TREATMENT
Overview
This topic covers management of lymphedema and its complications (e.g., infection, ulcers). For details on surgical management of lymphedema see "Lymphedema - Surgical Management". For clinical guidelines and quality measures specific to lymphedema, see "Lymphedema - Overview". For evidence-based review and assessment of lymphedema including epidemiology, risk factors, etiology, pathophysiology, history, physical examination, diagnosis, differential diagnoses, documentation and ICD-10 coding, see "Lymphedema - Introduction and Assessment". For patient education, see topic "Patient education - Lymphedema"
Background
Coordination of care is an important aspect for management of lymphedema, as patients frequently have co-morbidities usually treated by different healthcare teams. Lymphedema is best managed by a multidisciplinary team including certified lymphedema therapists, medical physicians, and lymphedema surgeons.[3] The team creates a personalized treatment based on a comprehensive assessment that:
- Includes comprehensive history and physical examination
- Identifies underlying causes of lymphedema (i.e., primary or secondary) and assesses risk factors contributing to development of lymphedema and its complications (e.g. ulcers, infection)
- Identifies conditions associated with lymphedema (e.g. if on lower limbs: chronic venous insufficiency, peripheral artery disease, etc)
- Differentiates from other types of edema (systemic or local), which require different treatments
- Identifies pertinent patient's/caregiver's concerns
- Stages lymphedema using a validated classification system, such as the one created by the International Society of Lymphology (ISL) [1]:
- Stage 0: subclinical disease, swelling absent despite underlying impaired lymphatic transport (at risk for lymphedema)
- Stage I: tissue swelling present but reduced by limb elevation. Pitting may occur
- Stage II: limb swelling partially reduced by elevation, pitting is manifest. Later in stage 2, limb may not pit due to excess subcutaneous fat or fibrosis
- Stage III: lymphostatic elephantiasis with skin changes (e.g., acanthosis, thickening, further fibrosis, warty overgrowths) and recurrent infections
- Assesses lymphedema severity: within each stage above, severity can be assessed and classified according to simple volume differences of the affected part, that is, the change from baseline in swelling not due to weight change (i.e, if limbs, determined by measuring both limbs). Severity can be classified as follows [1]:
- Minimal: typically > 5 to < 20% increase in limb volume
- Moderate: 20-40% increase
- Severe: > 40% increase
- Some clinics prefer to use > 5 to < 10% as minimal and > 10 to < 20% as mild
An adequate treatment plan for lymphedema aims to:
- Treat the cause and other factors impeding healing
- Assess patient's and caregiver's concerns
- Manage lymphedema complications effectively
If lymphedema without complications (e.g., ulcers, infection) does not improve within 3 months of adequate treatment, the initial treatment plan should be reassessed, differential diagnoses re-evaluated and adjunctive therapies considered.[4] If infection or ulcers are present, the initial plan should be reassessed within a few days or weeks depending on the severity level.
Of note, in all practice settings, it is essential to ensure that the treatment plan and care provided adheres to the specific facility's policies, procedures, protocols, and formulary.
See Algorithm for Management of Lymphedema (Algorithm 1)
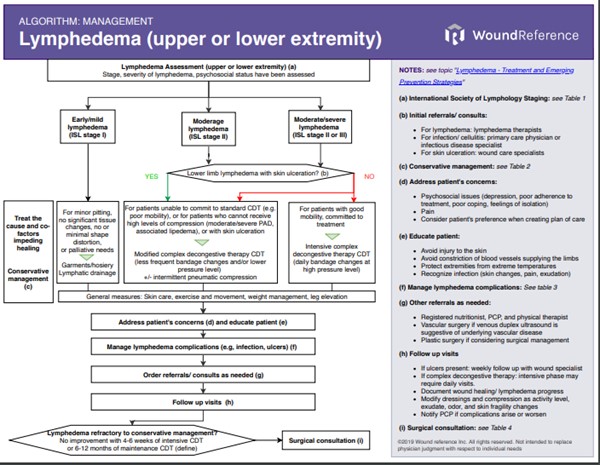
Algorithm 1. Management algorithm for lymphedema (click on this link to see algorithm)
Treatment goals
- In general, the goals are to reset patients to a subclinical or latency stage, regardless of their diagnosed stage or classification[5], and ultimately improve quality of life by achieving better social, functional and psychological adaptation.[6]
- Lymphedema treatment is focused on minimizing swelling, restoring cosmesis and limb function, and preventing complications (e.g. ulcers, infections).[7]
- Physiologically, this is accomplished by restoring balance to the lymphatic system through measures that decrease lymph fluid production and increase lymphatic flow.
Treat the cause and co-factors impeding healing
A graduated approach is recommended according to clinical stage of lymphedema and individual patient presentation[1]:
- For all cases, regardless of severity grades, conservative management is initially recommended.[2]
- For disease that is refractory to conservative treatment or if there are specific indications for surgery, surgical management may be indicated. See section 'Surgical Management'.
Conservative management by lymphedema stage
Mild lymphedema
- 2CFor patients with mild limb lymphedema (typically ISL stage I), minor pitting, no significant tissue changes, no or minimal shape distortion, or palliative needs, expert committees suggest initial management with lymphatic drainage and compression garments/hosiery.[8][9] (Grade 2C)
- Rationale: Lymphatic drainage is safe and may offer additional benefit to compression bandaging for swelling reduction of mild-to-moderate lymphedema, such as breast cancer related lymphedema.[10] Lymphatic drainage for mild limb lymphedema can be taught by lymphedema specialists and self-applied by patients. As for compression garments, pressure level is guided by the patient's vascular status and their ability to tolerate compression and manage the garment. Lymphatic drainage and compression garments should be accompanied by proper skin care, exercise/movement and limb elevation. [8][9]
Moderate or severe lymphedema
- 2CFor patients with moderate or severe limb lymphedema (ISL stage II or III) without contraindications, expert committees suggest complex decongestive therapy with a standard intensive phase to reduce limb volume.[8][9] (Grade 2C) See section on 'Complex Decongestive Therapy' below.
- Rationale: Patient adherence is paramount for successful outcomes, thus patient selection for the standard intensive therapy phase of complex decongestive therapy must be carefully done. Patients must be willing and able to commit physically and emotionally to daily intensive therapy, including participation in exercise programs.[8][9]
- For patients unable to commit to the standard intensive phase of complex decongestive therapy (e.g. due to poor mobility), or for patients who cannot receive high levels of compression (e.g. due to moderate/severe peripheral arterial disease or associated lipedema), or who have skin ulceration, expert committees suggest that the intensive phase of the complex decongestive therapy be modified according to the patient's needs and conditions.[8][9]
- Modified intensive therapy with high pressure: for patients who are able to tolerate high levels of compression, but are unable to commit to standard intensive therapy for physical, social, psychological or economic reasons.[9] Consists in less frequent changes of lymphedema compression bandaging (e.g., 3 times a week as opposed to daily), skin/nail care, exercise, lymphatic drainage.
- Modified intensive therapy with reduced lymphedema compression bandaging pressure: for patients who cannot tolerate or receive high levels of compression (e.g, due to associated moderate peripheral arterial disease [ankle-brachial index between 0.5 and 0.8] or lipedema). Consists in lymphedema compression bandaging changed daily to 3 times a week, skin/nail care, exercise, lymphatic drainage, with or without intermittent sequential pneumatic compression.[9]
- For patients with limb ulcers, lymphedema compression bandaging may need to be modified. Intermittent sequential pneumatic compression is usually helpful as an adjunct to complex decongestive therapy.[9]
Conservative lymphedema management - Interventions
Interventions frequently used in conservative management of lymphedema are summarized below.
physical therapy and adjuvants
Compression Garments/Hosiery
- For initial and subsequent management of mild limb lymphedema, and following lymphedema compression bandaging in cases of moderate or severe limb lymphedema, expert consensus suggests the use of compression garments/hosiery to reduce progression of lymphedema.[8][9][11] For more information on compression garments/hosiery for lower extremities, see topic "Gradient compression stockings".
- Regular use of compression garments has been shown to prevent progression of lymphedema, however, they do not aid in the volume reduction in the acute phase.[12]
- When patients are provided elastic compression garments, care should be taken to ensure they are well fitted, providing the highest tolerated compression.[11]
- The choice of circular versus flat knit elastic compression depends on the severity of lymphedema and limb shape (Figure 1).[11]
- For patients with mild/moderate limb lymphedema, expert consensus suggests the use of circular knit garments, as they provide relatively less containment than flat knit elastic compression (i.e. circular knit garments tend to stretch during the day allowing some degree of expansion and swelling).[11]
- For patients with significant differences in leg circumference as well as deep skin folds and edema of the toes/forefoot, expert consensus suggests use of flat knit garments.[11] Unfortunately, these garments tend to be significantly more expensive which is often a barrier to acquisition.[11]
- Practical considerations regarding choice of compression garments include issues related to patient compliance, cost, and reimbursement.[11]

Fig. 1. Difference between circular and flat knit compression garments for lymphedema
Velcro adjustable circumferential compression wraps
- As an alternative to compression garments, velcro circumferential wraps may be used for limbs with lymphedema stage II and III to prevent progression of lymphedema.[11]
- Velcro circumferential wraps are inelastic devices that have advantages in very large legs, as they can accommodate a variety of leg shapes and are adjustable. However, they require education to ensure proper donning and when and how to adjust the wrap.[11]
- Coverage: as long as criteria are met, Medicare covers lymphedema compression treatment items such as compression garments (both standard and custom-fitted garments for daytime and nighttime use), compression bandaging systems (supplies for the initial and maintenance phases of lymphedema treatment), gradient compression wraps and accessories. See topic " Expanding Care: Medicare's 2024 Coverage Update for Lymphedema Compression Treatment Items".
Complex Decongestive Therapy (CDT)
Complex decongestive therapy (CDT) is also known as Combined Physical Therapy (CPT) or Complex Decongestive Physiotherapy (CDP). It is the standard of care for management of lymphedema, although high certainty evidence endorsing this concept is lacking.[13][14] CDT is regarded as a noninvasive and cost-effective therapy that reduces limb volumes in both upper and lower extremity lymphedema.[15]
- CDT is performed by lymphedema specialists. In the United States, those are usually physical therapists (PT) or occupational therapists (OT). Massage therapists, nurses and physicians can also perform certain kinds of lymphedema interventions if they have been specially trained in lymphedema.[16]
- The beneficial effects of CDT are thought to be secondary to improved collateral lymphatic drainage, increased lymphatic pumping function and reduced fibrogenesis.[17]
- Indications for CDT therapy are: lymphedema, high clinical suspicion of lymphedema or inability to rule out other causes of disease in patients with low clinical suspicion of lymphedema.[6]
- Phases: CDT generally involves 2 stages, intensive CDT and maintenance CDT [1][4][15]:
-
- Intensive CDT: Ideally, this initial intensive phase is administered 1 or 2 times a day, every day for 1 to 6 weeks.[16] Weekly volumetric limb assessments are conducted at the end of each week to determine if volume reduction occurred or if a plateau has been reached. The initial treatment phase combines 4 elements of therapy:
-
- Compression therapy with lymphedema compression bandages. The type of compression device should be selected by a lymphedema specialist and can include inelastic bandages, multicomponent bandage systems (Figures 2-4), garments or adjustable compression wraps.[18][19]
- Pressure: recent studies suggest that pressure in the range of 20–30 mg Hg and 40–50 mm Hg seems to be effective in arm lymphedema and lower limb lymphedema respectively.[18] For patients with combined peripheral arterial disease and lower limb lymphedema, pressure should not exceed 40 mmHg.[18]
- Manual lymphatic drainage (MLD)[11]
- Therapeutic exercise, and
- Skin/nail care
- Maintenance CDT: the second phase is a life-long therapy initiated after the patient’s response to intensive CDT has plateaued. It involves self-care to maintain limb size, compression with low-stretch compression garments, continued exercise, self-skin/nail care, and self-applied simple lymphatic drainage, as needed.[1]
- Coverage: CDT is covered by Medicare if criteria are met (i.e.: documentation supports diagnosis of lymphedema and not other tissue edema, states prior unsuccessful therapies (e.g, elevation), demonstrates clear clinical improvement with therapy, service is provided by a qualified clinician. Education on how to apply compression is covered (up to 3 sessions using CTP 97535), however compression bandaging alone for lymphedema is not covered if the patient does not have an ulcer.[20]
 Fig. 2. Multicomponent bandage system. Application of the foam layer (By Feijoeiro F., PT) Fig. 2. Multicomponent bandage system. Application of the foam layer (By Feijoeiro F., PT)
|  Fig. 3. Multicomponent bandage system. Application of the short stretch compression bandage (By Feijoeiro F., PT) Fig. 3. Multicomponent bandage system. Application of the short stretch compression bandage (By Feijoeiro F., PT)
| 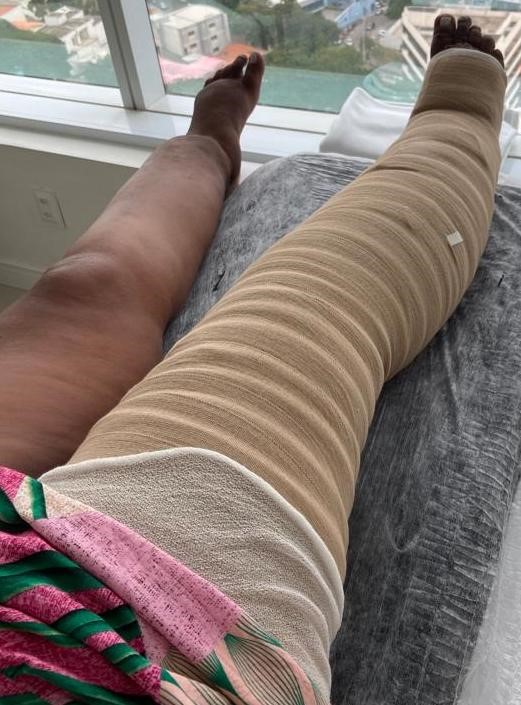 Fig. 4. Multicomponent bandage system (tubular bandage, foam, short stretch) (By Feijoeiro F., PT) Fig. 4. Multicomponent bandage system (tubular bandage, foam, short stretch) (By Feijoeiro F., PT)
|
Intermittent Sequential Pneumatic Compression (IPC)
- Intermittent Sequential Pneumatic Compression (IPC) consists in application of compression therapy with a sequential gradient pump, followed by application of a form-fitting low stretch stocking or sleeve to maintain edema reduction.[1]
- For patients with ulcers, compromised mobility or physical exercise, IPC may be used in the treatment of lymphedema as an adjunct to CDT.[11][21]
- For all other cases, evidence suggests that IPC may not provide additional benefits when used in combination with routine management of lymphedema.[11][21] There is some evidence that IPC with higher pressure may reduce lymphedema effectively, however safety of IPC operating at high pressure remains to be determined.[21]
- Contraindications to IPC include infections on the affected region, metastatic disease to the edematous extremity or the axilla, administration of chemotherapy (other than tamoxifen), ongoing radiation therapy for active cancer in the limb or surrounding area, or the presence of lymphangiosarcoma. Congestive heart failure, renal insufficiency, and hypertension are relative contraindications. [22]
- Coverage: Medicare does not cover pneumatic compression devices (i.e. IPCs) as initial therapy for lymphedema in the home setting. A patient must first undergo a 4-week trial of conservative therapy, which includes the use of an appropriate compression garment, exercise and elevation. This garment does not need to be custom-fabricated; however, it does need to be a graduated compression stocking/sleeve. A pneumatic compression device is covered if a physician determines after such a trial that there has been no significant improvement, or if significant symptoms remain. [23]
Leg elevation
- For all stages of lymphedema, particularly in Stage I, leg elevation often reduces swelling. It decreases intravascular hydrostatic pressures, reducing lymph load and improving lymph drainage. It should be performed at night and during periods of inactivity as to not discourage exercise. If effective, its results can be prolonged during the day by wearing a low-stretch elastic stocking/sleeve.[7][1]
Weight control
- Given the association between increasing body mass index (BMI) and lymphedema progression, patients with lymphedema should be encouraged to maintain a healthy lifestyle with weight control and BMI<=25.[1][24][25][26] No specific diet has proven effective in managing lymphedema, however.[1][2]
Pharmacological therapy
Diuretics
- Diuretics are of limited use in decreasing swelling in patients with lymphedema. Expert committee recommends clinicians reserve use of diuretics in the initial treatment phase of CDT for specific patient populations, namely for patients with [1][8]:
-
- Effusions in body cavities (e.g., ascites, hydrothorax),
- Protein-losing enteropathy
- Peripheral lymphedema from malignant lymphatic blockage, who may benefit from a short course of diuretic drug treatment.
- Intolerance to fluid overload, such as those with congestive heart failure, cirrhosis, nephrotic syndrome and older adults in whom enhanced lymphatic drainage as a result of lymphedema therapy precipitates cardiac failure
- For these patients above, a short course or temporary increase in the dose of diuretics can be administered during the first weeks of compression therapy. Dose can be reassessed 24-48 hours after initiation of compression therapy. Long term use is discouraged due to potential to induce fluid and electrolyte imbalance.[1]
Benzopyrones
- Pharmacological therapy using benzopyrones is not recommended as part of lymphedema management.[27]
Address Patient’s Concerns
Psychosocial issues
Lymphedema has a negative psychosocial impact on affected individuals.[28]
- From the psychological perspective, lymphedema can lead to negative self-identity, emotional disturbance, psychological distress.[28]
- From the social perspective, lymphedema can result in marginalization, financial burden, perceived diminished sexuality, social isolation, perceived social abandonment, public insensitivity, non-supportive work environment).[28]
Psychosocial issues can be initially addressed by generalists, however if problems are not solved within 3 months, referral to specialists is recommended.[8]
Depression
According to severity of depression, generalist interventions include [8]:
- Patient education, support advice on sleep and anxiety management, guided self-help programs
- Problem solving therapy, cognitive behavioral therapy (short-term)
- Counseling (short-term)
- Antidepressant therapy
- Referral to mental health services
Referral to a specialist (mental health services) is warranted if no improvement has happened in 3 months, or sooner if one of the following is observed [8]:
- Active suicidal ideas, psychotic symptoms, severe agitation, self-neglect, poor or incomplete response to two interventions, recurrent episode of depression within 1 year of last episode, patient or relatives request referral
POOR ADHERENCE to treatment
Poor adherence is common among patients with lymphedema. It is important to understand reason for poor adherence and attempt to address reasons behind it.
- If needed and feasible, treatment should be modified to accommodate patient's preference
- Depression should be ruled out
Poor coping, feelings of isolation
- Understanding reasons for these conditions is helpful in order to direct proper care. Care may include patient education, support groups, family/caregiver involvement, financial support, etc.
- If problems are not solved in 3 months, referral to social worker or psychologist is indicated.[8]
Pain
Pain has been reported to affect 50% of patients with lymphedema, with most taking regular analgesia.[8] Pain can be caused by a myriad of conditions including tissue distention, infection, ischemia, lipedema, etc.
- For pain caused by tissue distention due to lymphedema, adequate compression therapy will help alleviate pain.
- For patients who have just initiated compression therapy, pain may be an indicator of excessive pressure, and should trigger reassessment.
- Pain may also indicate infection, which if confirmed, should be treated promptly.
Management of lymphedema complications
The two most common lymphedema-associated skin changes seen in wound clinics are ulcers and skin infections.[29]
Skin infections
Management
- Assessment: Immediate attention should be given to signs of infection:
- Symptoms and signs of cellulitis include pain, swelling, warmth, erythema, lymphangitis and lymphadenitis. In more systemic disease, chills, rigors, fever, and gastrointestinal upset can be seen. Rarely, necrotizing fasciitis can occur caused by the rapid spread of bacteria through underlying fascial tissue, which must be promptly recognized and treated.[4]
- Antibiotic therapy: Prompt initiation of antibiotic therapy is essential in preventing sepsis and decreasing the risk of lymphedema progression.[2] Initial antibiotic choice should cover Staphylococcus and Streptococcus species. Antibiotics should be prescribed for at least 14 days after an acute episode begins responding to treatment.[2] It should be noted that the most common cause of treatment failure is lack of patient adherence.[4]
-
- Antibiotic choice for home care [8]:
-
- First-line: amoxicillin 500 mg every 8 hours, add flucloxacillin 500 mg every 6 hours if infection with S. aureus suspected, clindamycin 300 mg every 6 hours if penicillin allergic
- Second-line: clindamycin 300 mg every 6 hours
- Antibiotics for inpatient settings: Most hospitals will follow their antibiograms for empiric antibiotic therapy coverage. As a suggestion, antibiotic choice for hospital admission includes [8]:
-
- First-line: amoxicillin IV 2 g every 8 hours + gentamicin IV 5 mg/kg daily, clindamycin IV 1.2 g every 6 hours if penicillin allergic
- Second-line: clindamycin IV 1.2 g every 6 hours
- Additional measures to manage cellulitis include limb elevation, non-steroidal anti-inflammatory drugs (NSAID), and increased fluid intake. Manual lymphatic drainage should be avoided during an acute episode of cellulitis, but compression treatment should be maintained as tolerated and long periods without compression should be avoided.[4]
Prevention
Infection recurrence rate among patients who present with a first episode of infection reaches 25%.[2]
- Patient education on daily skin and nail care reduces the risk of cellulitis and unnecessary exposure to antibiotics.[4]
- Antibiotic prophylaxis is indicated to patients with two or more episode of cellulitis per year.[4]
- Antibiotic prophylaxis should be done only after a risk/benefit analysis, with particular attention to antibiotic resistance rates in the community, and after consultation with the patient’s general practitioner and an infectious disease specialist [8].
- Antibiotic selection [8]:
- First-line: phenoxymethyl penicillin 500 mg once daily, erythromycin 250 mg once daily if penicillin-allergic
- Second-line: clindamycin 150 mg once daily or clarithromycin 250 mg once daily
- After one year of therapy, antibiotic dose can be halved. After two years of therapy, antibiotics can be discontinued. However, if cellulitis recurs after antibiotic prophylaxis is discontinued, lifelong antibiotics should be considered.[8]
Ulcers
Ulcers are a common complication in patients with lymphedema. Clinical presentation of ulcers includes [30]:
- Lymphorrhea with superficial eczematous ulcerated lesion
- Established ulcerated lesions
- Ulceration with deep sinus tract in elephantiasis legs
- Ulcers developing after excision procedures
Most ulcers can be managed initially with a non-surgical approach as outlined in this section below. However, an initial surgical approach should be considered when indicated (e.g., surgical debridement if extensive necrotic tissue is present, or surgical wound coverage if tendon, nerve or vessels are exposed). See indications in 'Surgical Wound Coverage' below
determine Wound prognosis (healability)
Upon careful assessment of patient and ulcer, categorization of wound healability is of particular importance.[31] This designation defines for the clinician, patient, and family an expected course of action, plan of care, and healing rate. As a prerequisite to setting realistic treatment objectives, wounds are differentiated as:
- Healable wound: the cause is corrected, there is enough blood supply to heal; moist interactive healing
- Maintenance wound: the wound could heal, but the cause is not corrected due to patient unwillingness to adhere to treatment or a lack of required system resources
- Non-healable wound: underlying cause cannot be treated conservatively, blood supply to the wound is inadequate (e.g., critical limb ischemia), or co-factors impede ulcer healing (e.g., terminal organ failure, ulcer is a malignant tumor). If cause or co-morbidities impeding healing cannot be treated with a conservative approach but can be surgically treated, surgical consult/ referral is recommended. If patient is a poor surgical candidate, ulcer is treated as non-healable.
Treat the cause of the ulcer
- For healable ulcers, the underlying cause of the ulcer and factors impeding healing must be addressed:
- Lymphedema should be adequately managed as it delays ulcer healing. Lymphedema inhibits wound healing via impaired acid-base balance, inhibited cellular transport and decreased immune function in response to infection.[32]
- Other associated conditions and co-factors impeding wound healing must be addressed as well. Factors that impede wound healing include pressure, edema, necrosis, infection, dryness, trauma and systemic factors (e.g. smoking, nutrition, etc).
- For non-healable or maintenance ulcers, underlying causes should be addressed to the extent tolerated by the patient. See section above 'Treat the cause and co-factors impeding healing'
Address Patient's Concerns
For all ulcer prognosis (i.e., healable, non-healable, maintenance), patient's concerns should be addressed properly.
Pain control
- Pain control is an essential component of ulcer care. Two types of pain are associated with open wounds: nociceptive pain from direct tissue damage and neuropathic pain from damage to local sensory nerves .
- A trial of systemic NSAIDs (ie, ibuprofen) is a reasonable first step in the management of ulcer pain. Systemic opioids have significant analgesic effects but are associated with systemic side effects and potential risk of abuse; they are therefore usually reserved for patients with intractable pain. Early studies with topical opioids and dressings containing ibuprofen show promising evidence for their use in ulcer pain control.[33][34] Topical anesthetics such as lidocaine gel can also be applied with dressing changes to minimize discomfort.
Provide effective Local wound care
Cleansing
Promoting a clean wound base minimizes the risk of local infectious complications.
- Non-infected ulcers should be cleansed using a pH-neutral, non-irritating, non-toxic solution during each dressing change without exposing the wound to additional shear or pressure resulting in mechanical trauma. This is best accomplished using mild unscented soaps or soap-free cleaner available over the counter.[35]
- Abrasive soaps and scented soaps should be avoided because they risk changing the natural acidic pH of the skin or drying out the skin excessively. Appropriate care should be administered during skin cleansing to avoid removing the natural sebum secreted to protect the skin from excessive drying.[35]
Debridement
- For superficial wounds (e.g., epidermal loss only), debridement is not usually needed; these wounds often require only lymphorrhea management with absorptive dressings, skin care and compression therapy.
- Rationale: Evidence on use of debridement to improve outcomes of lymphedema ulcers is poor, as seen by the lack of published randomized controlled trials or retrospective studies in this area. However, it makes sense to remove necrotic tissue and debris to control bioburden and facilitate the wound healing process. Furthermore, insurers in the United States will only cover surgical dressings and other adjunctive interventions if debridement is performed, and is part of a comprehensive wound care plan.
- For full-thickness lymphedema ulcers that are not of mixed arterial etiology, we suggest debridement of necrotic tissue and debris.
- Among debridement techniques, clinicians might opt for sharp, mechanical, enzymatic or autolytic debridement. However, surgical debridement is recommended if conservative techniques are ineffective, if soft tissue infection is present, if necrotic tissue is present in large quantity or in areas that are difficult to debride (e.g. sinus tracts).
-
- Rationale: There is little evidence to support the use of one type of debridement over another; type of debridement used is dependent on provider/patient preference and availability. Sharp debridement is faster, however enzymatic debridement is convenient (e.g., with collagenase, papain - the latter is not available in the United States and may cause significant inflammatory response if prolonged use).[35][36] The use of proteolytic enzymes has also been shown to be an effective treatment for skin hyperkeratosis when associated with adequate wound bed preparation with debridement of necrotic non-viable tissue as well as management of chronic inflammatory exudate.[35] After surgical debridement is performed, clinicians might opt for adjunctive Negative Pressure Wound Therapy to promote granulation. Vascularized flap transfer might be indicated for critically exposed structures. See 'Surgical Wound Coverage' below.
Moisture Management and Dressing Selection
Wound dressings are used to reduce exposure to excess exudate, protect local tissue from trauma, reduce infectious complications, and maintain a moist wound bed.[37]
Selection of primary and secondary dressings should follow established wound bed preparation principles. [31][38][39][40] For customized, wound-specific recommendations and customized wound documentation, use Wound Prep and Dress Tool. For practical information on dressing indications, contraindications, application, Medicare coverage and brands see "Dressing Essentials" and "Dressing Brands Quick Reference". General recommendations regarding dressings for lymphedema are listed below.
- There are no high quality studies to recommend one dressing over another in the management of lymphedema-associated ulcers.[37] Since wound dressings should be used alongside compression therapy, dressings should be chosen based on their characteristics under compression and their absorption properties, odor control and cost.[41]
- For exudative wounds, absorptive dressings such as gelling fibers and alginates are suggested. Multi-component bandages can be applied on absorptive dressings to provide simultaneous absorption and compression.[42]
- Silicone dressings have gained popularity in promoting wound healing for lymphedema ulcers in the elderly.[43] Daily dressing changes using silver hydrocellular dressings (e.g., foam dressings with silver) have been also shown to be effective in managing lymphedema-associated non-healing ulcers, given silver’s broad-spectrum antimicrobial properties.[37]
Peri-wound care
The skin around an ulcer is subject to elements that may cause skin changes such as skin maceration, desquamation and contact dermatitis.
- Skin maceration: skin maceration is the softening and breakdown of skin in response to prolonged exposure to moisture. It is a common complication of bandaging during compression therapy or lymphedema-associated wound care, as a result of exposure of skin to exudate or perspiration.[37]
- Management: Skin care in cases of maceration involves the use of a moisture barrier applied on the area surrounding the wound, plus the use of dressings with absorptive qualities.[37]
-
- Skin desquamation: application of topical over the counter skin emollients containing lactic acid, urea, ceramides, glycerin, dimethicone have been used to prevent hyperkeratotic skin desquamation in patients with lymphedema.[41]
-
- Contact dermatitis: a hypersensitivity reaction to an irritant causing red, itchy and scaling skin that may progress to blistering.[4] Exposure to irritants can come from bandages, garments and devices used during compression therapy or from topical ointments using during local skin care.
- Management: acute episodes are managed with topical corticosteroids, with the potency chosen based on the severity and chronicity of dermatitis.[44] Clinical guidelines suggest using a moderate potency steroid such as betamethasone 0.1% daily for mild to moderate cases, and a high potency steroid such as clobetasol 0.05% daily for severe cases. Treatment should be administered for 3-4 weeks with gradual tapering of dosage or frequency.[4]
- For patients who fail to respond to standard therapy, a referral to dermatology for allergy testing should be considered, as patients might be allergic to certain components of lymphedema therapy (ointments, compression bandages/garments).[45]
Wound coverage
Cellular and/or tissue products
Also known as 'skin substitutes', Cellular and/or Tissue Products (CTP) may be adjunctive options for non-healing ulcers on lymphedematous areas, as long as adequate conservative management is maintained.
- Studies utilizing CTP to treat ulcers in patients with lymphedema are scarce. A randomized controlled trial that compared two different kinds of CTP (Apligraf® and TheraSkin®) to treat refractory venous ulcers included patients with associated lymphedema (phlebolymphedema) in both arms and did not find statistically significant difference between both groups.[46]
- Medicare covers application of CTPs as long as coverage criteria are met. See coverage eligibility and documentation checklist for CTPs in section 'Medicare Coverage and Utilization' in topic "Cellular and/or Tissue Products"
Surgical wound coverage
Surgical coverage should be considered if patients have ulcers that fail to reduce in size with conservative measures, if nerve, tendon or vessels are exposed, or for large-area defects that are unlikely to heal without surgery.[30]
- Methods: Surgical wound coverage can be accomplished via the following surgical methods [30][47]:
- Split-thickness skin grafts: initial surgical therapy for patients with ulcers that failed to decrease in size after standard treatment. Accessible surgical option but results in the creation of other wounds at donor sites. Generally performed with additional use of a Negative Pressure Wound Therapy device.
- Free flaps: Consists of the harvesting and autotransplant of flaps containing lymph nodes. More complex procedure than skin grafting and requires hospitalization.
- Pedicled and perforator flaps: Pedicled and perforator flaps result in additional scarring in tissue surrounding the ulcer, and are therefore less preferred than free flaps. Can be designed as a muscle flap as long as the overlying skin tissue is not moved.
- Indications for surgical wound coverage:
- For full-thickness non-healing ulcers, pre-operative wound bed preparation including debridement of necrotic tissue and wound culture to rule out infection is indicated. These ulcers are generally covered via split-thickness skin grafts if nerve, tendon or vessels are not exposed.
- For full-thickness ulcers with exposed nerve, tendon or vessels, coverage with flap is recommended.
- For full-thickness ulcers with severe lipodermatosclerosis, initial coverage with free flaps is preferred. If reconstruction with free flap is not amenable (e.g., in the case of poor donor sites), pedicled or perforator flaps are indicated.
- For ulcers with sinus tracts containing necrotic tissue, excision and debridement of the ulcer and sinus tract, followed by autologous reconstruction with one of the surgical methods described above are recommended.
- Ulcers that arise following surgical excision are characterized by near-continuous lymphorrhea and recurrence even after excision and surgical wound coverage. They are the most difficult to treat and mandate the use of graduate compression therapy and prolonged follow-up to monitor recurrence.
-
Fibrosis
- Management of lymphedema-associated fibrosis is often challenging. Established guidelines are lacking, however reasonable non-surgical interventions to provide symptomatic relief include [48]:
-
- Early initiation of physical therapy to minimize loss of limb function, along with early lymphatic drainage to improve articular contractions.
- Adequate analgesia to minimize pain associated with fibrosis.
- Careful consideration before performing surgical or mechanical interventions that could worsen the underlying fibrosis.
- While a non-surgical approach may be used and preferred in extensive cases of fibrosis, a more definitive approach to management of lymphedema-associated fibrosis includes surgical excision followed by compression therapy.[49]
- Lymphedema-associated fibrosis results in extensive underlying structural changes to interstitial tissues. Therefore, it is unlikely that nonoperative interventions alone are sufficient to manage it. Moreover, fibrotic lymphedema causes irreversible lymphatic vessel injury that precludes surgical reconstruction of the lymphatic vasculature.[49]
- Promising animal studies suggest that various medical therapies may be effective in reducing lymphedema-associated fibrosis. These include hyaluronic acid injections, topical tacrolimus, and topical collagen I gel, all of which have demonstrated reduced fibrogenesis.[50][51][52] However, more research is needed before these interventions can be incorporated in clinical practice.
Lymphangiosarcoma
- Stewart-Treves syndrome is a form of lymphangiosarcoma that is secondary to chronic lymphedema.[53] Survival rates for this malignancy are low, with a median survival of 2.5 years after diagnosis.[54] Some cases of long-term survival are attributed to misdiagnosed cutaneous Kaposi sarcoma which can look indistinguishable from cutaneous lymphangiosarcoma.[53]
- Management of Stewart-Treves syndrome relies on surgery with adjuvant chemotherapy and/or radiotherapy [53]:
- Chemotherapeutic agents include 5-fluorouracil, methotrexate, bleomycin, CHOP combination therapy, or dacarbazine.
- Chemotherapy has been used alone in patients with advanced and inoperable disease.
- Some studies show promising results for the use of immunotherapy in the treatment of Stewart-Treves syndrome with recombinant interleukin-2.[55]
Elephantiasis nostras verrucosa
- Assessment: since superficial debris in cases of elephantiasis nostras verrucosa (ENV) can mask appearance of the underlying skin, a thorough skin assessment should only be performed once the skin has been cleaned.
- It is recommended that cleaning be performed with emollients and tweezers before considering debridement or surgical options.[56]
- Emulsion with 10% urea water-in-oil has been reported to be effective, resulting in keratolysis and thinning of the epidermis.[57]
- Management: debridement of hyperkeratosis, papillomatosis and verrucae is frequently indicated in management of ENV.
- Maggot debridement therapy is an option for patients who do not have bias against larval debridement. Hyperammonemia is a rare but potential side effect of maggot therapy. Symptoms of hyperammonemia include lethargy, irritability, gastrointestinal upset, respiratory depression and seizures.[58]
- Surgical debridement can be considered for cases of ENV with hyperkeratotic verrucous lesions, with simultaneous use of physiotherapy to manage disease progression.[57]
Plan Reassessment
- Periodic functional lymphatic assessments are recommended in order to assess complications and response to treatment.[1] Frequency of assessment depends on the presence of complications. For instance, if an ulcer is present, follow up visits should be done at least weekly.
- Generally, the initial care plan should be reassessed when:
- Management challenges occur (e.g., compression garments not fitting)
- Patient does not adhere to treatment
- Complications occur or do not improve (e.g., worsening wounds, recurrent skin infections)
- Lymphedema without complications fails to respond after three months of adequate treatment.[4] As a benchmark, successful management outcomes include [4]:
-
- Reduction in limb size
- Improved shape
- Improved skin condition and subcutaneous tissue consistency
- Improved limb function
- Improved symptom control
- Plan reassessment should be done in conjunction with lymphedema specialists. Before considering use of adjunctive therapies, a thorough evaluation should be performed to:
- Ensure that the current treatment modality is used correctly
- Other co-morbidities preventing lymphedema improvement are addressed
- Patient adherence is optimized
- Differential diagnoses are considered
Surgical management
For details on surgical management of lymphedema see topic “Lymphedema - Surgical Management". A summary is provided below:
Conservative management of lymphedema with complex decongestive therapy (CDT) remains the first-line treatment for chronic lymphedema. Surgical intervention may be of benefit to a few, well-selected individuals who do not respond to conservative management.[32] Expert committees recommend at least 6 months of CDT before any surgical intervention for lymphedema is undertaken.[1][59]
There are three general surgical approaches to lymphedema: 1) debulking/ ablative/ excisional operations, 2) liposuction, 3) lymphatic reconstructions.[32]
- Debulking/ ablative/ excisional surgery: aims at reducing volume of the limb by excision of the excess cutaneous and fibrous overgrowth. Excisional procedures may be performed alone or with lymphatic reconstructions.[32]
- Liposuction: aims at reducing volume of the limb by removal of excessive adipose tissue.[32]
- Lymphatic reconstruction (also known as physiologic or functional procedures): aims at restoring or improving lymph flow in patients with lymphedema with microsurgical lymphatic reconstruction procedures.[32] With the advent of advanced microsurgical techniques, surgical management of lymphedema has evolved significantly in the past decades. For selected patients, microsurgical functional procedures may now represent the closest chance for a cure of lymph flow disorders.[1]
Care coordination
- Patients under consideration for surgical lymphedema management interventions should be referred to centers with an expert in lymphatic microvascular reconstruction and plastic surgeons with experience treating lymphedema patients. These specialists are more likely found in tertiary centers.
- At these centers, patients are evaluated and carefully selected. For surgical candidates, microsurgical functional lymphatic reconstruction will act as an adjunct to CDT or will be undertaken if CDT is clearly unsuccessful (or has removed the fluid component). [1]
Adjunctive Therapy
Established guidelines on indications for adjunctive therapies are scarce. Adjunctive interventions can be considered when initial treatment modalities have failed. See section on 'Plan Reassessment' above.
Adjunctive physical agent modalities
Physical agent modalities (PAMs) can be used as medical adjuncts to lymphedema therapy. They exert their effects through the use of temperature, light, water, mechanical force, sound or electricity.
- Based on evidence from randomized controlled trials (RCTs) and systematic reviews, the following PAMs are "likely to be effective" in reducing upper limb volumes in patients with breast cancer-related lymphedema:
- Low-level laser therapy [60][61]: use of light waves to stimulate photochemical reactions resulting in anti-inflammatory and analgesic effects.
- Extracorporeal shock wave therapy [62][63]: use of low energy short-wave pulsations to stimulate lymphangiogenesis.
- As for the PAMs below, there is not enough evidence to support their routine use as adjuncts in clinical practice. However, some non-RCT studies suggest these PAMs have beneficial effects:
- Hyperbaric oxygen therapy: administration of pure pressurized oxygen to increase oxygen supply and enhance musculoskeletal function.[64] Treatment of lymphedema with hyperbaric oxygen therapy is not covered by Medicare.
- Electrically stimulated lymphatic drainage: use of electrical waves to stimulate muscle contraction and promote lymphangion function.[65]
- Acupuncture: applied pressure on the skin in the form of needles for the purposes of reducing pain and stress.[66]
-
Adjunctive pharmacological therapy
The following pharmaceutical agents are currently being studied in the management of lymphedema, although none has been established as both safe and effective in large scale human trials [1]:
- Mesotherapy: use of hyaluronidase injections to loosen the extracellular matrix in cases of lymphedema associated with fibrosis.
- Immunological therapy: use of autologous lymphocytes to stimulate the adaptive immune system.
- Molecular therapy: use of vascular endothelial growth factors, VEGF-C and VEGF-D to promote lymphangiogenesis.
- Anti-proliferative therapy: use of octreotide, rapamycin to suppress lymphatic overgrowth in cases of lymphangiodysplasia associated with lymphedema.
-
Adjunctive operative therapy
Implantation of artificial lymphatics in the form of silicone tubings has been attempted in select patients with promising results. This procedure is associated with reduced upper and lower limb circumference without tissue reaction, and might be indicated in cases of advanced lymphedema with large amounts of tissue edema.[67][68]
PREVENTION - EMERGING STRATEGIES
Prevention of lymphedema secondary to lymphadenectomy
For pre-lymphadenectomy patients
Prophylactic Lymphovenous Bypass at the time of lymph node surgery
- 2CFor patients undergoing lymphadenectomy, we suggest consultation with a specialized reconstructive microsurgeon for consideration of prophylactic lymphovenous bypass to prevent development of lymphedema (Grade 2C)
- Rationale: A 2017 meta-analysis (n=82) [69] demonstrated that there is some evidence (level C, low certainty evidence) that prophylactic lymphovenous bypass at the time of lymphadenectomy may prevent development of lymphedema postoperatively. Also known as lymphatic microsurgical preventive healing approach (LYMPHA), a prophylactic lymphovenous bypass consists of microsurgical anastomosis between an accessory branch of the axillary or femoral vein prepared during lymphadenectomy of the axilla or groin, with one or more afferent lymphatic vessels identified by blue dye injection. It may offer a potential life-long avoidance of further treatment, however, the procedure requires specialized resources/skills and its not available at all centers.[70][71] The International Society of Lymphology does not yet routinely recommend the procedure, but recognizes it is currently being explored for high-risk patients.[1]
Other promising INTERVENTIONS
- Axillary reverse mapping (ARM) [72]
- Distinguishes breast-draining lymphatics from arm-draining lymphatics to avoid disruption of the latter during breast cancer surgery.[73]
- Immediate delivery of sustained-release 9-cis retinoic acid to the lymphedenectomy site (experimental stage).[74]
- Experimental studies in mice have shown that 9-cis RA incorporated into an implantable, 30-day sustained-release pellet placed within the surgical site at the same time that hind limb lymphedema was induced improved lymphatic function and structure, reducing lymphedema progression. 9-cis RA contained within a single-use depot DDS seems to have favorable properties in limiting pathologic responses to lymphatic injury and may be an effective strategy against secondary lymphedema.[74]
For post-lymphadenectomy patients
FOR ALL POSt-LYMPHADENECTOMY PATients (upper and lower extremity)
- Patient education is an essential component of lymphedema prevention. Early recognition of transient swelling following cancer-related surgery can help implement a care/prevention plan that attempts to prevent progression to advanced stages [4].
- Compression therapy: For patients with no lymphedema who have undergone lymphadenectomy, it is unclear whether compression stockings can prevent development of lymphedema. An RCT that compared thigh-high 21-32 mmHg stockings with usual care for patients after lymphadenectomy reported no significant differences between groups in terms of incidence of lymphedema or complications.[13][75] However, once lymphedema has developed, compression therapy is recommended.
For patients post-upper extremity lymphadenectomy
- Preoperative, bilateral upper extremity function should be assessed to provide a baseline before treatments. Postoperative assessments should occur regularly up to 1 year after surgery.[76]
- Clinical guidelines recommend minimizing the extent of axillary dissection, preventing infection, and avoiding obesity.[76]
- Early physical therapy has been shown to be effective in preventing lymphedema during the first year after surgery.[77]
-
- Physical therapy should begin the first week after surgery with gentle range of motion exercises. Physical therapy includes manual lymph drainage, scar tissue massage, active and active-assisted shoulder range of motion exercises.[77]
- Stretching exercises should begin one week after surgery or when drains are removed and be continued for 6-8 weeks.
- Strengthening exercises should begin 4-6 weeks after surgery with light weights (1-2 pounds).
- Use of the ipsilateral arm for intravenous lines or blood pressure measurements is not contraindicated in patients with lymphedema.[72] However, when possible, the contralateral arm should be used for venipuncture to minimize the risk for infection or lymphedema progression.[78]
PATIENT EDUCATION
- See topic and handout for patient education in “Patient education - Lymphedema” (coming soon)
APPENDIX
Summary of evidence
SOE - Prophylactic lymphovenous bypass at the time of lymph node surgery to prevent lymphedema
(back to text)
Meta-analyses
- A 2017 meta-analysis included 4 randomized controlled trials (82 patients), and showed that patients treated with prophylactic lymphovenous anastomosis (LVA) had a significant reduction in lymphedema incidence compared to patients receiving no prophylactic treatment (Relative risk: 0.33, 95%CI: 0.19 to 0.56). However, it cautioned that evidence was of low certainty (level C) due to lack of blinding, allocation concealment and sequence generation. Thus, evidence was not strong enough for clinical guidelines to routinely recommend prophylactic LVA. [69]
REVISION UPDATES
| Date | Description |
| 8/16/22 | Expanded section on 'Conservative lymphedema management' |
| 1/09/20 | Added coverage information to section on Intermittent Pneumatic Compression |