Treatment Protocol Guidelines
The following hyperbaric medicine treatment protocol is based upon the recommendations of the Hyperbaric Oxygen Committee of the Undersea and Hyperbaric Medical Society. Clinical protocols and/or practice guidelines are systematically developed statements that help physicians, other practitioners, case managers and clients make decisions about appropriate health care for specific clinical circumstances.
Protocols allow health providers to offer evidence-based, appropriate, standardized diagnostic treatment and care services to patients undergoing hyperbaric oxygen therapy (HBOT). This section discusses osteoradionecrosis of the mandible (ORN). Evidence-based medicine offers clinicians a way to achieve improved quality, improved patient satisfaction, and reduced costs. Utilization Review should be initiated when clinical decisions result in deviation from, or modification of, treatment protocols. This includes any course of treatment at or above the recognized threshold limits.
Medical Necessity
Medicare.gov defines “medically necessary” as “health-care services or supplies needed to prevent, diagnose, or treat an illness, injury, condition, disease, or its symptoms and that meet accepted standards of medicine.”
The following condition meets coverage indications per the National Coverage Determination (NCD) 20.29.[1] Continued HBOT therapy without documented evidence of effectiveness does not meet the Medicare definition of medically necessary treatment. Thorough re-evaluation should be made at least every 30 days for documentation of response to therapy.
|
HBO request requirements checklist | Sample Physician Order | $ ICD-10 Crosswalk | Treatment Table |
| Background | Delayed effects of radiation are a complication of modern radiotherapy that can be ameliorated with hyperbaric oxygen therapy. Some examples of delayed radiation effects include soft tissue radionecrosis, osteoradionecrosis (ORN), radiation cystitis, radiation proctitis, and laryngeal chondroradionecrosis. The basic pathophysiology of delayed radiation tissue damage is an obliterative endarteritis with resultant tissue hypoxia and secondary fibrosis. ORN can be characterized according to the Marx classification.[2] Robert Marx, DDS, developed a staging system and treatment protocols for osteoradionecrosis.[2] HBOT is an effective adjunctive therapy for existing ORN Marx Stage I-III (Figures 1 and 2).[1] For successful mandibular reconstruction, Dr. Marx lists 6 goals that must be met. Those include [3]:
- Restoration of jaw continuity
- Restoration of alveolar height
- Restoration of alveolar width suitable for dental implants
- Restoration of arch form
- Maintenance of bone over time
- Restoration of facial contours
|
|
Goals of HBOT
|
- Reduce tissue fibrosis and breakdown by inducing neovascularization and improving tissue oxygenation
- Prevent wound dehiscence following soft tissue reconstruction in previously irradiated areas
|
|
Diagnosis
|
- Osteoradionecrosis is the result of avascular, aseptic necrosis in a previously irradiated field. Irradiated bone becomes exposed through a wound in the overlying skin or mucosa. [4]
- After radiation to the mandible, salivary glands are non-functional. Saliva is imperative for tooth health. Mandibular teeth (with a single blood supply) tend to suffer most and begin to breakdown. The underlying mandible may also breakdown and become exposed.
- Clinical findings include pain on mastication, fibrotic skin, orocutaneous fistula, exposed mandible, trismus, infection, pathologic fracture, and/or bad breath
- Radiographic findings include moth-eaten appearance of the mandible, pathological fractures
- The teeth or mandible involved must be within the radiation ports
|
|
Hyperbaric Criteria
|
- Referral documenting confirmed diagnosis of osteoradionecrosis (This generally comes from an oral surgeon.)
- History and documentation of radiation treatment to the region of the pathology including laterality.
- Radiation treatment terminating at least 6 months prior to onset of signs, symptoms, or planned surgical intervention at the site.
|
|
Evaluation
|
- Comprehensive history, to include:
- Date cancer diagnosed
- Date radionecrosis diagnosed
- Type, amount, number of treatments and location of the radiation
- Radiation date span
- Previous treatment or therapies
- Obtain the radiation-oncology reports
- Physical examination
- Labs to order or review:
- CBC
- Serum Albumin
- Pre-albumin
- Sedimentation Rate
- C-reactive protein
- Wound photographs
- Chest x-ray: order or review
- ECG: order or review
- Determine present oncological status
- Evaluation of tympanic membranes pre- and post-treatment as needed
- Baseline visual acuity assessment
- Smoking/nicotine cessation
- Nutritional screening. These patients generally lose weight after radiation and may be continuing to lose weight with the oral lesions. Be sure to monitor overall weight loss, and include a statement that says weight is stable, increasing, or decreasing.
- Coordination of hyperbaric therapy and surgical procedures, in conjunction with referring oral-maxillofacial surgeon.
- Classify mandibular necrosis with the Marx scale to describe the severity of the ORN. [2][5]
- Stage I ORN: patients with exposed bone which has been chronically present or which developed rapidly.
- Stage II ORN: patients who do not respond favorably to 30 pre-operative HBOT treatments, or when a more major operative debridement is required.
- Stage III ORN: Presence of orocutaneous fistulae, evidence of lytic involvement extending to the inferior mandibular border, full thickness bone damage or pathologic fracture. Usually requires complete resection and reconstruction with free tissue transfer. There are 2 sources of free flaps for this repair. One in a rib flap (with artery and vein) and the second is a fibular free flap (with artery and vein) If mandibular resection is anticipated, patients are advanced to Stage III from outset.
- Rule out cancer recurrence or residual tumor when possible.
|
|
Treatment
|
Stage I: - Consists of HBOT sessions followed by minor bony debridement, then additional HBO sessions
- HBOT:
- 2.0-2.5 ATA for 90 minutes of oxygen breathing with two air breaks (may be 5 or 10 minutes in length) at the 30 and 60-minute portions of oxygen breathing (Table 3)
- HBOT once daily for 30 treatments
- Surgery:
- HBOT (to complete the Marx treatment protocol)
- If adequate response, continue with 10 additional daily HBOT and follow patient to complete clinical resolution
- If little or no improvement after 30 treatments, the patient may be advanced to stage II in consultation with the oral/maxillofacial surgeon.
Stage II: - Consists of HBOT (30 treatments) followed by more radical surgical debridement, then additional HBOT (10 postoperative treatments)
- Surgery:
- Stage II patients receive a more aggressive surgical debridement, immediately followed by 10 postoperative daily HBOT.
- Surgery for stage II patients must maintain mandibular continuity.
- If mandibular segmental resection is required, patients are advanced to stage III
Stage III: - Consists of 30 preoperative HBOT, followed by transoral mandibular resection then additional 10 postoperative HBOT
- Patients who originally classified as stage I or II who fail the initial therapy above, or patients who present initially with serious signs of pathologic fracture, orocutaneous fistulae, or evidence of lytic involvement extending to the inferior mandibular border, are treated as stage III.
- Surgery:
- Mandibular segmental resection and eradication of all necrotic bone are planned as part of the treatment. The usual surgery involves a free bone flap with attached soft tissue, and/or extensive titanium plate and screw reconstruction (Figure 3). If present, orocutaneous fistula or large tissue loss are also surgically addressed in this phase.
- After surgical resection and HBOT post-resection, the patient is advanced to stage III-R (reconstructive protocol)
Stage III-R - Consists of mandibular reconstruction and additional HBOT
- After a period of several weeks following completion of stage III, definite mandibular bony reconstruction with techniques such as free flaps or myocutaneous flaps is conducted
- Discuss with the oral/maxillofacial surgeon whether 10 postoperative treatments should follow in order to support initial free tissue flap.
For all patients
- Wound photography post-reconstruction
- Evaluation of tympanic membranes pre- HBOT and as needed
- Smoking Cessation
|
|
Follow-Up
|
- Visual acuity assessment for progressive myopia on a weekly basis (See topic "Visual Acuity and HBOT")
- Wound assessment and photography as indicated
|
|
Treatment Threshold
|
30 – 60 treatments (Peer review should be requested after 60 treatments) |
Coding
| Refer to the ICD-10 Guideline for the appropriate ICD-10 code
|
Comments
| - If the patient has diabetes, blood glucose should be checked within one 1 hour prior to treatment and post-HBOT by unit personnel.
- Avoid petroleum based dressings and ointments whenever possible. If these are a necessary part of the surgical dressing, ensure that they are not exposed and are completely covered with 100% cotton.
- Of note, Dr. Marx recommends that when patients are treated with HBOT for radiation injuries at 2.0 ATA, the HBOT exposure be increased from 90 to 120 minutes.
- The Marx rabbit mandible study and the authors’ experience suggest that 2.4 ATA is the preferred pressure for treating radiation injuries. However, treatment at 2.0 ATA has also been successfully employed.[6][7]
- A study out of the Hennepin Hyperbaric Medicine facility, which was presented at the 2019 Annual Scientific Meeting of the UHMS and printed in abstract form, compared treatments for radiation cystitis at either 2.0 or 2.4 ATA for 90 minutes of 100% oxygen. Both groups had eventually comparable results, but the group treated at the University of Pennsylvania at 2.0 ATA on average required five treatments more than the group treated at 2.4 ATA at Hennepin to obtain a comparable clinical result.[8]
|
Primary Sources: Whelan and Kindwall [3]
, Weaver [9][10],
Huang [11]
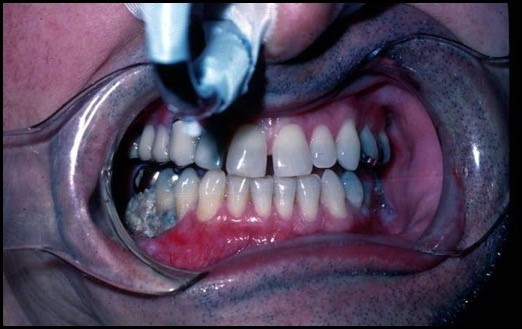
Fig. 1. Mandibular osteoradionecrosis. Marx Stage I with exposed mandible. By Worth E.R., M.D.

Fig. 2. Mandibular osteoradionecrosis. Marx Stage III with orocutaneous fistula.
By Worth E.R., M.D. Fig. 3. Mandibular reconstruction. By Worth E.R., M.D.
Fig. 3. Mandibular reconstruction. By Worth E.R., M.D.DOCUMENTATION
Radiation doses in excess of 40 Gray (4,000 cGy) are more commonly associated with compromised/delayed healing and persistent soft tissue/ integumentary breakdown within a previously irradiated field that has failed conservative and/or surgical management. The role of hyperbaric oxygen is one of overcoming hypovascular - hypocellular - hypoxic tissue bed and the late-radiation effects. Acute inflammatory responses to ongoing or recently completed radiation therapy need to be differentiated. Relieve the radiotherapy-induced hypoxic/ischemic state by stimulating angiogenesis, thereby overcoming radiation-induced obliterative endarteritis. Below we explain in detail what needs to be documented and provide sample statements that can be adapted to suit your needs. See also:
-
HBOT request requirements checklist
History
Sample history for osteoradionecrosis is shown below:
"Mr. Smith had a diagnosis of squamous cell cancer of the left side of the tongue. His radical neck surgery was in May, 2017. This was followed by 3 rounds of chemotherapy and 40 treatments of external beam electron therapy. The dosage ports were reviewed with the oral surgeon. He had an overall dose of 6,500 cGy with a boost at the base of the tumor to a total dose of 7,800 cGy. He comes to us now (10 months after radiation has finished) with a large area of visible mandible. We are asked to provide HBOT support for surgical resection and repair."
Documentation should include:
- An initial assessment including a history and physical that clearly substantiates the condition for which HBOT is recommended.
- Documentation of total radiation dose
- Documentation of any prior medical, surgical and/or hyperbaric treatments.
- Past medical records must support history of radiation therapy at the site identified by anatomical location.
- Documentation of optimization of nutritional status
Physical Exam
- Evidence of radiation therapy at the site identified by anatomical location (e.g., "Mrs. Smith has a permanent sunburn within the radiation tattoo marks over her left lateral mandible. The skin is taught and there are scattered telangiectasis noted within this area. There is no noted hair growth. The skin appears thin and friable.") You have just described the hallmark physical signs of radiated tissue.
- Measure and describe the non-responding wound within the previously radiated field.
- Clearly state what the intended clinical course will be.
- Note any prior failure of definitive coverage attempts, flap or graft.
Impression
- Osteoradionecrosis of the Mandible See ICD-10 Crosswalk
Plan
"For preventing wound healing complications in heavily irradiated tissue areas, in the face of established osteoradionecrosis of the mandible, we treat patients with 30 hyperbaric oxygen treatments at 2.0 or 2.4 ATA, breathing oxygen for 90 to 120 minutes. The patient then undergoes surgical repair followed by 10 or more treatments postoperatively, until the wound shows signs of continued healing."
Risk and Benefit of Hyperbaric Oxygen Treatment
Please refer to topic "Documentation HBO: Risks and Benefits"
Indication for Hyperbaric Oxygen Therapy (HBOT)
"Delayed effects of radiation are a complication of modern radiotherapy that can be well treated with hyperbaric oxygen therapy. Some examples of delayed radiation effects include soft tissue radionecrosis, osteoradionecrosis, radiation cystitis, radiation proctitis, and laryngeal chondroradionecrosis. The basic pathophysiology of delayed radiation tissue damage is endarteritis with resultant tissue hypoxia and secondary fibrosis.
Delayed radiation complications are often manifest as non-healing wounds located in previous irradiated areas and are precipitated by an additional insult such as surgery or trauma within the field of radiation. Hyperbaric oxygen has been shown to induce neovascularization and increase cellularity in irradiated and other hypoxic tissues. The success with randomized controlled studies in patients with mandibular osteoradionecrosis is clearly noted. Dental extractions or other surgical procedures are fraught with high complication rates when performed in heavily irradiated tissues without the benefit of preoperative hyperbaric oxygen therapy."
Sample Order
-
See Sample Physician Order
Documentation by Certified Hyperbaric Nurse and Certified Hyperbaric Technician
The certified hyperbaric technician (CHT) and certified hyperbaric nurse (CHRN) should maintain an accurate record of the care and related support services delivered during each patient’s course of hyperbaric oxygen therapy. The patient's chart is a legal document and as such, must reflect in meaningful terms the patient's condition, progress, and care rendered. The provider and nurse's notes must be concise, accurate, and support medical necessity for the treatment ordered. The notes must reflect responsibility for the care rendered. For details, see topic "Documentation: Hyperbaric Treatment Notes by the CHRN and CHT"
Daily treatment should include, but is not limited to the following:
- Physician Order - confirm daily HBO treatment order.
- Patient Assessment (by the CHRN or CHT)
- Pre-treatment Safety Check (by the CHRN or CHT)
- Documentation of the Treatment Log (by the CHRN or CHT)
- Physician Supervision (by the CHRN or CHT)
- Care Provided (by the CHRN or CHT)
- Plan of Care (by the CHRN or CHT, based on physician orders)
CLINICAL EVIDENCE AND RECOMMENDATIONS
-
1BFor patients with ORN Stage I, II and III, we recommend HBOT as an adjunctive therapy (Grade 1B)
-
Stage I: HBOT followed by minor debridement, then additional HBOT;
-
Stage II: HBOT followed by more radical surgical debridement, then additional HBO;
-
Stage III: HBOT followed by mandibular resection, additional HBOT, then mandibular reconstruction and additional HBOT
-
-
Rationale: A small percentage of patients treated with radiation for head and neck cancers will develop osteoradionecrosis (ORN) of the jaw. Patients treated with more than 6000 centigray (cGy) of radiation have an approximately 9% incidence of developing mandibular ORN.[2] Radiation doses in excess of 40 Gray (4,000 cGy) are more commonly associated with compromised/delayed healing and persistent soft tissue/ integumentary breakdown within a previously irradiated field that has failed conservative and/or surgical management.
- The Marx scale classifies mandibular necrosis and is used to describe the severity of the ORN.[2] Stage I ORN: patients with exposed bone. Stage II ORN: patients who do not respond favorably to 30 pre-operative HBOT treatments and a minor debridement (usually in the dentist/surgeon office), or when a more major operative debridement is required. Stage III ORN: presence of orocutaneous fistulae, evidence of lytic involvement extending to the inferior mandibular border, full thickness bone damage or pathologic fracture. Usually requires complete resection and reconstruction with free flap transfer.[5] If mandibular resection is anticipated, patients are advanced to Stage III from outset.
- Use of HBOT for management as an adjunctive therapy to treat ORN is supported by moderate certainty evidence (evidence level B).[12][13][14][15] The role of hyperbaric oxygen is one of overcoming hypovascular - hypocellular - hypoxic tissue bed and the late-radiation effects. Acute inflammatory responses to ongoing or recently completed radiation therapy need to be differentiated.
- A frequently expressed concern by those considering hyperbaric oxygen for a patient with radiation injury is the fear that hyperbaric oxygen will somehow accelerate malignant growth or cause a dormant malignancy to be reactivated. An overwhelming majority of both clinical reports and animal studies reviewed showed no enhancement of cancer growth.[13]
- Coverage: Medicare covers perioperative use of HBOT as an adjunctive therapy for existing ORN Marx Stage I-III. Data to justify HBOT prophylaxis for osteoradionecrosis in a previously irradiated mandible undergoing tooth extraction is lacking at this time; subsequently this is a non-covered service. See section on 'Coverage and Reimbursement' below.
OPERATIONAL CONSIDERATIONS
Providing HBOT in a safe manner is the primary objective with each and every treatment. The clinical team (physician, CHT, CHRN, etc) providing HBOT goes to great lengths to ensure patient safety with every treatment. A summary of operational considerations pertaining to adjunct HBOT for osteoradionecrosis of the mandible is provided below:
Chamber Inspections
- Routine chamber inspections should be conducted to confirm chamber maintenance procedures and the safe operation of all equipment utilized during HBOT.
- To ensure the safety of the environment in the hyperbaric medicine facility, chamber inspections are to be performed routinely (i.e. daily, monthly, semi-annually, as needed). Processes and systems that meet standards set forth by The Joint Commission (TJC) and the Undersea and Hyperbaric Medical Society (UHMS) should be utilized, and elements that have been developed within the field (in some cases through “near misses”) may be incorporated. These processes and systems can be implemented through customized patient-centered checklists. Checklists have a wide range of applications, with the potential to improve patient education, pre-procedure planning, discharge instructions, care coordination, chronic care management, and plans for staying well.
- For resources on chamber inspections, see topic "HBO Safety Inspections".
Ground testing
- NFPA 99 requires that all hyperbaric chambers are grounded and patients inside chambers filled with 100% oxygen are likewise grounded. Wrist continuity tests prior to each treatment and daily chamber checks including chamber stud to wall measurements and patient ground jack to chamber stud measurements ensure ongoing continuity.
Prohibited Item(s), Assessment and Authorization
- Wound dressings, devices, and other objects that go in the hyperbaric chamber with the patient may raise important safety concerns, including the production of heat, production of static electricity, production of flammable vapor, ignition temperature, and total fuel load. It is critical that clinicians understand which dressings, devices, and objects are prohibited, restricted or allowed inside a hyperbaric chamber during HBOT for osteoradionecrosis of the mandible.
- Frequently, questions arise in regard to which items are prohibited, restricted or allowed inside a hyperbaric chamber during HBOT. The NFPA 99 2018 edition, chapter 14 "Hyperbaric Facilities", provides the process for effectively managing patient care product(s) during HBOT. The NFPA 99 2018 edition - 14.3.1.6.4.4 states “Physician and Safety Director approval to use prohibited items shall be stated in writing for all prohibited materials employed”.[16]
- Each hyperbaric facility should maintain an internal list of items that are approved for use, should be used with caution and should not be used in the chamber. An authorization form is required for items that should be used with caution. To facilitate assessment, clinicians might opt to utilize the Go-No-Go Risk Assessment Tool. The tool is an interactive process that enables hyperbaric technicians, Safety and Medical Directors to document the product information necessary to complete the risk assessment process. Upon completion of the process, the user will have the ability to print or email the document. In addition, clinicians can review lists of items that are approved for use, should be used with caution and should not be used in the chamber. See " Go-No-Go Lists / Prohibited Items" and "Go-No-Go: Frequently Asked Questions".
Ancillary Equipment
- All equipment utilized by the hyperbaric medicine facility is maintained through a program of regular preventative maintenance. The manufacturer maintains the hyperbaric chambers during the regularly scheduled service contract. Ancillary equipment (e.g. cardiac monitor leads, TCOM sensors) should be serviced by the Hospital's Biomedical Department and maintained in accordance with the recommendations of the manufacturer. See topics "Ear Exam - Barotrauma" and "Ancillary Equipment".
Air Breaks
- During HBOT for osteoradionecrosis of the mandible, it is necessary to provide an alternative air breathing source. This may also be necessary to reduce the risk of central nervous system oxygen toxicity. The air breathing system consists of an independent high-pressure air source, capable of providing flow that is sufficient to meet the patient's inspiratory demand. Air breathing systems may be provided by institutional gas outlet (wall outlet) or via portable "H" cylinders utilizing a diameter index safety system (DISS) regulator. Delivery of the air break to the patient may be provided by disposable non-rebreather mask, demand valve and resuscitation mask or trach collar. For purposes of infection control, masks should be single patient use and cleaned or replaced (per patient) as needed.
- While the use of air breaks to decrease the incidence of CNS oxygen toxicity has not been directly demonstrated, there is a large amount of published data on the cause of oxygen toxicity related directly to a combination of the level of FiO2, and time. As such, these air breaks limit the interval time exposure and are expected to decrease the risk of oxygen toxicity. [17]
Infection Control - Cleaning/ Disinfection of the Hyperbaric Chamber
- The cleaning and disinfection of acrylic monoplace chambers is done with caution because many commercial biohazard-cleaning agents contain alcohol. While alcohol is adequate to kill many pathogens, it is destructive to acrylic and can produce flammable fumes and vapors creating a fire hazard in the chamber and the immediate area. Therefore, only manufacturer-approved cleaning products are used for disinfection of the hyperbaric chamber and accompanying equipment. Particular cleaning and disinfecting tasks are divided among staff from environmental services, equipment or biomedical device services, respiratory services, nursing, and other departments of the health care team.
- Adverse outcomes related the risk of infection stands at the forefront of concern for patients with osteoradionecrosis of the mandible. Particular attention should be given to cleaning and disinfection of the hyperbaric chamber acrylic, stretcher and associated equipment, including observing the specific kill times of the solution used prior to the delivery HBOT. See topic: "Cleaning and Disinfection of Hyperbaric Oxygen Monoplace Chamber"
Barotrauma
- Assessment of the patient and their past medical history is necessary to identify and minimize the risk of barotraumas. If the patient has history of sinus, tooth, and ear problems, compress slowly and observe patient for signs of pain.
- Middle-ear barotrauma: The most common type of barotrauma experienced by patients receiving HBOT is middle-ear barotrauma (MEB). Normally, the middle ear is a closed, vascular lined space and therefore subject to pressure change. The normal means by which the middle ear remains at an equal pressure with the surrounding tissue is through the opening of the Eustachian tube. During descent, the increasing pressure of the surrounding water leads to “middle-ear squeeze.” If a patient is suffering from Eustachian tube dysfunction, he or she likely will be unable to equalize and will subsequently develop middle-ear barotrauma. [3]
- Pneumothorax: A complete and accurate pulmonary assessment is essential to avoid or prevent gas expansion problems. Assessing the patient with a significant pulmonary history is a key starting point. Identify any history of asthma, chronic obstructive pulmonary dysfunction (COPD), fibrosis, spontaneous pneumothorax, or chest trauma.
- The absolute contraindication for HBOT is the unvented pneumothorax.
- Signs and symptoms of pneumothorax include: Sudden shortness of breath, Sudden stabbing chest pain, Tracheal shift to affected side in cases of tension, Asymmetrical chest movement (lack or reduced chest excursion on affected side), Increase in respiratory distress with decompression, with relief on recompression. [3]
Blood Glucose Level and HBOT
- It is necessary to ensure that all patients who are currently being treated for irregular blood glucose levels do not experience a hypoglycemic event as a result of ongoing HBOT. Most studies suggest that blood glucose decreases in patients with diabetes who undergo HBOT. This decrease in blood glucose is estimated at 50 mg/dl. HBOT might not be as effective in patients with non-insulin dependent diabetes mellitus patients, compared to patients without diabetes. [3]
- If diabetes mellitus is present, blood glucose should be checked within 1 hour prior to treatment and immediately post-HBOT. The goal is to ensure that all patients who are currently being treated, do not experience a hypoglycemic event as a result of ongoing HBOT. Hypoglycemia in the hyperbaric environment may present similar symptoms as oxygen toxicity, including but not limited to sweating, pale skin, shakiness, anxiety, tingling or numbness of the tongue or cheek. See section 'Blood Glucose Level and HBOT' in topic "Patient Care".
Medications in the hyperbaric environment
- The hyperbaric environment creates numerous considerations for the use of drug therapies within it. Physiologic changes to the body due to HBOT may lead to pharmacokinetic changes in drug disposition. In addition, HBO acting as a drug can interact and enhance or lessen the physiologic effect of the drug. Most drugs will not interact unfavorably with oxygen. Unless specific contraindications or precautions have been addressed, it is generally safe to assume a medication can be used. [3] Pharmacodynamic interactions result in modification of the pharmacologic effect of the drug after administration. These interactions will increase or decrease the effects of oxygen or the drug. [3]
- Patients receiving antibiotics with elastomeric infusion pumps (also known as "space balls") may continue to receive antibiotics in the chamber, however infusion amounts during changes of atmospheric pressure should still be monitored. If the pump is full of air, then it may not provide standardized infusion rate. A study shows that the antibiotic flow rate for ceftazidime 6 g is not significantly affected by increases in ambient pressure across the pressure range of 101.3 kPa to 284 kPa. However, there was evidence that the specific antibiotic solution might affect flow rates and this requires further study.
- For potential interactions of drugs administered to patients with osteoradionecrosis of the mandible, see topic "Medications In The Hyperbaric Environment".
Oxygen Toxicity
- The hyperbaric staff should be skilled in reducing the potential for and management of oxygen toxicity for the patient receiving HBOT.
- Central Nervous System (CNS) oxygen toxicity can occur in patients breathing oxygen at pressures of 2.0 ATA (atmosphere absolute) or greater. Convulsions may occur abruptly or may be preceded by other signs of central nervous system irritability.[3] Early estimates of the seizure rate during therapeutic oxygen exposures at 2-3 ATA reported a convulsion incidence of about one per 10,000 therapies or 0.01%.[3]
- The hyperbaric physician will be notified immediately if a patient experiences signs or symptoms of oxygen toxicity during HBOT.
CNS toxicity
Observe patient closely for premonitory signs and symptoms of CNS Oxygen Toxicity such as:
- V : vision-visual changes, blurred vision, visual hallucinations
- E : Ears- Auditory hallucinations, ringing in the ears
- N : Nausea- May include emesis
- T : Twitching- Restlessness, numbness, focal twitching (note time, duration and site)
- I : irritability- Change in personality
- D : Dizziness- Vertigo
- C : Convulsions- Seizure activity
- C : Change in mentation - Change in affect or of a non-descriptive complaint like, “I just don’t feel right” [18]
Pulmonary Oxygen Toxicity
- Most current applications of HBOT do not cause pulmonary symptoms or clinically significant pulmonary functional deficits.[19] Prolonged exposure to oxygen pressures greater than 0.5 ATA is associated with the development of intratracheal and bronchial irritation. Pulmonary oxygen toxicity is not expected from routine daily HBOT. The possibility of development does exist with prolonged exposure most typically related to long treatment tables such as US Navy Treatment Table 6 used for decompression illness, but even these cases would be mild and self-limiting.[20]
- Continued oxygen exposure may lead to impaired pulmonary function and eventually acute respiratory distress syndrome (ARDS). Symptoms include:
- Substernal burning,
- Chest tightness
- Cough
- Dyspnea
- These changes are seen over the course of days to weeks at lower oxygen pressures and occur more rapidly as the oxygen pressure is increased. [3]
Ocular Oxygen Toxicity
- Vision changes as a side effect of HBOT have been observed in patients undergoing prolonged periods of daily HBOT. The rate of these changes has been reported in the literature to be ∼0.25 diopter per week and progressive throughout the course of ongoing treatment. Myopia has been reported in 25–100% of patients undergoing HBOT after several weeks at pressures of 2.0 ATA and greater. [20] When providing HBO for the patient being treated for osteoradionecrosis of the mandible it is important to discuss the risks, hazards and potential side effects with the patient and family. Documentation of this discussion and patient/ family's understanding should be clearly stated in the patient record. For further information See topic:"Hyperbaric Oxygen Therapy and Visual Acuity"
NURSING INTERVENTIONS
Nursing interventions that are relevant for adjunct HBOT of osteoradionecrosis of the mandible are presented below. [21] For further information see topic "Nursing Interventions".
Knowledge deficit related to hyperbaric oxygen therapy and treatment procedures
- Assess and document the patient and/or family's understanding of purpose and goals of hyperbaric oxygen therapy (HBO), procedures involved, and potential hazards of HBO.
- Utilize the teach-back method to confirm patient understanding and identify and address barriers to learning. Involve an interpreter if indicated, apply age-specific teaching, consider cultural/religious factors, assess readiness to learn, and identify patient's expectations of treatment.
- See section 'Knowledge deficit related to hyperbaric oxygen therapy and treatment procedures' in topic "Nursing Intervention"
Anxiety related to hyperbaric oxygen treatments or other medical problems
- Assess the patient for a history of confinement anxiety and implement preventative measures as appropriate. It is important to reinforce to patient that someone will always be with them, and the staff are well trained for emergency procedures. Identify signs of symptoms of anxiety before and during HBO treatment such as:
- Patient states anxiety
- Tense-appearing facial/body posturing
- Complaint of nausea or diarrhea
- Feelings of being confined or smothered
- Defensive or argumentative attitude
- Hyperventilation
- Diaphoresis and hyperventilation
- Tachycardia
- Restlessness
- Sudden feeling of being hot
- See section 'Anxiety related to hyperbaric oxygen treatments or other medical problems' in topic "Nursing Intervention"
Potential for injury within the hyperbaric facility related to transferring the patient in and out of the chamber
- When transferring a patient that is being treated for osteoradionecrosis of the mandible, comply with the facility's fall risk prevention policy. Prior to transfer of the patient assess fall risk and safety precautions. Communicate and involve patient with the plan of action and provide patient education regarding safety precautions. Provide assistance with transfers as patient needs apply to ensure compliance with institutional/facility policy.
Potential for injury related to fire within the hyperbaric chamber
- Fuel sources in an oxygen-enriched environment are an unavoidable circumstance of HBOT and include linens, equipment, dressings, and the patient. The fire triangle consists of oxygen, fuel, and an ignition source (heat). In HBOT an ignition source is needed to complete the fire triangle. This may occur due to a spark in the chamber. Follow facility fire prevention steps and NFPA chapter 14 probes for Class A and Class B hyperbaric chambers. HBOT teaching and consent should include the risks of fire in the chamber. Provide the patient and family with written instructions regarding the risk of prohibited materials during HBOT. Prior to each hyperbaric treatment, staff should perform and document the pre-treatment safety checklist. Ensure this has been performed and time-stamped prior to descent. Patients receiving treatment for osteoradionecrosis of the mandible may have surgical dressings that are ordered to remain intact. A risk assessment per the Safety Director in collaboration with the Medical Director to determine if they may enter the chamber on a case by case basis. Safety measures should be initiated if the risk assessment allows for item to enter the chamber as well as completion of a prohibited item's authorization form signed by the Safety and Medical Directors.
Potential for injury related to changes in atmospheric pressure within the hyperbaric chamber - Assess patient's and inside attendant's knowledge of ear clearing techniques and ability to equalize pressure. Collaborate with provider to assess tympanic membrane (TM) for suspected barotrauma prior to and after the first HBO treatment and per patient complaint. Collaborate with the provider to describe and document observations including color and visibility of TM, presence of wax, blood/fluid/air and any hearing deficits or changes. Methods to equalize pressure in the middle ear during HBO treatment include: yawning, swallowing, jaw thrust, head tilt, Valsalva, Toynbee, Roydhouse, Frenzel, etc. Reinforce the importance of notifying the chamber operator immediately when pressure or fullness is felt in the middle ear.
Potential for unstable blood glucose level related to hyperbaric oxygen therapy and disease pathology
- Literature notes hyperbaric oxygen therapy carries its own mechanism for increased glucose usage through oxygen-mediated transport of glucose into muscle cells and may also increase insulin sensitivity. Prevention of acute hypoglycemia in the hyperbaric chamber is vital for patient safety. Follow facility policy and procedure guidelines for pre and post-treatment glucose control. Prior to treatment assess the patient's knowledge level, recent hypoglycemic events, and patient-specific symptoms of hypoglycemia. Proper glucose control < 200mg/dL is vital for wound healing. Consider timing of short and long-acting glycemic control medications when scheduling HBO to avoid peak action time while at depth in the chamber.
CODING
ICD-10 Coding
COVERAGE AND REIMBURSEMENT
- Medicare covered condition: Medicare covers perioperative use of HBOT as an adjunctive therapy for existing ORN Marx Stage I-III. Prerequisite for treatment includes history of radiation treatment to the region of the documented injury, terminating at least 6 months prior to onset of signs or symptoms or planned surgical intervention at the site. The recommended daily treatments are designed around the stages of radionecrosis and typically last 90-120 minutes at 2.0 to 2.5 ATA. The duration of HBOT therapy for these patients is highly individualized but is not expected to exceed 4-8 weeks therapy. The Marx mandibular osteoradionecrosis protocol extends from 30-60 treatments based on stage I-III, adhering to the established principle that all necrotic bone must be debrided. No demonstrable evidence of improvement post two 30 day periods of HBOT (2.0-2.5 ATA, for 90 to 120 minutes, 5 days per week) suggests lack of benefit and subsequent treatments will be denied as not medically reasonable and necessary. [22]
- Medicare non-covered conditions: In a 2019 randomized control trial, HBOT was used to determine the incidence of delayed osteoradionecrosis in an irradiated mandible after tooth extraction and/or implant placement. At this time, there is no data to support HBOT prophylaxis for osteoradionecrosis in a previously irradiated mandible undergoing tooth extraction.[23] Prophylaxis is a non-covered service. HBOT is not covered to prepare the patient for dental extraction when radiation therapy has been done less than 6 months prior to extraction/implant. [22] We know of wound care/ HBOT clinics who view the need for teeth extraction (without visible mandible) as a form of soft tissue radiation necrosis where there is evidence of a highly radiated field that will need a surgical repair. Since there are no open wounds, the HBOT regimen would follow a Marx protocol with 20 preoperative treatments, then following surgery for 10 postoperative treatments.
APPENDIX
Summary of Evidence
Use of HBOT as an adjunctive therapy for existing ORN Marx Stage I-III.
Systematic reviews
- A 2016 Cochrane systematic review [12] included 14 trials (753 participants). A meta-analysis of three RCTs showed that there was some moderate quality evidence that HBOT was more likely to achieve mucosal coverage with osteoradionecrosis (ORN) (risk ratio (RR) 1.3; 95% confidence interval (CI) 1.1 to 1.6, P value = 0.003, 246 participants, 3 studies). There was also moderate quality evidence of a significantly improved chance of wound breakdown without HBOT following operative treatment for ORN (RR 4.2; 95% CI 1.1 to 16.8, P value = 0.04, 264 participants, 2 studies). As for bony continuity, 1 RCT (104 participants) showed that the experimental group (HBOT) had statistically significant higher chance of establishment of bony continuity compared to the control group (P value = 0.002). 1 RCT (74 participants) contributed results to healing of tooth sockets following extraction in irradiated field at six months [12]. There was an increased chance of successful healing with HBOT compared to the control group (P value = 0.02)
- A 2016 systematic review [24] included 7 studies (RCTs and observational studies) that analyzed HBOT in ORN management. Authors concluded that HNO‐DF/BWCC does not recommend the routine use of HBOT for the prevention or management of ORN. Adjunctive HBOT may be considered for use on a case‐by‐case basis in patients considered to be at exceptionally high risk who have failed conservative therapy and subsequent surgical resection. The highest level of evidence available to date on the management of ORN using HBOT comes from the Annane et al. 2004 multicenter trial. The trial was terminated early due to worse outcomes in the HBOT arm (19% resolution with HBOT versus 32% resolution with placebo). This study did not include the same RCTs evaluated by Bennett et al [12] and thus generated different conclusions. The Annane study was flawed by design. This study demanded that the ORN lesions would close without any surgical procedure, by HBOT alone, in order to be counted as successful. Marx, in original publications, recognized that HBOT was adjunctive to surgical procedures to extirpate dead bone and close the resulting defects.
Guidelines:
- The 2014 UHMS HBO Therapy Indications Book (Guidelines) [13] and the 2017 European Committee for Hyperbaric Medicine Guidelines [14] support the use of HBOT as an adjunctive therapy to treat ORN, and although the guidelines use different evidence grading methodologies, they are in agreement regarding strength of recommendation. See table below:
|
HBOT for Mandibular Osteoradionecrosis (ORN)
|
UHMS, 2014 |
ECHMG, 2017 |
|
HBOT followed by debridement and additional HBO for Stage I Mandibular ORN
|
AHA Class Ib |
Grade 1B |
|
HBOT followed by a more radical surgical debridement for Stage II Mandibular ORN
|
AHA Class Ib |
Grade 1B |
| Mandibular resection followed by HBOT, then mandibular reconstruction and HBOT in the treatment of Stage III Mandibular ORN |
AHA Class Ib |
Grade 1B |
Observational
- A 2023 retrospective study by Dang et al studied prophylactic HBOT to prevent ORN (n=121 patients). Authors found that ORN occurred in 9.1% of individuals and delayed healing for 3.3%; fifteen individuals (12.4%) were unable to complete the HBOT protocol. The individuals who were diagnosed with ORN had a significant association with age (P = 0.006) and binary analysis showed alcohol consumption to be a significant predictor. Authors concluded that prophylactic HBOT protocol had a lower proportion of individuals diagnosed with ORN and those who were diagnosed were more likely to be younger males and have current alcohol consumption.[15]
- A 2018 retrospective study by Jenwitheesuk et. al. [25] aimed to determine the benefits of adjunctive treatment of HBOT in ORN (n=84 patients). Authors found that HBO improved wound healing of ORN patients with stages I-III.
- A 2017 observational study by Dieleman et al [26] evaluated the success of HBOT and surgery in the treatment of mandibular osteoradionecrosis (ORN) in relation to the extent of the ORN. Twenty-seven patients with ORN with a history of primary oral or base of the tongue cancer who had been treated with radiation therapy with curative intent between 1992 and 2006, with a radiation dose to the mandible of ≥50Gy. The ORN was staged according to the classification of Notani et al. The time from completion of radiation therapy to the development of ORN varied (median 3 years). Forty HBOT sessions were offered. After HBOT alone, 3 of 11 stage I lesions, 0 of 8 stage II lesions, and 0 of 8 stage III lesions had healed (P=0.0018). An absolute incidence of 5.3% ORN was found in this population. Of all sites irradiated in this study, the floor of the mouth was most associated with ORN (8.6%), whereas the cheek was least associated (0%). Based on the results of this study, HBOT can be recommended for stage I and II ORN and for selected cases of stage III ORN (evidence level C)
- A 2016 observational study by Niezgoda et. al. [27] analyzed the effect of HBOT on a cohort of patients (588 participants) with ORN and found that 92% presented with improved scores.
HBOT prophylaxis for osteoradionecrosis
Randomized clinical trials (RCT)
- A 2019 RCT (100 analyzed patients) compared effect of prophylactic HBOT on the incidence of ORN in patients who required dental extractions or implant placement in the mandible with prior radiation therapy >50 Gy. There was no statistically significant difference between both groups.The incidence of ORN at 6 months was 6.4% and 5.7% for the HBOT and control groups, respectively (odds ratio, 1.13; 95% confidence interval, 0.14-8.92; P = 1). Authors concluded that the low incidence of ORN makes recommending HBOT for dental extractions or implant placement in the irradiated mandible unnecessary.[23]
Negative studies
- In 2019 Shaw et al published the results of the HOPON trial (n=144 patients), a phase 3 RCT that studied establish the benefit of HBOT in the prevention of ORN after high-risk surgical procedures to the irradiated mandible. Patients in the hyperbaric arm had fewer acute symptoms but no significant differences in late pain or quality of life. Authors concluded that the low incidence of ORN makes recommending HBO for dental extractions or implant placement in the irradiated mandible unnecessary. Of note, the study was underpowered and included patients with less than 6000 cGy and patients getting dental implants rather than extractions.[23]
- In 2011 Delainian et al studied the effect of a pharmacological treatment for late radiation injuries including mandibular ORN. Authors used a protocol called PENTOCLO which consisted of pentoxifylline, vitamin E and bisphosphonate clodronate, as well as prednisone and ciprofloxacin for 16 months, and reported complete response in all patients ina median time of 9 months. Of note, patients included in the study were possibly overstaged by the researchers prior to treatment (e.g. patients with a Stage 1 of the Marx protocol were possibly staged and included in the study as Stage 3). Complete resolution of exposed bone was not required to be counted as a success, in contrast with what Dr Marx describes as treatment success.[28]
REVISION UPDATES
| Date | Description |
| 9/05/24 | Expanded topic and protocol for Stage I, updated references |
2/24/23
| Operational Considerations and Nursing Interventions sections added
|
| 5/21/19 | Updated section on 'Documentation'. Added figures |
4/22/19
| Updated section on 'Clinical Evidence and Recommendations' with recent studies. Added section 'Coverage and Reimbursement'
|