeostasis through the balanced release of nitric oxide (NO) and endothelin-1. However, a hemodynamically significant arterial stenosis leads to tissue ischemia upon physical activity. Repetitive cycles of exercise-induced ischemia followed by reperfusion leads to the formation of reactive oxygen species (free radicals), which leads to endothelial activation and dysfunction. This in turn leads to an endothelin-mediated baseline vasoconstrictor tone and a prothrombotic milieu (see Table 1).[14]
- Thus, recurrent ischemia/reperfusion due to atherosclerosis leads to the following events that collectively lead to variable clinical symptoms [14]:
-
- Increased oxidative stress
- Altered metabolism
- Mitochondrial dysfunction
- Fiber type switching
- Altered myosin heavy chain expansion
- Myocyte apoptosis
- Muscle fiber denervation
- Fat infiltration
- Muscle atrophy
- AUs are often precipitated by trauma or infection. Limbs with arterial compromise may have minimal but adequate blood flow to maintain tissue viability. Trauma or infection increase demand for blood supply, leading to a non-healing ulcer. [15]
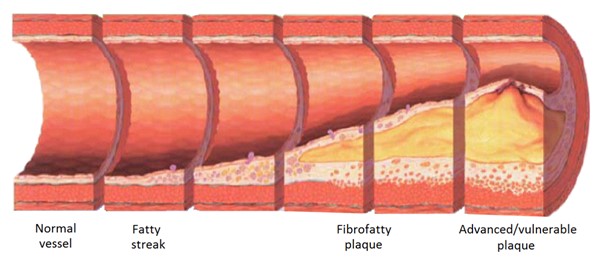
Fig. 2. Development of atherosclerosis. Atherosclerosis refers to the formation of fibrous and fatty plaques in the arterial wall. As the atherosclerotic plaque builds up, it can obstruct the arterial lumen (stenosis) or it can erode, decreasing blood flow and leading to ischemia.[30]
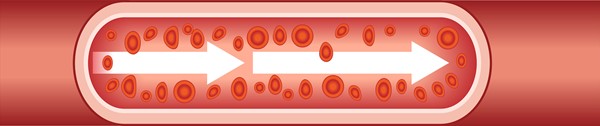
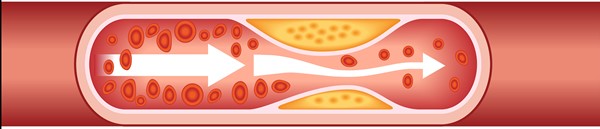
Table 1. Pathophysiology of peripheral artery disease and arterial ulcers. [14]
| Normal artery |
Artery with stenosis |
|
|
 |
- Flow: laminar, with normal shear stress
- Nitric oxide (NO)-dependent vasodilation
- Endothelial surface: antithrombotic
- Ankle-brachial index: normal
- Blood flow: increases with exercise
- Ischemia/reperfusion injury: not common
|
- Flow: turbulent, with abnormal shear
- Increased endothelin-1 vasoconstriction
- Endothelial surface: prothrombotic
- Ankle-brachial index: low
- Blood flow: does not increase with exercise
- Ischemia/reperfusion injury: recurrent
|
ASSESSMENT
Patients with AUs frequently present with other comorbidities that may impede healing. Thus, it is recommended that a qualified professional multidisciplinary team evaluate and perform a complete assessment of the patient. It is important to obtain a comprehensive history of the patient’s current condition, recurrence, past medical and surgical history, medications, and other risk factors related to AU and PAD, and perform a through physical examination, assessing for signs of ischemia and foot pulses.[4] Differentiating from other types of ulcers is key, as other ulcers require varying treatments that may be harmful for patients with AU (e.g. compression therapy for venous leg ulcers).[3]
For diagnosis of AUs, signs and symptoms must be attributable to arterial occlusive disease, objectively proven by non-invasive arterial tests such as ankle brachial index (ABI), toe-brachial index (TBI), transcutaneous oximetry (TcPO2), skin perfusion pressure (SPP), arterial duplex, computed tomography angiogram, or angiography.[2] See section 'Noninvasive Arterial Tests' below.
- Primary goals of assessment are:
-
- To assess blood supply to the ulcer
- To identify risk factors for AU or other ulcer etiologies, amputation, delayed healing, infection.
- To differentiate from other types of lower extremity ulcers, which may require different treatments. See 'Differential Diagnoses'
- To assess for infection and rule out urgent conditions affecting the lower extremity.
- Together with a vascular specialist, to classify the AU using the Society for Vascular Surgery Lower Extremity Threatened Limb Wound/Ischemia/Foot Infection (WIfI) classification system
- To determine healability, that is, if the ulcer has potential to heal with conservative interventions only. See 'Ulcer Healability'.
See Algorithm for Assessment of Arterial Ulcers below (Algorithm 1)
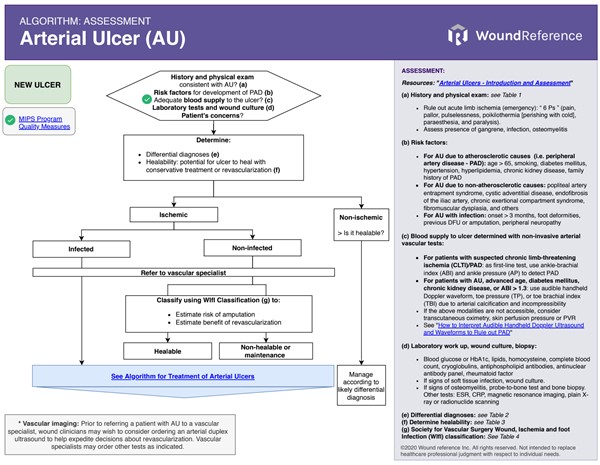 Algorithm 1. Algorithm for Assessment of Arterial Ulcers (click on this link to enlarge)
Algorithm 1. Algorithm for Assessment of Arterial Ulcers (click on this link to enlarge)
History
The non-exhaustive checklist below highlights relevant factors that should be assessed when taking history of a patient with an AU. Risk factors for development of AUs are indicated with the icon
Chief Complaint and History of Present Illness
-
Age: risk of atherosclerosis and PAD increases with age. See 'Risk Factors'
-
Ulcer: onset of the ulcer, recurrence, changes in size and presence of other ulcers. Frequently, patients with subclinical PAD present with a new AU triggered by trauma or infection as the first clinical manifestation.[1]
-
History of atherosclerosis and PAD : patients may be asymptomatic or have a history of [1]:
-
- Ischemic rest pain: constant burning sensation or numbness in the ankle or foot in the absence of activity. Pain on the foot typically occurs over the forefoot or metatarsal heads.[2] Leg elevation exacerbates pain, while placing the limb in a dependent position alleviates symptoms.
- Intermittent claudication: exercise-induced ischemic leg pain, usually relieved within 10 minutes of rest. Claudication may be absent in patients with predominantly infrapopliteal arterial disease, diabetic peripheral neuropathy or limited mobility from other medical conditions.
-
History of non-atherosclerotic conditions that involve the arterial system and may lead to AU : examples include popliteal artery entrapment syndrome, thromboangiitis, vasculitis, Raynaud, pyoderma gangrenosum, thalassemia, or sickle cell disease.[15] See section 'Etiology' above
-
Pain: pure AUs are usually painful. Assess pain onset, duration, location, precipitating and alleviating factors. Rest pain and/or intermittent claudication may be present, however peripheral neuropathy may mask pain.[3]
-
- Rest pain: typically occurs over the forefoot and metatarsal heads. If present for more than 2 weeks, it is a sign of advanced arterial disease (CLTI).[2]
- Claudication: may be unilateral or bilateral. It often affects calves, but can occur in the buttocks, hips, or feet depending on the level of the arterial disease.[2]
- History of other conditions that may lead to chronic non-healing ulcers: for instance, chronic venous insufficiency, peripheral neuropathy, pressure, inflammation, infection
- Past treatment history, including past surgical history: for instance, vascular and endovascular interventions for limb revascularization and amputation, prior surgical interventions that may have led to peripheral arterial occlusion.
Medications and Allergies
Assess medication history (prescribed and self-prescribed):
- Individuals with AU may be receiving medical therapy to reduce cardiovascular and limb-related events as recommended by clinical guidelines, such as [5][1] :
-
- Antiplatelet monotherapy with clopidogrel or aspirin.
- Moderate- or high-intensity statin therapy
- Angiotensin-converting enzyme inhibitors to treat hypertension
- Metformin as a hypoglycemic agent in patients with type 2 diabetes mellitus and CLTI.
- Certain medications may induce thrombosis, resulting in venous or arterial events.[31]:
-
- Heparin: heparin-induced thrombocytopenia (HIT) is an adverse drug reaction characterized by thrombocytopenia and a high risk for venous or arterial thrombosis.[32]
- Warfarin: warfarin-induced skin necrosis is an infrequent side effect following administration of warfarin in individuals with a thrombophilic history or after administration of large loading doses of warfarin, particularly without simultaneous initial use of heparin [33][34]
- Oral contraceptive pills: may alter the balance between the different coagulation factors, and promote thrombosis
- Intravenous immunoglobulin administration: can lead to decreased blood flow by increasing blood viscosity [31]
- Medications that delay wound healing: anticoagulants, antimicrobials (various antibiotic classes), anti-angiogenesis agents (eg, bevacizumab, aflibercept), antineoplastic drugs, anti-rheumatoid drugs (eg, methotrexate, aspirin/nonsteroidal anti-inflammatory drugs [NSAIDs]), colchicine (anti-gout drug), topical hydrogen peroxide, topical iodine, full-strength 0.5% Dakin’s solution (sodium hypochlorite), nicotine, steroids, and vasoconstrictors.[35][36]
Assess history of allergies and iodine sensitivity (relevant in the context of contrast imaging studies, such as arteriography, computed tomographic angiography, etc).
Family History
Assess history of AU, PAD and other variants of atherosclerotic vascular disease among parents or siblings
- Several risk factors for atherosclerotic PAD and resulting AU may be inherited (e.g., dyslipidemia, diabetes, and hypertension) .[37]
Social History
- Occupational: work settings that increase susceptibility for trauma may increase risk for development of AUs in individuals with PAD . Individuals with mixed etiology ulcers (arterial and venous) who need to stand for prolonged time may present with delayed wound healing.
-
Tobacco: smoking (including second-hand passive smoke) increases the risk for PAD .[2][25]
-
Drugs: many illicit drugs can either cause wounds (not related to arterial insufficiency per se) or instigate scratching that can produce wounds.[12]
Review of Systems
- General:
-
- Age: risk of PAD increases with age .[24]
- Obesity: increases risk factors for PAD
- Cardiovascular:
-
- Presence of an AU and PAD could hint for more widespread atherosclerotic disease and other comorbidities (e.g. coronary heart disease, metabolic syndrome, cardiovascular disease, and stroke).[2][37]
- Hypertension: risk factor for PAD and AU .[38]
- Congestive heart failure and angina: may limit mobility and mask PAD symptoms such as intermittent claudication.[2]
- Vasculitis: thromboangiitis obliterans (also known as Buerger’s disease) is a non-PAD cause of AUs
- Respiratory:
-
- Chronic obstructive pulmonary disease may limit mobility and thus mask intermittent claudication.[2]
- Endocrine:
-
- Dyslipidemia: risk factor for development of PAD and AU .[38][2][9]
- Diabetes: risk factor for development of PAD and AU .[38]
-
- Peripheral neuropathy: may contribute towards development of a non-healing foot ulcer (i.e. mixed arterial and neuropathic ulcer), and can alter pain perception, masking rest pain or intermittent claudication. [2]
- Hypothyroidism: may be a risk factor for PAD among men .[39]
- Gastro-intestinal/ Genito-urinary
-
- Chronic kidney disease: risk factor for atherosclerosis and PAD .[1]
- Neurological
-
- Radiculopathy: differential etiology for rest pain. Radiculopathy causes lower extremity pain due to nerve root compression. Radiculopathy pain is described as sharp, occurs along the distribution of a particular nerve root and is often positional. The straight leg raise maneuver may be useful in diagnosis.[2]
- Musculoskeletal
-
- Some musculoskeletal diseases may limit mobility and mask PAD symptoms such as intermittent claudication.[2]
- Skin
-
- Psoriasis: patients with psoriasis demonstrate a higher prevalence of cardiovascular risk factors including PAD. It is not clear whether there is a causal relationship or if merely an association resulting from multiple shared risk factors.[40]
Nutritional Assessment
- Poor diet with high caloric intake and low fibers leads to dyslipidemia, which in turn increases the risk for PAD, metabolic syndrome, cardiovascular disease, and stroke.[3][41]
- Guidelines recommend referring patients with AU for nutritional counseling to identify nutritional and vitamin deficiencies.[3][41] If nutritional status is poor, no amount of revascularization will suffice to heal a wound. [12]
- The following tools may be utilized:
-
- Standardized tools such as the "Nestlé MNA" and "Self-MNA®" by Nestlé can be used to screen for malnutrition.
- Medicare Quality Payment Program, Quality Measure:
-
- "Process Measure: Nutritional Screening and Intervention Plan in Patients with Chronic Wounds and Ulcers"
- "Patient Reported Nutritional Assessment and Intervention Plan in Patients with Wounds and Ulcers"
- "Preventative Care and Screening: Body Mass Index (BMI) Screening and Follow-Up"
Patients’ and caregivers' concerns
Patient's and caregiver's concerns and psychosocial status should be assessed and taken into consideration when creating a treatment plan:
- Evaluate patient's concerns: pain and ability to carry out daily activities
-
- Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"
-
Patients with an amputated limb may experience phantom limb pain, which feels like it originates from the amputated limb even though the limb is absent. This pain is described as shooting, stabbing, boring, squeezing, throbbing or burning.[7]
- Evaluate psychosocial aspects of the patient, caregiver and family: cognitive, functional, emotional status, presence of depression, understanding of the wound and risk factors, preference for treatment, motivation for adherence to the care plan, financial concerns
-
- We recommend use of "Patient-Reported Outcome Tools" to assess aspects above and measure impact of interventions.[42] See section 'Documentation' below.
Physical Examination
A comprehensive, bilateral lower-extremity examination that looks for signs and symptoms of ischemia and infection is recommended.[1][2][3] Urgent conditions affecting the lower extremity should be ruled out. Those include acute limb ischemia, necrotizing fasciitis (wet gangrene), compartment syndrome, spinal complications, deep-vein thrombosis, among others.[43]
-
Acute limb ischemia: is a clinical emergency characterized by the six ‘Ps’ (pulselessness, pain, pallor, poikilothermia - cold, paraesthesia and paralysis). Refer to vascular specialist as soon as possible, as limb may be at great risk if not managed in a timely and effective manner. [21]
Inspection:
Skin
- Surgical scars: depending on the location, they may indicate previous revascularization, potential fibrosis and resulting arterial occlusion, etc.
-
- Abdominal scars: may indicate previous surgery to the aorta or iliac vessels.
- Groin: stretch the skin folds open
- Legs: assess the medial aspect of the legs for scars caused by lower-limb bypass surgery (e.g. femoral-popliteal or femoral-distal bypass procedure) [43]
-
Ischemic skin changes: purpura; atrophy of the skin/subcutaneous tissue/muscle; shiny and taut skin; distal hair loss and/or dystrophic nails.[3]
-
Color: an ischemic foot due to PAD may appear pink and relatively warm due to arteriovenous shunting. Erythema may also be caused by infection (e.g., cellulitis). [15]
-
Tissue loss on the foot or lower leg: varies from small AUs (Figure 3) to widespread gangrene with eschars and mummification (Figure 4).[1] See section 'Ulcer exam' below.
- Gangrene: black, stiff, devitalized tissue resulting from ischemia (Figure 4). While the hardened tissue is not painful, significant pain is often present in the transition between viable and ischemic tissue.
|
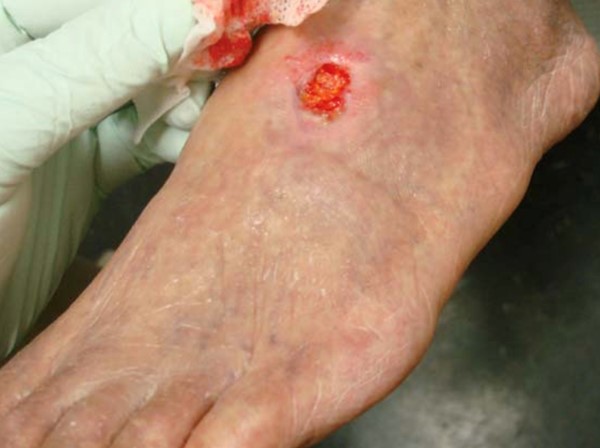
Fig. 3. Arterial ulcer on the dorsal aspect of the left foot
|
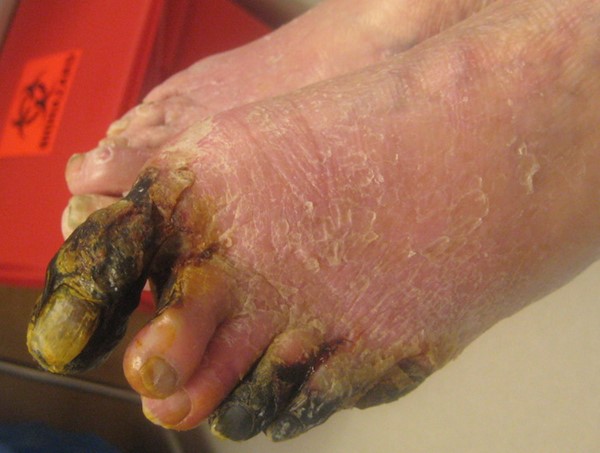
Fig. 4. Dry gangrene of the toes due to arterial insufficiency
|
Soft tissue
- Edema: assess for edema location (generalized/ localized, unilateral/ bilateral) and type (pitting/ non-pitting). Correlate edema with co-morbid conditions such as heart failure, nephropathy, venous insufficiency, lymphedema, phlebolymphedema.
-
- Pitting edema may be present if ulcer is of mixed venous and arterial etiology.
-
Wasting (cachexia): characterized by extreme loss of weight and skeletal muscle mass. Wasting can indicate ischemia, neurologic injuries, or long-term inactivity resulting from pain or a generally poor functional state.[43]
Bones
-
Amputations of the toes, feet, or legs (above or below the knee).[43]
-
Presence of deformities or fractures that may compress arteries and thus lead to ischemia.[43]
Palpation
- Skin temperature: focal or global decrease in skin temperature compared to the other foot may be predictive of either vascular disease or ulceration.[44]
-
Scars: palpate soft tissue near scars to identify any subcutaneous prosthetic grafts, such as bypass grafts.[43]
- Pulses: assess and document femoral, popliteal, dorsalis pedis and posterior tibial pulses.
-
-
Documentation of pulse strength [43]:
-
- If a pulse is absent, it should be documented as 0;
- If diminished, as 1+;
- If normal, as 2+;
- If bounding, as 3+
- Aortic pulse: to palpate the aorta, place both hands on the abdomen, above the umbilicus and slightly left of the midline. Auscultate the abdomen for bruits.
-
Femoral and popliteal pulses should be checked to assess for more proximal arterial disease (Figures 5 and 6).[43] Auscultate femoral/popliteal arteries for bruits.[3]
|
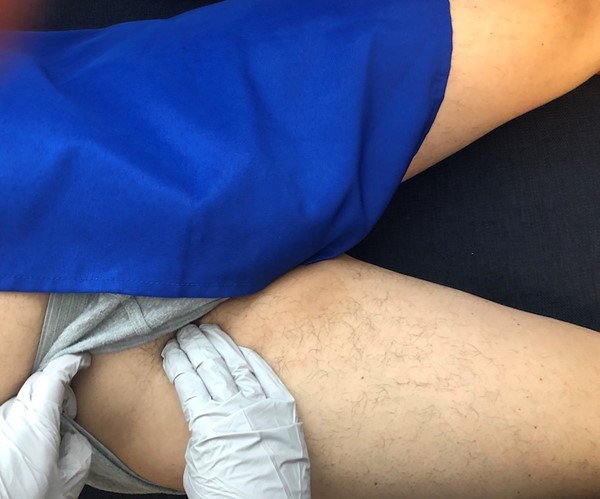
Fig. 5. Palpate the femoral pulse right below the groin crease, at the midinguinal point
|

Fig. 6. Palpate the popliteal pulse by pressing deeply into the popliteal space of a completely relaxed leg
|
-
Foot (pedal) pulses: palpate and determine whether the posterior tibial and dorsalis pedis pulses are “present” or “absent” (Figures 7 and 8).[3][44]
- If pulses are “absent”:
-
- A few healthy people have undetectable pedal pulses. The dorsalis pedis pulse is reported to be absent in 8.1% of healthy individuals, and the posterior tibial pulse is absent in 2.0%.[45][46]
- However, the absence of both pedal pulses, when assessed by an experienced clinician, strongly suggests presence of PAD.[47]
- If pulse is not palpable (e.g. due to edema), consider using a handheld Doppler to enhance examination
- If pulses are “present”:
-
- A palpable pulse does not rule out PAD, especially in people with diabetes suffering from medial calcinosis (abnormal deposition of calcium of the vessel wall).[45][46] More than two thirds of patients with PAD may have a detectable pulse.[48]
- Despite a palpable pulse, the ischemic ulcer might be situated in another angiosome whose blood supply is affected by PAD.[49]
- Even if pulse is palpable, the magnitude of clinical information derived from a pulse examination can be greatly enhanced with the use of a portable audible handheld Doppler.
|
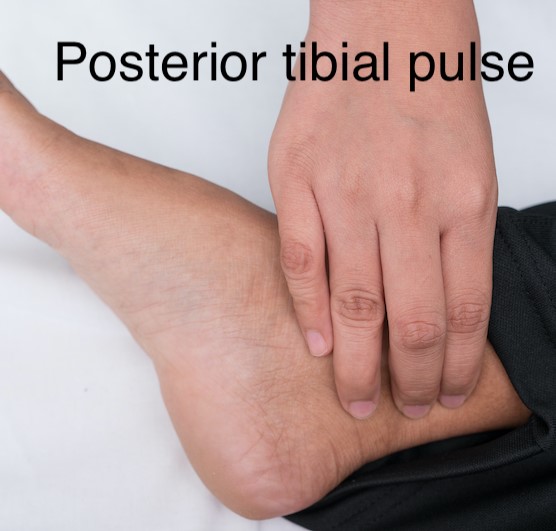
Fig. 7. Posterior tibial pulse
|

Fig. 8. Dorsalis pedis pulse
|
-
Capillary refill time: Gently press on the pulp of the toe or nail bed for 3 seconds and count the time needed for reperfusion to occur. Capillary refill greater than 3 seconds on dependency may indicate poor arterial perfusion.[43]
-
Portable audible handheld Doppler ultrasound: for all patients with AUs, we suggest use of a portable audible handheld Doppler ultrasound to enhance pulse examination and obtain additional clinical information on arterial patency, stenosis or occlusion.[1][12][50] For details, see topic “How to Interpret Audible Handheld Doppler Ultrasound and Waveforms to Rule out PAD".
-
- A non-palpable dorsalis pedis pulse may be “dopplerable” and show a biphasic or monophasic waveform. All patients with monophasic and some high-risk patients with biphasic waves (especially persons with diabetes) require further vascular studies and vascular surgery consults.[50]
- If a dorsalis pedis pulse is non-palpable and shows no audible signal by vascular Doppler the dorsalis pedis artery can be diagnosed as occluded with a great degree of certainty.[12][50] In these cases, revascularization is likely indicated.
-
Buerger sign: pallor of the foot on elevation and rubor (so-called sunset foot) on dependency is usually present in patients with CLTI and AU. [5]
-
-
Elevation pallor should be assessed by raising the affected leg to 60 degrees and observing the color of the soles of the feet.[12]
-
- No change in color: normal
- Pallor in a light skinned patient or ashen hue in a dark skinned patient indicates arterial perfusion deficit:
-
- Pallor within 25 seconds: severe arterial occlusive disease
- Pallor within 25 to 40 seconds: moderate arterial occlusive disease
- Pallor within 40 to 60 seconds: mild arterial occlusive disease
- If leg elevation leads to pallor, check for dependent rubor.
-
Dependent rubor: this is an important sign of PAD.[12]
-
- To assess dependent rubor, have the patient sit so that legs are hanging off the edge of the table and observe the color of the foot and lower leg. In patients with PAD, limb will be erythematous. When raised above the level of the heart, limb will normalize in color.
- This effect, suggestive of severe PAD, occurs due to the chronic dilatation (and loss of vasoconstriction) of dermal arterioles and capillaries despite increased hydrostatic pressure secondary to chronic ischemia. [51] This same effect is responsible for reperfusion edema after arterial revascularization. [12]
Assessment of peripheral neuropathy
Diabetes mellitus is a risk factor for PAD and AUs. Therefore, peripheral neuropathy may be present and contribute towards the development of a non-healing ulcer of mixed etiology (i.e. neuropathy and arterial).
- Assess loss of protective sensation (LOPS), motor neuropathy and autonomic neuropathy.[3] For details, see section ‘Assessment of Peripheral Neuropathy’ in topic “Diabetic Foot Ulcer - Introduction and Assessment”
Ulcer exam
-
Ulcer assessment: evaluate location, number and size of ulcers, edges, undermining, presence and type of nonviable tissue, signs of infection or biofilm, exudate type and quantity, periwound appearance (e.g., altered perfusion, maceration).[12][2]
-
Common AU characteristics (Figures 9 and 10) [2][12]:
-
- Anatomic location: distal toes, lateral ankle, or pressure points of the foot (i.e. often on bony prominences). If possible, determine the angiosomal distribution (see section 'Angiosome mapping' below)
- Size (width and length): measured in centimeters in two orthogonal planes, preferably one of which should be aligned with the patient’s head (for consistency)
- Depth: usually full-thickness
- Margins: well-demarcated edges, “punched-out” appearance.
- Tunneling or undermining: may be present
- Wound bed: pale, non-granulating, often necrotic base
- Exudate: low to none
- Periwound: capillary refill adjacent to the wound may remain normal even in PAD due to the presence of collateralization. However, if significantly delayed, severe PAD is present.
-
See history and physical exam typical findings of AU and associated conditions (e.g. mixed arterial and venous ulcer, neuroischemic ulcer) in Algorithm 1.
|
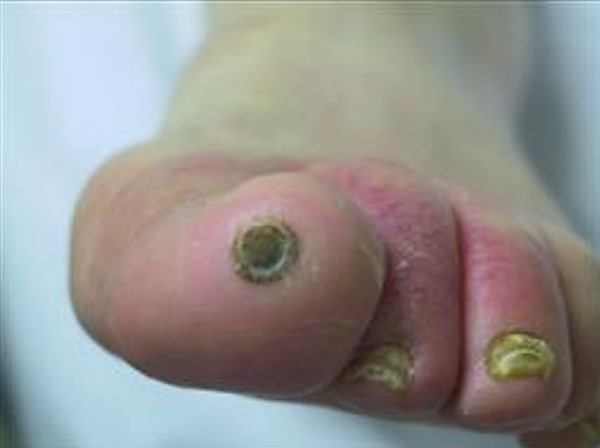
Fig. 9. Pure arterial ulcer on the tip of the great toe, without visible pressure component
|
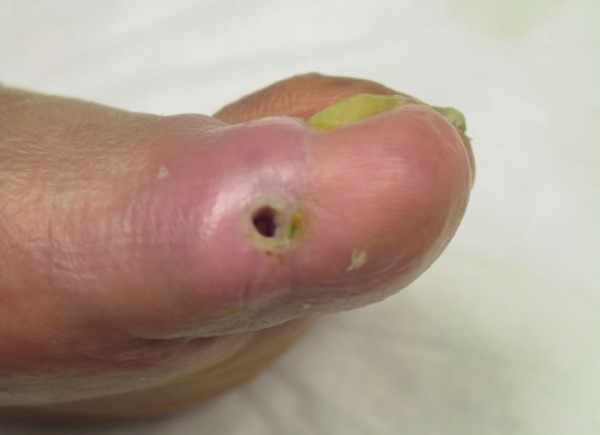
Fig. 10. Pure arterial ulcer on over interphalangeal joint, without visible pressure component
|
Ulcer complications
Soft Tissue Infection
Monitor signs of infection closely, as signs may be subtle due to reduced blood flow.[3]
-
1CFor patients with AUs and suspected limb infection, guidelines recommend immediate evaluation by vascular specialist, assessment of perfusion status and culture-guided antibiotic therapy (Grade 1C).[3][7]
-
-
Rationale: Infection of ischemic ulcers is a primary risk factor for major amputation.[7]
- The following mnemonics can help identify superficial and deep infections [8][52][49]:
-
- Superficial infection: The NERDS mnemonic can help identify soft tissue infection. If any 3 NERDS are present, superficial soft tissue infection is likely and topical antimicrobial treatment is justified. For arterial ulcers with superficial infection, topical antimicrobials alone may not be sufficient and systemic antibiotics may be needed.[7][53] NERDS stands for:
-
- Nonhealing ulcer
- increased Exudate
- Red-friable tissue
- Debris
- Smell
- Deep and surrounding infections: The STONEES mnemonic can help identify deep and surrounding infections. Systemic antibiotics and topical antimicrobial treatment are justified if 3 or more of the STONEES signs are present. STONEES stands for:
-
- increased Size,
- elevated Temperature of 3° F over a mirror image of the surrounding wound skin
- Os (latin for bone): probing to bone
- New breakdown or satellite areas of involvement,
- increased Exudate
- Erythema + edema (cellulitis)
- Smell
- Diagnosis is mainly clinical. Tissue biopsy or quantitative, validated swab cultures (e.g. Levine technique), or biofluorescent scans may be used to confirm diagnosis of infection (see ‘Diagnosis’ section below).[3] For details on the wound infection continuum and associated signs and symptoms, see section see section 'Relevance' in topic "Wound Culture - Swabs, Biopsies, Needle Aspiration'. Tissue biopsy or quantitative, validated swab cultures (e.g. Levine technique), or biofluorescent scans may be used to confirm diagnosis of infection. See topics "Wound Culture - Swabs, Biopsies, Needle Aspiration", "How to Collect a Wound Swab (Levine Technique) for Culture" and "How to Perform a Wound Biopsy".
The Wound, Ischemia, and foot Infection (WIfI) classification of the Society for Vascular Surgery (SVS) categorizes foot infection as mild, moderate and severe. See section ‘Classification’.
Osteomyelitis
Wounds that are chronic, large, deep, or overlie a bony prominence are at high risk for underlying bone infection. The presence of a 'sausage toe' (Figure 11) or visible bone is highly indicative of osteomyelitis.[47]
-
Probe-to-bone testing: In patients with suspected CLTI who have a foot ulcer, clinical guidelines recommend a probe-to-bone test to assess depth and the probability of underlying osteomyelitis.[5]
-
-
Probe-to-bone test: gently probe the ulcer with a sterile blunt metal probe (Figure 12). If the metal strikes bone (detected by its hard, gritty feel) in a patient at high risk for osteomyelitis, there is a high likelihood that the patient has osteomyelitis. In practice, for all open AUs with positive probe-to-bone test, clinicians might opt for further imaging to assess for osteomyelitis (e.g., magnetic resonance imaging). See section 'Diagnosis' below
|
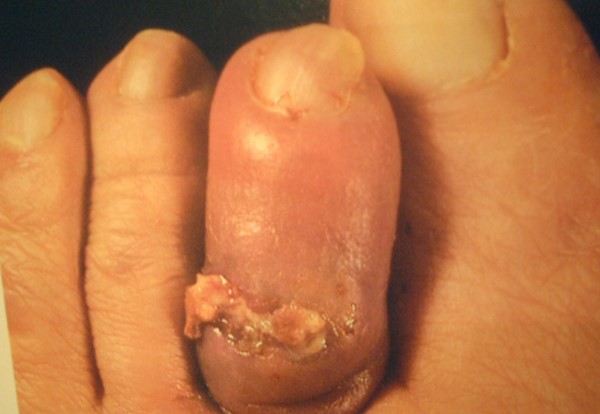
Fig. 11. Sausage toe suggestive of osteomyelitis [54]
|
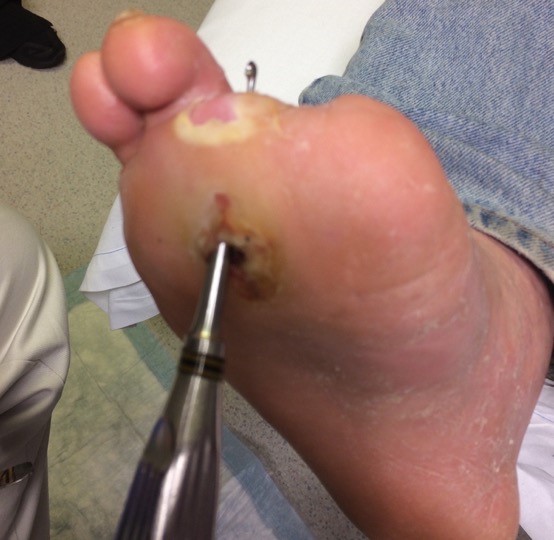
Fig. 12. Probe-to-bone in a DFU
|
Gangrene
- Dry gangrene is devitalized, ischemic tissue that eventually becomes black, hard and mummified (Figure 13). Gangrene itself is not painful, however the area surrounding it is often very painful. Gangrenous tissue may involve a small area or an entire limb, depending on the degree of the arterial disease. In smaller areas, gangrenous tissue often dry and fall off, exposing a skin lesion.
- Wet gangrene is a medical emergency caused by bacterial infections (e.g. necrotizing fasciitis, see Figure 14). This condition should be distinguished from dry gangrene, which results from ischemia.
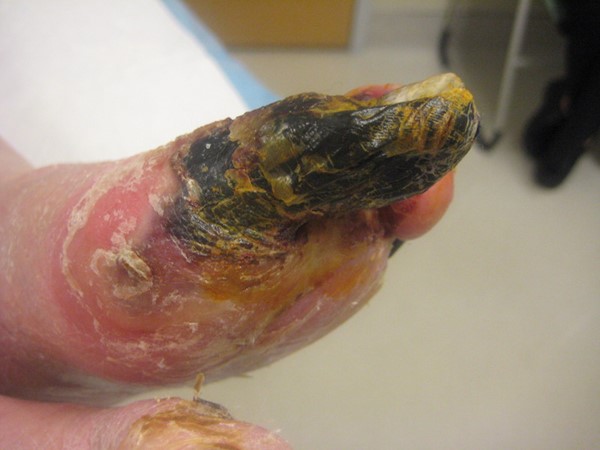
Fig. 13. Dry gangrene
|
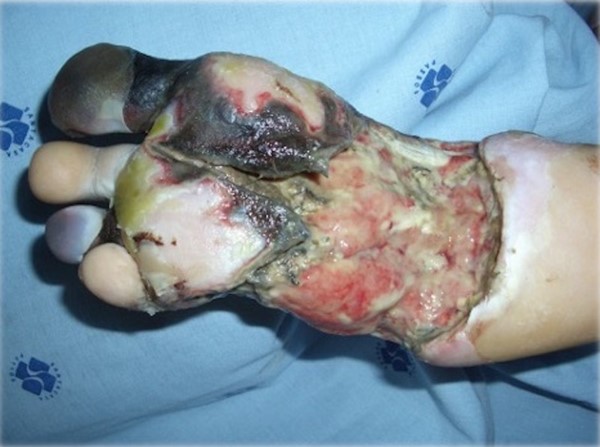
Fig. 14. Wet gangrene
|
Diagnosis
The diagnosis of AU requires identification of arterial insufficiency as a significant or primary etiology of the ulcer. This goal is accomplished through a comprehensive history and physical exam, and objective diagnostic evidence of impaired lower limb perfusion, which can be obtained with noninvasive arterial tests (e.g, ankle brachial index, absolute ankle pressure, absolute toe pressure, and others).[2][5] Diagnosis of PAD by manual palpation of lower limb pulses is a reliable method when resources are limited, however, a palpable pulse does not rule out PAD, particularly in diabetic patients with arterial calcification.[55] Given high prevalence of comorbidities among patients with chronic wounds, noninvasive arterial tests are recommended to rule out PAD in this population.[4]
Noninvasive arterial tests
Objective diagnostic evidence of impaired perfusion can be obtained through noninvasive arterial tests. For all patients with a lower extremity ulcer, noninvasive arterial tests are recommended to rule out PAD.[3][4][5][56][57][58][59] Guidelines recommend ankle brachial index (ABI) and ankle pressure (AP) as the first-line screening tests to detect PAD. For patients with advanced age, diabetes, chronic kidney disease or arterial calcification (ABI > 1.3), guidelines recommend measurement of toe brachial index (TBI) and toe pressure (TP) in addition to ABI to screen for PAD. If feasible and resources are available, for all patients with suspected CLTI/PAD or with a lower extremity ulcer, doppler arterial waveforms (DAW) or audible handheld Doppler ultrasound (AHDU) can be used as a screening test for PAD due to the higher sensitivity as an alternative to ABI.[60][56] In those instances, non-triphasic waveforms or the presence of clinical signs of PAD warrant further investigation with noninvasive arterial testing (e.g. ABI, TBI/TP) to confirm diagnosis of PAD.[60][61] For patients with AU, due to high prevalence of arterial calcification among these patients, ischemia should also be documented by TP, TBI, DAW or AHDU, transcutaneous oximetry (TcPO2 or TCOM) or skin pressure perfusion (SPP).[3][5][6][7] See Table 2 below.
Table 2. Bedside non-invasive arterial tests and likely interpretation (note: ABI, toe pressure, TBI values are frequently falsely elevated in patients with diabetes)
| Interpretation |
ABI (no diabetes) [6]
|
ABI (with diabetes)* [62]
|
AP (mmHg) [6]
|
TcPO2 or TCOM (mmHg) [63]
|
TP (mmHg)[6]
|
Pedal Doppler waveform [4][62][64]
|
TBI (diabetes) [4][62][64]
|
SPP (mmHg) [65][62]
|
| Non-compressible arteries |
>1.3 |
>1.3 |
|
|
|
|
|
|
| No relevant ischemia ** |
0.8-1.3
|
0.9-1.3 |
>100 |
>40 |
>=60 |
triphasic or
biphasic ***
|
>=0.70 |
>40 |
| Mild/ Moderate ischemia |
0.4-0.79
|
0.5 -0.89 |
50-100 |
30-40 |
30-59 |
biphasic or
biphasic/monophasic
|
0.25-0.69 |
30-40 |
| Severe ischemia |
<0.4 |
<0.5 |
<50 |
<30 |
<30 |
monophasic |
<0.25 |
<30 |
ABI: ankle brachial pressure, AP: ankle systolic pressure, TP: toe systolic pressure, TcPO2 or TCOM: transcutaneous oxygen pressure, TBI: toe brachial index, SPP: skin perfusion pressure
* Patients with diabetes should have pedal Doppler waveforms, in combination with ABI and TBI measurements. [4][6][63] If arterial calcification precludes reliable ABI or TP measurements, or if ABI is non-compressible (>1.3), ischemia should be documented by TcPO2, SPP, or PVR [6].
** No single modality has been shown to be optimal for the diagnosis of PAD, and there is no value above which PAD can be excluded. However, PAD is less likely in the presence of ABI 0.9 – 1.3; TBI ≥ 0.70; and triphasic or biphasic pedal Doppler waveforms. [4]
** Biphasic waveform may be normal in older individuals or when there is no clear transition from triphasic signal along the vascular tree [66].
NONINVASIVE ARTERIAL TESTs for determination of ulcer healing potential
- 1BFor all patients with lower extremity nonhealing wounds or ulcers, noninvasive arterial tests are recommended to rule out PAD and determine healability of the ulcer (i.e. healing potential) at the initial visit (Grade 1B).[3][4][5][56][57][58][59]
- Rationale: for all patients with lower extremity ulcerations (e.g., DFU, VLU and pressure ulcers of the lower extremity), and all chronic wounds of the lower extremity (e.g., traumatic wounds and nonhealing surgical wounds) that have not shown signs of improvement for >30 days, noninvasive arterial tests are recommended at the initial visit.[5][59] The results of these investigations assist the wound clinician to determine the etiology of the wound, predict healing capacity, and inform further management.[67] Comorbidities that contribute to lower extremity ulceration are also risk factors for PAD (e.g., diabetes, heart disease, advanced age, smoking, etc.), which is a major cause of limb loss.[59] According to a systematic review that included 40 studies, there is currently no single bedside test or a combination of bedside tests that has been shown to have superior diagnostic accuracy for PAD in people with diabetes with or without DFU.[68] However, an ABI <0.9 or >1.3, TBI of <0.70, and absent or monophasic pedal Doppler waveforms are useful to identify the presence of PAD.[68] See summary of guidelines recommendations in the 'Appendix' below.
- Indications:
- Noninvasive arterial tests are indicated for all patients with lower extremity ulcerations (e.g., DFU, VLU and pressure ulcers of the lower extremity), and all chronic wounds of the lower extremity (e.g., traumatic wounds and nonhealing surgical wounds) that have not shown signs of improvement for >30 days:
- At the initial visit for the wound or ulcer, at least once in a 12-month period.[59]
- Every 3-6 months for patients with nonhealing DFU wounds (ABI/TBI values can decrease over time, even if normal at first evaluation). [58]
- If there is clinical deterioration of the ulcer (e.g. increased pain, gangrenous changes, etc)
- Noninvasive arterial tests are indicated annually for patients with a prior history of DFUs or lower extremity arterial disease and other known risk factors.[58]
- For an algorithm on how to incorporate noninvasive arterial testing when evaluating a patient with nonhealing wounds, see topic "How to Assess a Patient with Chronic Wounds".
- Medicare Quality Payment Program, Quality Measure: "Non Invasive Arterial Assessment of patients with lower extremity wounds or ulcers for determination of healing potential"
- Quality measure: noninvasive arterial assessment of patients with lower extremity wounds or ulcers upon the first visit is a quality measure utilized in the Centers for Medicare & Medicaid Services (CMS) Quality Program. In 2020, clinician performance ranged from 0% to 100% with an average of 57.6% for practitioners that submitted data to CMS 44.6% among clinicians who did not submit quality data.[59] For quality measures, see topic "Arterial Ulcers - Guidelines and Quality Measures".
- Coverage: noninvasive arterial tests are covered by CMS as long as requirements are met. Covered peripheral arterial study testing methods include duplex scans; Doppler waveform or spectral analysis; volume, impedance or strain gauge plethysmography; and transcutaneous oxygen tension measurement. Providers should consult their Medicare Administrative Contractor (MAC) for specific requirements and limitations. See topic "Medicare Coverage Determinations for Wound Care".
- Non-invasive peripheral arterial examinations, performed to establish the level and/or degree of arterial occlusive disease, are medically necessary if (1) significant signs and/or symptoms of possible limb ischemia are present, (2) the patient is a candidate for invasive/surgical therapeutic interventions, 3) are performed by appropriately trained providers as specified by CMS. Acute ischemia is characterized by the sudden onset of severe pain, coldness, numbness and pallor of the extremity. Chronic ischemia can be manifested by intermittent claudication, pain at rest, diminished pulse, ulceration, and gangrene.[69][70][71]
- A routine history and physical examination, which includes ankle/brachial indices (ABIs), can readily document the presence or absence of ischemic disease in the majority of cases. It is not medically necessary to proceed beyond the physical examination for minor signs and symptoms such as hair loss, absence of a single pulse, relative coolness of a foot, shiny thin skin, or lack of toe nail growth unless related signs and/or symptoms are present which are severe enough to require possible invasive intervention.[69][70][71]
- An ABI is not a reimbursable procedure by itself; rather, ABI may be reimbursed when derived from a more comprehensive procedure which includes a permanent chart copy of the measured pressures and waveforms in the examined vessels. An ABI should be abnormal, e.g., and must be accompanied by another appropriate indication before proceeding to more sophisticated or complete studies, except in patients with severe diabetes or uremia resulting in medial calcification as demonstrated by artifactually elevated ankle blood pressure.[69][70][71]
- Documentation: providers should consult their MAC Articles for specific documentation requirements. In general:
- Medical record documentation maintained by the ordering/referring physician/nonphysician practitioner must clearly indicate the medical necessity of non-invasive physiologic studies of the upper or lower extremity arteries i.e., signs and symptoms, relevant history (including known diagnoses, and/or prior imaging). This documentation includes, but is not limited to, relevant medical history, physical examination, and results of pertinent diagnostic tests or procedures. Also, the results of arterial studies must be included in the patient's medical record. A hard copy or soft copy convertible to a hard copy provides a permanent record of the study performed and must be of a quality that meets accepted medical standards.[72]
- If the provider of non-invasive physiologic studies of arteries of the upper or lower extremity is other than the ordering/referring physician/nonphysician practitioner, the provider of the service must maintain a copy of the test results and interpretation, along with copies of the ordering/referring physician/nonphysician practitioner’s order for the studies.[72]
Ankle brachial index and ankle pressure
-
1BFor all patients with suspected CLTI/PAD (including patients with lower extremity ulcers), guidelines recommend ankle brachial index (ABI) and ankle pressure (AP) and as the first-line screening tests to detect PAD (Grade 1B).[3][5][57][58][56][73]
-
-
Rationale: ABI reports the ratio of highest arm to highest ankle blood pressure in the affected extremity.[1] Limitations include falsely elevated readings in patients with non-compressible arteries due to arterial calcification, often seen in patients with diabetes, renal insufficiency or advanced age.[5] About 80% of patients with diabetes mellitus and 20% of non-diabetic individuals develop arterial calcification.[74]
-
- An ABI between 1.00-1.3 is considered normal, and 0.91–0.99 is considered borderline.[7][56][73]
- If ABI results fall between 0.91 and 0.99 but the patient has symptoms of exertional leg ischemia or intermittent claudication, ABI is considered a borderline low ABI. In those cases, assessment of postexercise ABI should be taken into consideration to rule out PAD.[56]
- Although the criteria to evaluate postexercise ABI have not been standardized, postexercise ABI <0.90 or a drop of ABI >20% or ankle pressure drop >30 mm Hg are usually considered diagnostic of PAD.[56]
- For patients with nonhealing wounds or gangrene and a normal (1.00–1.30) or borderline (0.91–0.99) ABI, guidelines suggest using TBI with waveforms, transcutaneous oxygen pressure (TcPO2), or skin perfusion pressure (SPP) to diagnose CLTI/PAD. [73]
- An ABI <=0.90 is considered PAD.[56][73]
- An ABI < 0.4 or AP < 50 mmHg indicates severe ischemia.[1] See Table 2 above.
- If ABI falls between 0.4 and 1.3 but patient has a monophasic Doppler waveform or non-healing wounds/gangrene, further testing with TP/TBI, TCOM or SPP is recommended, as ABI values may be falsely elevated. [5][7]
- If ABI is >1.3, results are reported as non-compressible.[3] In those cases, additional vascular studies are needed to rule out PAD (i.e., pulse volume recordings, measurement of the toe pressures and TBI, transcutaneous oxygen measurements, or arterial duplex studies) [73][56]
- If ABI is inconclusive, TBI is indicated as a next step in evaluation.[75]
- See topic "How to Perform An Ankle Brachial Index"
Doppler Arterial Waveform and Audible Handheld Doppler
- 2CFor all patients with lower extremity ulcers, if feasible, clinicians might opt for bedside Doppler arterial waveforms (DAW) or audible handheld Doppler Ultrasound (AHDU) as a suitable alternative to ABI for first-line screening test for PAD (Grade 2C).[61][60][55]
- Rationale: evidence derived from cross-sectional and retrospective studies supports the use of DAW or AHDU as a potential screening tool for PAD instead of ABI, due to their higher sensitivity or higher specificity[61] in detecting PAD when compared to ABI in all patients, including persons with diabetes.[60], As shown by the San Diego Population Study, the addition of tibial artery Doppler assessment identified 20% additional diseased legs missed by the ABI.[56] AHDU waveforms are diagnostic of PAD and a triphasic pulse can reliably rule out significant PAD.[55]
- Doppler arterial waveforms (DAW): DAW analysis measures the quality of arterial blood flow as well as the level and severity of occlusion[60], and enables clinicians to detect occlusive disease despite calcified arteries in patients with diabetes, and to identify those at high risk of cardiovascular disease (CVD) and limb events.[56] Kashetsky et al. observed that DAW's sensitivities to detect PAD range from 81.87 to 100% and specificities range from 64.5% to 96%.[56] Because of the higher sensitivity of DAW compared with ABPI, the authors recommend DAW as a potential screening tool for PAD.[60] DAW can be measured with continuous Doppler or ultrasound probes in pulse-wave Doppler mode.[60] Doppler waveforms are interpreted by a grading system.[56] Limitations of DAW analysis include the feasibility of incorporating it into primary care practice, as the ability to produce accurate results depends directly on the operator's skills and experience.[60]
- Audible handheld Doppler Ultrasound (AHDU): AHDU utilizes Doppler waveforms as with DAW and may act as a reproducible equivalent to DAW.[60] Alavi et al. calculated a specificity of 97.5% and sensitivity of 42.8% to detect PAD, and concluded that AHDU proved to be a reliable, simple, rapid, and inexpensive bedside exclusion test of PAD in patients with or without diabetes.[61] Despite being operator dependent, AHDU results are not influenced by arterial calcification, edema, or woody fibrosis and can be used before commencing therapy (e.g., compression therapy) or referring the patient, although more definitive testing should be pursued.[61] Furthermore, the audible signal can be recorded in an MP3 format using a smartphone and included in a patient’s medical record or shared with a remote provider for faster clinical decision making.[60] For more details on AHDU see topic “How to Interpret Audible Handheld Doppler Ultrasound and Waveforms to Rule out PAD".
- Of note, Kashetsky et al. defined any waveform that was not triphasic or grade A as abnormal, whereas Alavi et al defined only monophasic waveforms as abnormal.[61][60] This difference in diagnostic threshold may explain the high sensitivity and low specificity of DAW in Katshetsky's review versus the high specificity and low sensitivity of AHDU in the study by Alavi et al.[60]
- Waveforms interpretation and considerations are summarized below:
- Triphasic: no relevant ischemia. A triphasic pulse can reliably rule out significant PAD.[55]
- Biphasic: may indicate mild/moderate ischemia or no relevant ischemia. Biphasic waveform may be normal in older individuals or when there is no clear transition from triphasic signal along the vascular tree [66]
- If higher risk for PAD (see 'Risk Factors' above) and/or if signs/symptoms of PAD (e.g. atypical leg symptoms, claudication, rest pain) are present, further testing should be considered to rule out PAD [60]:
- If peripheral arterial calcification is likely (i.e., if advanced age, diabetes mellitus, chronic kidney disease), TP/TBI is recommended.[3][5][6][7][56]
- If peripheral arterial calcification not likely, ABI or TP/TBI may be performed.[56]
- Arterial duplex ultrasound may be ordered, if available/feasible.
- Monophasic: may indicate severe ischemia, based on signs/symptoms of PAD (atypical leg symptoms, claudication, rest pain). As it is difficult to obtain ABI values in patients with monophasic signals, clinicians should confirm presence of PAD with TP/TBI or refer to vascular specialist for further testing (e.g. arterial duplex ultrasound).[61]
- No dopplerable pulses: refer to a vascular specialist for further investigation. Urgency of the referral is based on the presence of signs/symptoms of PAD (atypical leg symptoms, claudication, rest pain) or a limb threatening clinical presentation.
- If there is no indication for surgical revascularization, mitigate all comorbidities and control underlying causes (e.g. smoking, secondhand smoking), and provide adequate local wound care. See topic "Arterial Ulcer - Treatment".
- For suspected mixed arterial and venous leg ulcers, see algorithm 'based on audible handheld Doppler ultrasound or continuous waveform analysis' in topic "How to Select Adequate Compression Therapy Pressure Levels and Products" and topic "How to Interpret Audible Handheld Doppler Ultrasound and Waveforms to Rule out PAD“.
Toe pressure and toe brachial index
-
For patients with suspected arterial calcification (i.e. advanced age, diabetes mellitus, chronic kidney disease), or ABI > 1.3, guidelines recommend measuring toe pressure (TP) and toe brachial index (TBI) in addition to ABI in order to screen for PAD.[3][5][6][7][56]
-
-
Rationale: advanced age, diabetes mellitus, chronic kidney disease, or ABI >1.3 are associated with arterial calcification and vessel incompressibility, which may result in falsely elevated ABI. Guidelines recommend that regardless of ABI values, patients with these conditions have their TP and TBI measured, because medial calcification rarely affects digital arteries and thus TP/TBI are less susceptible to falsely elevated readings.[5][56] TBI's accuracy (the proportion of tests that were either true positive or true negative) to detect PAD has been shown to be consistent in patients with diabetes versus those without diabetes (74% versus 78%).[75] However, ABI's accuracy is lower in patients with diabetes (66%) compared to patients without diabetes (81%). Thus, the American Heart Association guidelines recommend simultaneous measurement of ABI and TBI in patients with diabetes or chronic kidney disease for detecting PAD.[56]
- Of note: for patients with diabetes, the Wound Healing Society recommends TBI as the first-line screening test.[55] In practice, the time required to perform an ABI is the most widely nominated barrier to conducting appropriate vascular assessment. A study with 119 participants has shown however, that ABI and TBI (without simultaneous ABI) have similar diagnostic accuracy (74% versus 73%), and that TBI without simultaneous ABI may save time when evaluating at risk populations (e.g. with diabetes, renal failure, or age > 75 years).[76]
- TP/TBI are less affected by incompressibility compared to AP/ABI. TP is measured with an appropriately sized mini-cuff typically placed around the base of the great toe and attached to a standard manometer. A photoplethysmographic or continuous-wave Doppler flow detector is then used to determine when flow returns while the inflated cuff is slowly deflated.[5]
- Patients with severe ischemia will have TP<30 mmHg and TBI<0.3. See Table 2 above.
-
-
1BFor all patients with AU, guidelines recommend measuring TP and TBI in addition to ABI/AP (Grade 1B).[5][77][78]
-
-
Rationale: TP is more sensitive than ABI/AP in the diagnosis of CLTI and more predictive of amputation risk.[5][77][78]
- 1CFor patients with diabetes and a foot ulcer or gangrene, guidelines recommend pedal Doppler waveforms in combination with ABI and TBI measurements to diagnose PAD.[4]
- Rationale: PAD is present in approximately half of the people with a DFU.[79] Therefore, in any person with diabetes and a foot ulcer or gangrene, PAD should be ruled out with the appropriate diagnostic strategies. All people with a DFU or gangrene should undergo a complete physical examination, including palpation of the lower limb pulses. Pulse palpation is considered the first step in a detailed evaluation of the limb, but when DFU is present further diagnostic procedures should be performed with non-invasive bedside testing techniques as clinical examination alone is not sufficient to exclude PAD. Evidence shows that no single modality has been shown to be optimal for the diagnosis of PAD among people with diabetes. However, PAD is less likely if ABI falls between 0.9 – 1.3; TBI ≥ 0.70; and if pedal Doppler waveforms are triphasic or biphasic.[4]
- The aforementioned non-invasive bedside tests (ABI, TBI, Doppler waveforms) are less resource intensive relative to other methods of diagnosing PAD such as angiography.[4]
Other noninvasive arterial tests
- When ankle and toe pressures, indices, and waveforms cannot be assessed (e.g. if an ABI cannot be measured due to noncompressible arteries at the ankle, or if TPs cannot be measured due to toe amputations), other methods for noninvasive assessment of perfusion should be considered [56][58] (e.g. pulse volume recording - PVR, transcutaneous oximetry - TCOM, or skin perfusion pressure - SPP).[5] Table 3 summarizes indications and limitations of each modality. Even if the above macrocirculatory tests are normal it would behoove the practitioner to assess the microcirulation as well (e.g. with TCOM, SPP), because patency not always equals perfusion.
noninvasive arterial tests for PROGNOSIS evaluation
- 2CFor patients with diabetes and a foot ulcer or gangrene, clinicians might opt to perform noninvasive arterial tests to asses the likelihood of healing and amputation (Grade 2C). [4][80]
- Ankle pressures (AP) and ABI: ankle pressure and ABI are weak predictors of healing. A low ankle pressure (e.g., < 50 mmHg) or ABI (e.g., < 0.5) may be associated with a greater likelihood of impaired healing and greater likelihood of major amputation.[4][80]
- Toe pressure (TP): TP ≥ 30 mmHg increases the pre-test probability of healing by up to 30% and a value < 30 mmHg increases the pre-test probability of major amputation by approximately 20%.[4][80]
- Transcutaneous oximetry (TCOM) or SPP: if TP cannot be performed, consider performing a TCOM measurement or a SPP to assess likelihood of healing.[4]
- A TcPO2 ≥ 25 mmHg increases the pre-test probability of healing by up to 45% and value < 25 mmHg increases the pre-test probability of major amputation by approximately 20%.[4][80]
- An SPP ≥ 40 mmHg increases the pre-test probability of healing by up to 30%.[4][80]
Table 3. Noninvasive arterial tests
Noninvasive arterial test
|
Indications |
Limitations |
Ankle-brachial index (ABI)
|
- ABI is the first-line test for the screening and diagnosis of PAD.[5][3]
- ABI has high sensitivity and specificity in the detection of severe PAD (84.7% sensitivity, 97% specificity) in patients without diabetes.[74]
- Among patients with diabetes, ABI sensitivities range from 53% to 100% and specificities range from 88% to 95.8%.[75]
|
-
For patients with diabetes, renal insufficiency or advanced age, ABI may be falsely elevated due to arterial calcification. In those cases, further testing with waveforms, TP or TBI is recommended.[5][64]
|
| Toe-brachial index (TBI) and toe pressure (TP) |
-
Indicated for patients with AU, advanced age, diabetes mellitus, chronic kidney disease, ABI > 1.3, or patients a normal ABI and with a monophasic Doppler waveform or non-healing wounds/gangrene [5][3][7][6]
- TBI tests both large and small vessels of the foot and thus does not provide reliable readings for patients with Raynaud’s disease or scleroderma.[74]
|
- TBI tests both large and small vessels of the foot and thus does not provide reliable readings for patients with Raynaud’s disease or scleroderma. [74]
|
| Audible handheld doppler ultrasound (AHDU) and dopper arterial waveforms (DAW) |
-
Indicated for patients with advanced age, diabetes mellitus, chronic kidney disease, ABI > 1.3, or patients a normal ABI and non-healing wounds/gangrene [5][3][7][6], or
- Active ulcer is present over the area of blood pressure cuff occlusion, or
- Severe local pain limits the ability to perform the ABI test, or
- Patient has lower limb lymphedema [81]
- Dopper arterial waveforms (or AHDU) have been suggested as the first-line screening test to rule out PAD in patients with lower extremity ulcers, as a suitable alternative to ABI.[61][60][55]
- Among patients without diabetes, DAW sensitivities range from 81.87 to 100% and specificities ranged from 64.5% to 96%.[75]
- Among patients with diabetes, DAW sensitivities range from 81.8% to 100% and specificities ranged from 66% to 93.2%.[75]
|
- Interpretation of audible Doppler signals or waveforms requires training
- DAW results are operator-dependent. Also, several factors can alter the Doppler waveforms and decrease specificity, including the potential for vasodilation secondary to heat leading to a decrease in early diastolic reversal of flow, uncompensated congestive heart failure dampening waveforms following exercise, and sympathetic nerve system signals influencing postexercise blood flow and DAWs. It is recommended that patients rest supine for 5 to 10 minutes before waveforms measurements are taken.[60]
|
| Transcutaneous oximetry (TcPO2 or TCOM) |
-
Indicated for patients with AU, advanced age, diabetes mellitus, chronic kidney disease, ABI > 1.3, or patients a normal ABI and with a monophasic Doppler waveform or non-healing wounds/gangrene [5][3][7][6],
- If TBI, TP, doppler ultrasound and waveforms and are not available
|
- Several factors can affect TcPO2 readings.Those include obesity, edema, infection, inflammation, scar tissue, irradiated tissue, sclerosis, patient position, skin temperature.[74]
- See topic "Transcutaneous Oximetry"
|
| Skin perfusion pressure (SPP) |
-
Indicated for patients with AU, advanced age, diabetes mellitus, chronic kidney disease, ABI > 1.3, or patients a normal ABI and with a monophasic Doppler waveform or non-healing wounds/gangrene [5][3][7][6],
- If ulcers on the toes or previous digital amputations preclude measurement of toe pressures [65]
|
- Requires costly equipment and training
|
Angiosome mapping
- For patients with AUs on the midfoot or hindfoot, advanced diagnostics might include evaluation of foot angiosomes with a handheld Doppler ultrasound.[5][12][82]
-
- Angiosomes are a concept described by Taylor and Palmer (Figure 15).[83] These authors defined angiosome as a three-dimensional network of vessels that are present in all tissue layers between the skin and the bone. According to this concept, the location of an ulcer may offer a hint on which arteries are affected by stenosis or occlusion, in light of the corresponding angiosome.
- Assessment of angiosomes may be helpful in planning for revascularization if the AU is on the midfoot or hindfoot, but it is likely to be of marginal value for most forefoot lesions and minor ulcers.
-
- For AUs on the midfoot or hindfoot, a unique angiosome may be identified in up to 80% of patients. However, for toe lesions, which typically represent more than half of the lesions encountered, assignment to a unique angiosome is not possible, as toes have a dual blood supply (anterior tibialis and posterior tibialis).[5]
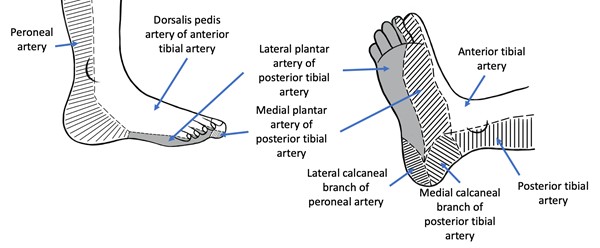
Fig. 15. Foot and leg angiosomes
Infection Work Up
Soft tissue infection
Soft tissue infection should be diagnosed clinically, based on the presence of local or systemic signs or symptoms of inflammation. See section ‘Ulcer complications - Soft Tissue Infection’ above.
Tissue biopsy or quantitative, validated swab cultures, or biofluorescent scans may be used to confirm diagnosis of infection and to guide antibiotic therapy. [3]
- For wound culture collection methods (e.g. Levine technique), see section ‘Soft Tissue Infection - Wound Culture’ in topic “DFU - Introduction and Assessment”
Osteomyelitis
- Accurate diagnosis of osteomyelitis can be difficult, as the clinical presentation varies and signs may not be obvious. All open infected AUs need to be inspected and gently probed with a sterile blunt metal probe (probe-to-bone test). See section ‘Ulcer complications - Osteomyelitis’ above.
- Definitive diagnosis is achieved with a bone biopsy. However, because bone biopsy results are not always easily obtained, clinicians must often use surrogate diagnostic markers. Therefore, once osteomyelitis is suspected, serum inflammatory markers such as erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), magnetic resonance imaging (MRI), plain X-ray or radionuclide scanning (e.g. WBC tagged study, nuclear medicine 3 phase bone scan) can be used to aid in diagnosis and monitor progress.
-
- ESR over 70 mm/h is clinically suggestive of osteomyelitis and can be used to monitor progression of osteomyelitis.[84]
- For more on diagnostic tests for osteomyelitis see section ‘Diagnosis: Diabetic Foot Infection - Osteomyelitis’ in topic “DFU Intro and Assessment”.
Laboratory Work Up
To accurately manage the patient with AU, the clinical order set should include the following laboratory tests [85]:
- Glucose
- Lipids: the following laboratory tests help identify risk factors for PAD[3]:
-
- Elevated low-density lipoprotein (LDL) cholesterol, total cholesterol and triglycerides; reduced high density lipoprotein (HDL) cholesterol.
- Elevated homocysteine
- Complete blood count
- Eosinophils
- Cryoglobulins
- Antiphospholipid antibodies
- Antinuclear antibody panel
- Rheumatoid factor
Vascular Imaging
Prior to referring a patient with AU to a vascular specialist, wound clinicians may wish to consider ordering an arterial duplex ultrasound to help expedite decisions about revascularization. Other vascular imaging modalities can be ordered by the vascular specialist, if the patient is identified as a surgical candidate. See Table 4 for a comparison of imaging modalities.
-
2BGuidelines suggest duplex ultrasound imaging (DUS) as the initial arterial imaging modality to determine the presence, extent and severity of arterial disease and to help inform decisions about revascularization (Grade 2B).[5]
-
- Rationale: DUS provides information on the anatomic location and extent of disease as well as information about flow volume and velocity. DUS imaging is usually the first choice for vascular imaging and in some health care settings may be the only modality available. In some cases however, it may be appropriate to proceed directly to computed tomography angiography (CTA), magnetic resonance angiography (MRA) or catheter digital subtraction angiography (DSA) instead of DUS imaging.
-
Advantages: although DUS has been shown to be inferior to other imaging techniques such as digital subtraction angiography (DSA), DUS offers many advantages as a first-line imaging modality, including its noninvasive nature, low cost, no iodinated contrast media, no ionizing radiation, and wide availability.[5][86]
-
Limitations: DUS is time-consuming and highly operator dependent. Furthermore, calcification may result in an incomplete examination. DUS is also poor at estimating collateral blood supply and reserve.[5]
For patients who are candidates for revascularization, additional vascular imaging is indicated. Invasive vascular imaging (e.g. CTA, MRA, catheter angiography), should only be obtained after the decision to intervene has been made. The decision to intervene is based on history and physical examination and on the potential for improvement in quality of life, and not solely on anatomical findings.
-
2BIn order to guide revascularization planning for patients who are candidates for revascularization, guidelines suggest evaluation with noninvasive vascular imaging studies (i.e. computed tomography angiography, magnetic resonance angiography) before invasive catheter angiography (Grade 2B).[5][87][88]
-
-
Rationale: In candidates for revascularization, high quality arterial imaging helps plan and guide the intervention. Choice of imaging modality varies according to the availability of equipment and local expertise, and according to history and physical examination. For patients who are suitable candidates for limb salvage, complete diagnostic angiography that includes the ankle and foot should be performed before revascularization is attempted or denied.[5]
Table 4. Comparison of different imaging modalities for patients with chronic limb-threatening ischemia (CLTI) [5][7]
Vascular imaging modality
|
Indications and Advantages
|
Limitations |
|
Duplex ultrasound imaging (DUS)

|
Indications
- For patients with suspected arterial disease, DUS is the preferred initial imaging modality, if available. DUS can help inform decisions regarding revascularization
Advantages
- Noninvasive
- Low cost
- Quick, widely available worldwide
- Useful to monitor efficacy of therapeutic intervention
|
- Time-consuming
- Highly operator dependent
- Calcification may produce incomplete examination.
- Poor at estimating collateral blood supply and reserve
|
|
Magnetic resonance angiography (MRA)
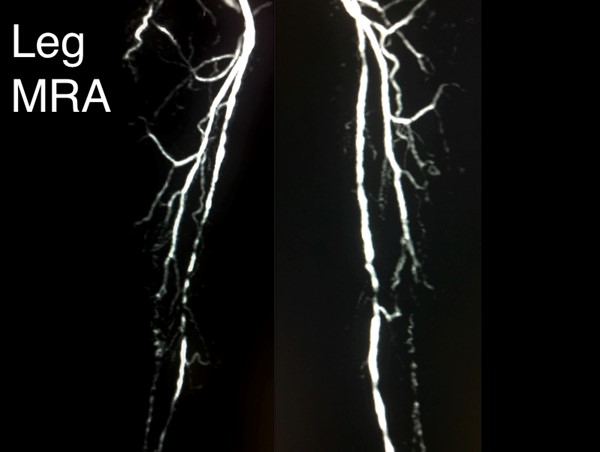
|
Indications
- MRA is indicated for patients with arterial disease above the knee
- For patients with arterial disease below the knee, a foot CE-MRA may be indicated. Available at specialized centers, a CE-MRA has sensitivity high enough (92%) to detect significant disease in infra-popliteal and pedal vessels.
Advantages
- Noninvasive
- Eliminates exposure to ionizing radiation
- Unaffected by arterial calcification
- Easily produce arterial maps that greatly aid in surgical planning
|
- For patients with arterial disease below the knee (which generally includes patients with foot/ankle AU), a simple MRA may fail to completely image the ankle and foot vessels with sufficient resolution for procedural planning. In those cases, a foot CE-MRA or a catheter digital subtraction angiography (DSA) should be obtained
- Patients with pacemakers and defibrillators and some cerebral clips cannot be scanned safely
|
|
Computed Tomography Angiography (CTA)
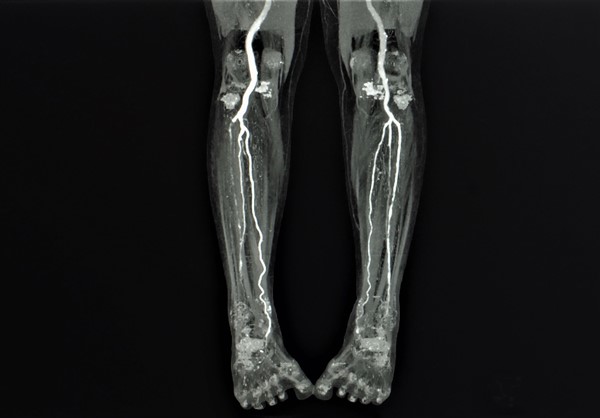
|
Indications
- For arterial disease involving the femoropopliteal or aorto-iliac segments, CTA has high sensitivity and specificity (compared to DSA), and thus may be used to image and plan interventions [88]
- For patients with contraindications to MRA
|
- For arterial disease below the knee (which generally includes patients with foot/ankle AU), sensitivity and specificity are not as high as those of a DSA. [88]
- Calcified arteries may interfere with imaging
- Potentially nephrotoxic contrast agents
- Radiation exposure
|
|
Catheter digital subtraction angiography (DSA)
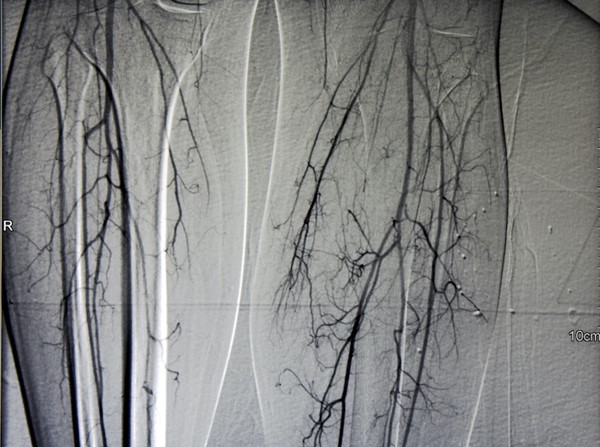
|
Indications
- Indicated for patients with arterial disease below the knee, when MRA and/or foot CE-MRA are not available or fail to provide adequate visualization of tibial and foot vessels
- If CTA or MRA are not viable options
Advantages
- If available, DSA may be performed immediately after a DUS, with the option for endovascular intervention in the same setting
|
- Exposure to ionizing radiation and contrast media
- Complications of catheterization
|
Differential Diagnosis
AU differential diagnoses include [89]:
- Mixed ulcers (i.e. ulcer with mixed etiology, for instance, arterial/venous, arterial/neuropathic, etc)
- Diabetic foot ulcers (see topic "Diabetic Foot Ulcer - Introduction and Assessment")
- Venous ulcers (see topic " Venous ulcers - Introduction and Assessment")
- Pressure ulcers (see topic " Pressure Ulcers/Injuries - Introduction and Assessment")
- Trauma
- Acute burns, frostbite (see topic " Acute Burns - Introduction and Assessment")
- Calciphylaxis
- Scleroderma
- Vasculitis (see topic "Cutaneous Vasculitis")
- Spider bite
- Hypertensive ulcers
- Pyoderma gangrenosum (see topic "Pyoderma Gangrenosum - Introduction and Assessment")
- Neoplasms (e.g. melanoma)
- Skin manifestations of COVID-19 (e.g. chilblains-like lesions, acute limb ischemia due to hypercoagulability)
Classification
-
1CFor all patients with AU and CLTI, clinical guidelines recommend using the Society for Vascular Surgery Lower Extremity Threatened Limb Wound/Ischemia/Foot Infection (WIfI) classification system (Grade 1C). [4][5][6] See WIfI classification in 'Algorithm 1.' or in topic "Diabetic Foot Ulcers - Classification Systems"
-
-
Rationale: The WIfI system correlates with limb salvage, amputation risk, wound healing and can identify patients who are likely to benefit from revascularization. Limb staging should be repeated after vascular intervention, foot surgery, or treatment of infection and whenever there is suspected clinical deterioration.[5]
- Other classification systems for PAD and ischemia were developed in the past (See Table 5). However, those classification systems were primarily developed for patients suffering from pure ischemia and atherosclerosis, prior to the global epidemic of diabetes mellitus. Because patients with diabetes mellitus make up the majority of patients with CLTI, absolute perfusion now needs to be considered in the context of neuropathy, wound characteristics, and infection. This need is contemplated by the WIfI system. [5]
- Of note, the WIfI classification is not to be used for patients with vasospastic and collagen vascular disease, vasculitis, Buerger’s disease, acute limb ischemia, or acute trauma (mangled extremity).
Table 5. Ischemia and PAD classifications (The Rutherford and Fontaine classification systems), adapted from Tummala S. et al. [2]
Rutherford Classification Systems
|
Fontaine Classification Systems
|
|
Description: assigns severity based on performance on the 5-minute treadmill test at 2 mph on a 12% incline.
|
Description: assigns severity based entirely on symptoms.
|
|
Indication: used primarily and routinely in patient care.
|
Indication: used primarily for clinical research and not routinely used in patient care.
|
|
Grade
|
Category
|
History
|
Stage
|
Clinical findings
|
|
0
|
0
|
Asymptomatic
|
I
|
Asymptomatic
|
|
I
|
1
|
Mild claudication
|
IIa
|
Mild claudication
|
|
I
|
2
|
Moderate claudication
|
IIb
|
Moderate to severe claudication
|
|
I
|
3
|
Severe claudication
|
III
|
Ischemic rest pain
|
|
II
|
4
|
Ischemic rest pain
|
IV
|
Ulceration or gangrene
|
|
III
|
5
|
Minor tissue loss
|
|
|
|
III
|
6
|
Major tissue loss
|
|
|
Ulcer healability
Categorization of wound healability (i.e., healable, maintenance, or non-healable) is of particular importance.[8] This designation defines for the clinician, patient, and family an expected course of action, plan of care, and healing rate. A framework is summarized below. For details, see topic “How to Determine Healability of a Chronic Wound".
As a prerequisite to setting realistic treatment objectives, wounds may be classified as [8]:
- Healable wound: the underlying cause of the ulcer can be corrected, there is enough blood supply to heal the wound, existing co-factors, conditions or medications that could potentially delay healing can be optimized or ideally corrected.
- Maintenance wound: the wound may be healable, but the cause cannot be corrected due to patient unwillingness to adhere to treatment or a lack of required system resources
- Non-healable wound: the patient is severely ill or may have negative protein balance or inadequate blood supply that is not bypassable or dilatable
For patients with AUs, if any of the conditions below are present, consider a non-healing program:
-
Non-correctable arterial insufficiency (as evidenced by ABI, Doppler, TCOM, other exams). That is, revascularization is not possible or not indicated. (e.g. patients with DFU and PAD for whom, from the patient perspective, the risk-benefit ratio for the probability of surgical success is unfavorable, or unclear. See topic “Diabetic Foot Ulcer Associated with Ischemia - Management”.
- WIfI Stage 5: unsalvageable foot (most commonly due to wound extent, severity of infection)
- Co-morbidities that impede healing:
-
- Ulcer is malignant tumor,
- Major organ failure
If patient has any of the conditions that impede wound healing below, consider a maintenance-healing program until element impeding healing is mitigated:
- Comorbidities:
-
- Uncontrolled diabetes
- Immunosuppression
- Obesity: BMI > 40
- Inadequate nutrition (abnormal serum protein, unintended weight loss)
- Drugs and interventions:
-
- Steroids, immunosuppressants
- Chemotherapy/ radiation
- Regular smoking, illicit drugs, alcohol abuse
- Financial or resource constraints
- Non compliance with treatment
Documentation
Documentation of noninvasive arterial testing
To support the diagnosis of AU, signs and symptoms must be attributable to objectively proven arterial insufficiency by noninvasive arterial tests (e.g. ABI/TP, TBI/TP, TcPO2, Doppler waveform, skin perfusion pressure, etc). Waveform documentation is required for reimbursement of ABI tests.
Documenting signs of improvement to support medical necessity (Medicare):
- Documenting signs of improvement to support medical necessity (Medicare):
-
- Reimbursement for wound care services on a continuing basis for a particular wound in a patient requires documentation in the patient's record that the wound is improving in response to the wound care being provided.
- It is not medically reasonable or necessary to continue a given type of wound care if evidence of wound improvement cannot be shown.
- Medicare expects the wound-care treatment plan to be modified in the event that appropriate healing is not achieved. Such evidence must be documented with each date of service provided.
- Evidence of improvement includes measurable changes (decreases) of some of the following:
-
- Drainage
- Inflammation
- Swelling
- Pain
- Wound dimensions (diameter, depth)
- Tissue type: necrotic tissue/slough
Documentation tools
- Tools that facilitate standardized assessment and documentation should be used whenever possible:
-
- Validated vascular assessment tools:
-
-
Rose Questionnaire: Determines pain experience of patients with cardiac-ischemic disease. Standardizes identification of angina to exercise, pain due to myocardial infarction and intermittent claudication.[7][90] Includes 8 questions:
-
- 1. Do you get pain in either leg on walking?
- 2. Does pain begin when standing still or sitting?
- 3. Do you get this pain in your calf/calves?
- 4. Do you get it if you walk uphill or hurry?
- 5. Do you get it when you walk at an ordinary pace on the level?
- 6. Does it ever disappear while walking?
- 7. What do you do if you get it while walking?
- 8. What happens to the pain if you stand still?
-
Edinburgh Claudication Questionnaire (ECQ): Updated Rose Questionnaire with improved sensitivity while maintaining high specificity.[7][91] Includes 6 elements:
-
- 1. Pain or discomfort in the legs with walking
- 2. Pain on sitting or standing
- 3. Pain going up stairs or hurrying
- 4. Regular walking pain
- 5. Pain duration of 10 minutes or more
- 6. Site of pain
- Validated wound assessment tools:
-
- Bates-Jensen Wound Assessment Tool [92]
- Wound Reference Wound Prep & Dress Tool creates notes to help support medical necessity that can be copied and pasted to electronic medical records
- Wound imaging: digital photographs at the first consultation and periodically thereafter to document progress is helpful and ensures consistency of care among healthcare practitioners, facilitates telemedicine in remote areas, and illustrates improvement to the patient.
-
Wound measurement: methods include manually measuring length and width (the longest length with the greatest width at right angles), manual tracing, digital photography, and software programs that calculate wound dimensions from a photograph of the lesion. Wound tracings that calculate the area via digital software are slightly better than linear measurement. [93]
-
Patient reported outcome (PRO) Tools: Validated instruments allow clinicians to obtain feedback on whether a given treatment plan is resulting in the best outcomes possible and is improving patient's quality of life (QOL). PRO assessment will continue to gain importance as health care systems transition from a volume-based reimbursement model to a value-based model.[94]
-
-
QOL tools validated for AU patients: 36-item Short Form (SF-36), Walking Impairment Questionnaire, EQ-5D, and the Vascular Quality of Life Questionnaire.[42]
-
-
Medicare Quality Payment Program, Improvement Activity "Promote Use of Patient-Reported Outcome Tools" suggests use of Wound-Quality of Life (QoL) and patient-reported Wound Outcome.[95]
-
Validated pain measurement tools: for neonates up to 6 months - CRIES (Crying; Requires O2 for Saturation > 95%; Increasing vital signs; Expression; Sleepless)[96]; for children 2 months to 7 years of age - FLACC (Face, Leg, Activity, Cry, and Consolability)[96], and the Wong-Baker FACES scale; for adults - visual analog scale (VAS).
-
- Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"
Referrals to other specialists
When to refer to specialists [9][3][7] :
- Vascular specialist: if arterial disease is suspected as indicated by history, physical exam, vascular assessment and noninvasive arterial tests. Specific indications include:
-
- Absence of both pedal and posterior tibial pulses.
- ABI:
-
- An ABI less than 0.90 plus any one of the following: wounds failing to improve within 2 to 4 weeks of appropriate therapy, severe ischemic pain, and/or intermittent claudication.
- ABI less than 0.50.
- ABI greater than 1.3 or noncompressible vessels and unable to obliterate pulse signal at cuff pressures greater than 250 mm Hg.
- Toe pressure less than 30 mm Hg (< 50 mm Hg if diabetes).
- Ankle pressure less than 50 mm Hg.
- Urgent referral if:
-
- Acute limb ischemia, manifested by six ‘Ps’ (onset < 2 weeks): pain, pallor, pulselessness, poikilothermia (cold), paresthesias, and paralysis.
- CLTI (onset >2 weeks): ischemic rest pain, non-healing wound/ulcers, or gangrene attributable to objectively proven arterial occlusive disease.
- ABI less than 0.40
- Gangrene
-
Surgeon with experience in foot surgery (e.g. podiatrist, orthopedic surgeon): tissue loss, infection with palpable pulses, urgent surgical intervention for deep abscesses, compartment syndromes, and all necrotizing soft tissue infections.[97]
- Internal medicine/ family medicine specialist: assessment and optimization of systemic comorbidities such as smoking status, lipid lowering, blood glucose and hypertension
- Infectious disease specialist: if infection of AU is suspected
- Plastic surgeon: flaps and grafts after limb amputation, significant debridement of revascularized tissue
- Physical therapist, occupational therapist: exercises to improve symptoms of intermittent claudication, reconditioning program post amputation
- Psychologist, social worker: psychological, financial, social adjustment
- Nutritionist at initial evaluation, pain management specialist, orthotist for offloading or post-amputation prosthetics, podiatrist for foot deformities, hyperbaric oxygen therapy specialist for Wagner 3 and above. Respective specialists if associated metabolic, hematologic, autoimmune, oncologic diseases are suspected.
- Hospice if the patient is certified as terminally ill (prognosis of 6 months or less) by their attending provider (if they have one) and the hospice provider.[98]
CODING
ICD-10
- First code the underlying condition (see codes in the Appendix) , then code the ulcer with a chronic ulcer ICD-10 code (L97.-) as follows:
- Specify laterality
-
- Right, left or unspecified
- Specify ulcer severity (L97.-):
-
- Limited to breakdown of skin
- With fat layer exposed
- With necrosis of muscle
- With necrosis of bone
- Unspecified severity
Diagnostic Related Group
ICD-10-CM I70.25 is grouped within Diagnostic Related Group(s) (MS-DRG v37.0):
- 299 Peripheral vascular disorders with mcc
- 300 Peripheral vascular disorders with cc
- 301 Peripheral vascular disorders without cc/mcc
APPENDIX
Summary of Evidence (SOE)
Noninvasive Arterial Tests for Determination of Ulcer Healing Potential
(back to text)
Relevant clinical guidelines are summarized in the table below
| Intervention | Joint guidelines [5]
| AHA [73] | WHS [55] | WOCN [57] |
| PAD |
|
|
|
|
In patients with history or physical examination findings suggestive of PAD (includes nonhealing lower extremity wound), the resting ABI, with or without segmental pressures and waveforms, is recommended to establish the diagnosis.
|
| IB |
|
|
For patients with history or physical examination findings suggestive of PAD (includes nonhealing lower extremity wound), audio handheld Doppler waveforms are diagnostic of PAD and triphasic pulse is reliable to rule out significant PAD.
|
|
| yes |
|
For all patients with diabetes, measure TBI as the first-line noninvasive test to detect PAD
|
|
| yes |
|
| For patients with ABI > 1.4, TBI should be measured to diagnose patients with suspected PAD |
| IB |
|
|
| CLTI (CLTI is an advanced manifestation of PAD) |
|
|
|
|
For all patients with suspected CLTI, measure AP and ABI as the first-line noninvasive test
| 1B | |
|
|
For all patients with suspected CLTI and tissue loss, measure TP and TBI
| 1B |
|
|
|
In patients with normal (1.00–1.40) or borderline (0.91–0.99) ABI in the setting of nonhealing wounds or gangrene, it is reasonable to diagnose CLTI by using TBI with waveforms, transcutaneous oxygen pressure (TcPO2), or skin perfusion pressure (SPP)
|
| IIaB
|
|
|
When ankle and toe pressures, indices, and waveforms cannot be assessed, consider using alternative methods for noninvasive assessment of perfusion, such as PVR, transcutaneous oximetry, or skin perfusion pressure,
| 2C |
|
|
|
Obtain annual ABI exams for patients with a prior history of DFUs or lower extremity arterial disease and other known risk factors.
|
|
|
| yes |
Assess the ABI every 3 months for patients with nonhealing DFUs; ABI can decrease over time.
|
|
|
| yes |
Doppler arterial waveform and audible handheld doppler
(back to text)
- In 2022, Kashetsky et al [60] published a review study evaluating the diagnostic accuracy of the ankle-brachial pressure index (ABPI) compared with that of Doppler arterial waveforms (DAWs) to detect peripheral arterial disease (PAD). Results are summarized below:
- Results suggest that DAW has higher sensitivity for detecting PAD compared with ABPI, and ABPI has higher specificity for detecting PAD compared to DAW. That is, results demonstrated a higher DAW sensitivity (fewer false negative results) compared with ABPI in all patients, including persons with diabetes. Thus, DAW has better potential value as a screening tool for PAD compared with ABPI because it is less likely to miss individuals with the disease.[60] Despite the review limitations, the authors recommend DAW over ABPI as a screening test for PAD because of its higher sensitivity. In addition, ABPI is recommended as a diagnostic test for PAD because of its higher specificity compared with DAW [60],
- Sensitivity:
- In patients with PAD and peripheral diabetic neuropathy, all studies reported higher sensitivities (fewer false negative results) of DAW compared with ABPI
- Patients with diabetes without neuropathy had a sensitivity of 100% for both DAW and ABPI.
- In patients without diabetes, the sensitivity of DAW was 86% whereas the sensitivity of ABPI was 83%.
- Specificity:
- In patients with diabetic neuropathy: The higher specificity of PAD detection using ABPI in patients without diabetes was 95% versus 66%
- in patients with diabetes without neuropathy: William et al5 found that DAW had slightly higher specificity (92%) compared with ABPI (88%) in detecting PAD.
- Persons without diabetes had higher specificity for the ABPI detection of PAD in all studies compared with DAW
Toe pressure and toe brachial index
(back to text)
- In 2016 Tehan and Chuter [76] published a study comparing a targeted screening method and the American Heart Association (AHA) guideline for vascular screening to screen for PAD. One hundred nineteen participants were included. Sensitivity of the targeted screening method (62%, 95% CI 47.17–75.35) was higher than the AHA method (49%, 95% CI 34.75–63.40), however, specificity of the AHA method (94%, 95% CI 85.62–98.37) was higher than the targeted screening method (85%, 95% CI 74.26–92.60). Diagnostic accuracy was similar with the AHA method yielding 74% diagnostic accuracy and the targeted screening method 73%. Conclusion:The targeted screening method and the broad international guideline demonstrated similar accuracy, however clinicians may save time using the targeted screening method.
ICD-10 Coding
Codes in red indicate that the code is not billable as there are several codes below it that contain a greater level of detail and thus are billable (codes in black font)
- I70.23 Atherosclerosis of native arteries of right leg with ulceration
-
- I70.231 …… of thigh
- I70.232 …… of calf
- I70.233 …… of ankle
- I70.234 …… of heel and midfoot
- I70.235 …… of other part of foot
- I70.238 …… of other part of lower leg
- I70.239 …… of unspecified site
- I70.24 Atherosclerosis of native arteries of left leg with ulceration
-
- I70.241 …… of thigh
- I70.242 …… of calf
- I70.243 …… of ankle
- I70.244 …… of heel and midfoot
- I70.245 …… of other part of foot
- I70.248 …… of other part of lower leg
- I70.249 …… of unspecified site
- I70.25 Atherosclerosis of native arteries of other extremities with ulceration
- I70.26 Atherosclerosis of native arteries of extremities with gangrene
-
- I70.261 …… right leg
- I70.262 …… left leg
- I70.263 …… bilateral legs
- I70.268 …… other extremity
- I70.269 …… unspecified extremity