ABSTRACT
Lymphedema is defined as progressive swelling (edema) of a specific body part due to insufficiency of the lymphatic system.[1] This progressive swelling eventually results in thickening of the skin and soft tissue underneath. Lymphedema of lower extremities is frequently associated with chronic venous insufficiency (CVI) in a condition known as phlebolymphedema.
Typically, patients with lymphedema receive treatment at different types of care settings: cancer centers, wound clinics, or physical therapy/occupational therapy (PT/OT) clinics specialized in lymphedema. There are significant differences in the profile of lymphedema patients across venues [2]:
- Patients seen at cancer centers have cancer-related lymphedema, and most of them are women with breast cancer-related upper extremity lymphedema.
- Meanwhile, patients seen at wound clinics and PT/OT clinics usually have lymphedema unrelated to cancer; many have lymphedema of lower extremities caused by chronic venous disease and obesity impairing lymphatics.
See Lymphedema Assessment Form Template/Checklist
History: an accurate history and physical exam can diagnose lymphedema in up to 90% of cases.[3] Assessment includes risk factors for development, triggering events, complications, prior treatment, co-morbidities, functional and psychosocial status. See structured framework for history taking.
Physical Examination: a complete physical examination is essential to clinically assess lymphedema. A checklist for local exam of patients with lymphedema is described in this section, including skin changes and complications (e.g., ulcers, cellulitis), mobility and functional assessment, limb measurements, vascular assessment (including recent best practices related to non-invasive arterial testing such as ankle-brachial index and handheld Doppler waveform)
Diagnosis: diagnosis of lymphedema is mainly clinical, based on a comprehensive history and physical exam. However, many diagnostic modalities are used to corroborate evidence and assess severity. For cases with unclear diagnosis of lymphedema, referral to a lymphologist or lymphologic center is recommended for clarification of prognosis and treatment plan. Imaging modalities and genetic testing may be ordered. Lymphoscintigraphy is considered the gold imaging standard to confirm diagnosis. Duplex ultrasonography is the initial test to rule out venous disease. For patients at risk for development of lymphedema (e.g., post-radical mastectomy), surveillance and early diagnosis is recommended.
Differential diagnoses: includes systemic and local causes of edema. See differential diagnoses including lipedema.
Clinical staging: The most widely used by clinicians is by the International Society of Lymphology. The New Taiwan Lymphoscintigraphy classification has been gaining interest recently for incorporating objective measures into its staging assessment. See classification systems
Documentation: documentation to support ICD-10 of lymphedema and associated services/treatments
Treatment: See 'Algorithm for Management of Lymphedema' and topics "Lymphedema - Treatment and Emerging Strategies for Prevention" and "Lymphedema - Surgical Management"
Patient Education: See topic "Patient education - Lymphedema"
When to refer to specialists:
- Lymphedema specialist (e.g, physical therapist, occupational therapist or other healthcare professional specialized in lymphedema) for assessment and conservative management of lymphedema
- Nutritionist for weight management
- Wound specialist if ulcer is present
- Vascular specialist if lymphedema is in lower extremity and peripheral artery disease is suspected.
- Respective specialists if associated metabolic, hematologic, autoimmune, oncologic diseases are suspected.
- Social work for economic and social support.
- Psychologist/ mental health professional for depression and other psychological issues.
- Plastic surgeon: surgical management and prevention of lymphedema
- Hospice if the patient is certified as terminally ill (prognosis of 6 months or less) by their attending provider (if they have one) and the hospice provider.[4]
Coding: See ''ICD-10 Coding'
Clinical Guidelines and Quality Measures: See "Lymphedema - Overview"
INTRODUCTION
Overview
This topic provides a review and framework for assessment of lymphedema and its complications (e.g, ulcers, cellulitis), including epidemiology, risk factors, etiology, pathophysiology, history, physical examination, diagnosis, differential diagnoses, documentation and ICD-10 coding. For clinical guidelines and quality measures, see "Lymphedema - Overview". For management of lymphedema see "Lymphedema - Treatment and Emerging Strategies for Prevention" and "Lymphedema - Surgical Management"
Background
Definition
- Lymphedema is defined as progressive swelling (edema) of a specific body part due to insufficiency of the lymphatic system.[1] It has been regarded by an expert consensus as "a clinicopathologic condition related to intrinsic or extrinsic impairment of lymphatic resorption and/or transportation".[5] The body part affected depends on the anatomic location of the disrupted lymphatics, and can involve the limbs, head and neck, genitals or abdomen. This progressive swelling eventually results in thickening of the skin and soft tissue underneath, which gives chronic lymphedema its characteristic appearance.
- Lymphedema is characterized by high-protein edema, with protein concentrations of 1.0-5.5 g/mL which drive the accumulation of water in the interstitial space. This is in contrast to other forms of edema which have protein concentrations below 1.0 g/mL.[1]
- Lymphedema of lower extremities is frequently associated with chronic venous insufficiency (CVI) in a condition known as phlebolymphedema. As such, when assessing venous ulcers clinicians should maintain a high level of suspicion for associated undiagnosed lymphedema, which will need to be treated as a key component of ulcer management.
Relevance/burden of lymphedema
Care settings
Typically, patients with lymphedema end up receiving treatment at different types of settings: cancer centers, wound clinics, or clinics specialized in lymphedema. There are significant differences in the profile of lymphedema patients seen at these venues [2]:
- Patients seen at cancer centers have cancer-related lymphedema, and most of them are women with breast cancer-related upper extremity lymphedema.
- Meanwhile, patients seen at wound clinics and physical therapists (PT) and occupational therapists (OT) clinics usually have lymphedema unrelated to cancer; many have lymphedema of lower extremities caused by chronic venous disease and obesity impairing lymphatics.
-
- Patients can be referred to wound clinics for the management of complicated wounds, and/or to OT/PT clinics for conservative lymphedema management including compression therapy and manual lymphatic drainage.
- Patients with non-cancer etiologies may find more barriers to diagnosis and treatment. Early diagnosis leads to early treatment and decreased disease burden.[6] However, there is a relatively low awareness of lymphedema among clinicians outside of wound clinics. Most patients are not given a diagnosis until they are referred to a wound clinic.[2] Also, crucial elements for lymphedema management (e.g., compression therapy supplies, manual lymphatic drainage) may not be covered by insurers.[2]
psychoSocial burden of disease on individuals
Individuals with lymphedema report a poorer quality of life (QOL) on surveys compared to their non-diseased counterparts.[7] Impact on QOL frequently arise from feelings of decreased attractiveness, chronic pain and impaired motor function/mobility.[1][7][8]
Financial burden for the healthcare system
In addition to the social burden of disease on individuals, findings below suggest that lymphedema is associated with a significant financial burden in healthcare:
- Lymphedema is a common condition, affecting at least 3 million people in the United States.[9]
- Patients with lymphedema were reported to have more frequent overall hospitalizations and outpatient visits than non-lymphedema patients.[10]
- According to a review of cancer survivors afflicted with lymphedema, total lymphedema-related treatment costs per patient exceeded $2,000 over a 12-month period, with the majority of the burden coming from outpatient services.[10]
- The cost to manage late-stage breast cancer related lymphedema (BCRL) per patient per year is over $3,000.[11]
- Another study found that BCRL patients had an average increase in healthcare costs of $7,000 per year compared to their counterparts with breast cancer but no lymphedema.[12]
Epidemiology
By cause of lymphedema
Primary lymphedema
- Primary lymphedema is estimated to affect 1:6,000-10,000 live births, with female 2.5-10 times more likely to be affected.[13]
- The estimated prevalence of primary lymphedema in the United States is between 1 and 2 million cases.[9]
Secondary lymphedema
- In developed countries, the most common etiology of lymphedema is related to cancer and/or its treatment, affecting 55-80% of total patients. However, because most of these patients are seen at cancer centers, only a minority (~10%) of patients with lymphedema treated at wound clinics has cancer-related lymphedema.[2][14]
- In developed countries, non-cancer related lymphedema due to chronic venous disease, non-cancer surgery, obesity, etc, is also very common and comprises the majority (up to 75%) of patients with lymphedema treated at wound clinics.
- Worldwide, the most common etiology of secondary lymphedema is lymphatic filariasis affecting over 150 million people, primarily in sub-Saharan Africa and Southern Asia.
- Filariasis [15]:
- Currently, 58 countries and 1.23 billion people live in areas where filariasis is endemic
- In Ethiopia where the disease is endemic, prevalence is 9%
- In the United States, where filarial lymphedema is not endemic, incident cases are rare and can often be traced back to visits to endemic countries.[16]
- In the United States, the estimated prevalence of secondary lymphedema is between 2 and 3 million cases.[9]
By anatomical location
Upper extremity lymphedema
- Breast cancer is the main cause of upper extremity lymphedema in the U.S.
- In the general population, approximately 10% of patients with lymphedema have upper extremity lymphedema [17]
- Incidence of secondary upper extremity lymphedema after cancer-related surgery is listed below:
-
- Mastectomy: 24-49% [18]
- Lumpectomy: 4-28% [18]
- Axillary lymph node dissection: 20-30%.[13] More recent studies report lower incidence rates of 4.5-14%.[6]
- Sentinel lymph node sampling: 2-8%.[6] Given the lower incidence of lymphedema, sentinel lymph node sampling is preferred to axillary lymph node dissection in the treatment of breast cancer.[16]
- Because most of these patients are seen at cancer centers, proportion of patients with upper extremity lymphedema at wound clinics is usually low (~1.2% of total patients with lymphedema) [2].
Lower extremity lymphedema
- In the general population, approximately 90% of patients with lymphedema have lower extremity lymphedema.[17]
- Women are affected more frequently than men, and the majority of cases are unilateral (~66% of patients with lower extremity lymphedema) [19].
- Incidence of secondary lower extremity lymphedema after surgery, trauma and irradiation are listed below:
-
- Surgical management of malignant melanoma: 3-29% [20][21]
- Surgical management of soft tissue sarcoma: 19-30% [22]
- Surgical management of gynecological malignancies (cervical, vulvar, endometrial): 1-49% [20][21][23]
- Surgical management of genitourinary malignancies (bladder, penile, prostate), 3-21% [20][21]
- Trauma or irradiation to the groin/abdomen: 20-30% [24]
- In lymphedema patients seen at wound clinics, the prevalence of lower extremity lymphedema was as high as 99.7% according to one study, with the majority being bilateral (~82.9% of patients with lymphedema).[2]
Head and neck lymphedema
- Of the 650,000 people affected with head and neck cancer every year, approximately 54% to 75% develop some form of head and neck lymphedema (internal and/or external).[25] This means that in the general population, up to 351,000 to 488,000 of patients develop head and neck lymphedema annually.
- In lymphedema patients seen at wound clinics, the prevalence of head and neck lymphedema was about 0.3% according to one study.[2]
- Incidence of secondary head and neck lymphedema after squamous cell carcinoma of the head and neck is 5-8%. [21]
Genital lymphedema
- In the general population, less than 1% of patients with lymphedema have genital lymphedema.[17]
- In lymphedema patients seen at wound clinics, the prevalence of genital lymphedema was about 1.5% according to one study. [2]
- Primary lymphedema:
-
- Occurs in 49% of male patients with genital lymphedema [26]
- Occurs in 92% of pediatric patients with genital lymphedema [27]
- Secondary genital lymphedema can be caused by:
-
- Bladder, prostate, rectal cancer
- Hodgkin or non-Hodgkin lymphoma
- Aorto-bifemoral bypass
- Inguinal lymph node biopsy
- Operative intervention for undescended testes in children
By complication of lymphedema among patients seen in wound clinics
- Prevalence of complications among patients with lower extremity lymphedema [2]:
-
- Open ulcers (includes ulcers of mixed etiology): 64.1%
- Cellulitis: 39.9%
- Dermatitis: 34.7%
Risk factors
Many factors have been evaluated for contributing to lymphedema risk. Venous outflow abnormalities, such as deep vein thrombosis and venous hypertension, are currently being studied in association with lymphedema, although none has been clearly identified as risk factors [28].
Risk factors for the development of upper extremity lymphedema include:
- Body mass index in the overweight range or above (BMI >25) [29][30][31][32]
- Lymph node intervention (dissection or radiation) in the treatment of breast cancer and gynecological cancer [29][30][31][32]
- Age greater than 65 at the time of breast cancer treatment [29][33][34]
- Weight gain following breast cancer surgery [35]
- Appearance of cellulitis following breast cancer surgery [29][33][34]
- Adjuvant taxanes in the treatment of BCRL [29][33][34]
- Prior episodes of inflammation on the ipsilateral arm [36]
- Skin puncture and skin infections [36]
- Air travel [36]
- Ipsilateral arm blood pressure measurements [36]
- Extreme temperatures [36]
- Sauna use [30].
- Poor upper body function [37]
Risk factors for the development of lower extremity lymphedema include:
- Lymph node intervention (dissection or radiation) [29][30][31][32][23]
- Trauma and/or surgery on lower extremity (may affect lymphatics) [5]
- Body mass index in the overweight range or above (BMI >25) [29][30][31][32]
- Chronic venous insufficiency (causing phlebolymphedema) [2][38][5]
- Lipedema [2][38]
- Medical comorbidities, including diabetes, congestive heart failure and kidney disease [32]
- Infection [5]
Etiology
Lymphedema is usually classified as primary and secondary.
Primary lymphedema
Primary lymphedema is defined as lymphedema due to inborn defects of lymphangiogenesis resulting in structural abnormalities of the lymphatic system. Primary lymphedema can be sporadic, caused by inherited gene mutations or chromosomal abnormalities, or associated with genetic syndromes (Table 1, Figure 1).[1]
Table 1. Primary causes of lymphedema
| Sporadic lymphedema | Hereditary lymphedema | Syndrome-associated lymphedema |
|
Lymphedema praecox
|
Meige disease
|
Turner syndrome
|
|
Lymphedema tarda
|
Nonne-Milroy disease
|
Noonan syndrome
|
|
|
Milroy disease (hereditary congenital lymphedema)
|
Aagenaes syndrome (cholestasis-lymphedema syndrome)
|
|
|
|
Klinefelter syndrome
|
|
|
|
Patau syndrome
|
|
|
|
Edwards syndrome
|
|
|
|
Yellow nail syndrome
|
|
|
|
Adams-Oliver syndrome
|
|
|
|
Proteus syndrome
|
|
|
|
Hennekam syndrome
|
|
|
|
Kippel-Tranaunay syndrome
|
|
|
|
Lymphedema distichiasis syndrome
|
|
|
|
Prader-Willi syndrome
|
|
|
|
Neurofibromatosis
|
|
|
|
Protein-losing enteropathy |
Figure 1. Teenage girl with primary lymphedema of the left lower extremity. Pitting on the left foot due to shoe can be observed.
Secondary lymphedema
Secondary lymphedema encompasses acquired etiologies resulting from obstruction or disruption of the lymphatics [19][16]:
Lymphatics obstruction:
- Chronic venous insufficiency (phlebolymphedema): resulting from incompetent venous valves, causes pressure buildup of pooled blood within veins that damages the surrounding lymphatics [16]. One of the most common etiologies of lymphedema among patients with lymphedema seen in wound clinics.[2] Given the association between lymphedema and CVI, when seeing venous ulcers clinicians should have a high level of suspicion for associated lymphedema, which will help address all conditions contributing to ulcer formation.
- Obesity: impedes lymph flow through the accumulation of interstitial protein-rich fluid [19] Obesity is one of the most common etiologies of lymphedema among patients with lymphedema seen in wound clinics [2]
- Malignant infiltration or metastasis to lymph nodes: in the setting of lymphoma, melanoma, and squamous cell carcinoma.[1]
- May-Thurner Syndrome: left iliac vein compression by right iliac artery with resulting venous and lymphatic obstruction [39]
- Parasitic infiltration (Filariasis): the most common cause of lymphedema worldwide, but rarely seen in the U.S. Lymphatic filariasis is caused by mosquito-borne worms such as Wuchereria bancrofti, Brugia malayi and Brugia timori.[40]
Lymphatics disruption:
- Surgical disruption of lymphatics (e.g, total hip/knee replacement) and/or radical operative dissection:
-
- Together with irradiation, surgical disruption of lymphatics (accidental or due to operative dissection of lymph nodes) are the most common causes of secondary lymphedema in the United States. These patients are frequently treated at cancer centers, and not at wound clinics.[2]
- The most common cancers associated with iatrogenically acquired lymphedema are those of the breast, gynecologic, genitourinary and head/neck regions [13]
- Trauma involving lymphatics (e.g., long bone fractures, pelvic fractures)
- Recurrent skin infections: recurrent episodes of cellulitis, erysipelas and lymphangitis damage neighboring lymphatic structures and promote lymphatic fluid stasis, in turn leading to increased susceptibility to bacterial injury and repeated episodes of skin infections [41]
- Inflammation due to podoconiosis: another common cause of lymphedema outside the U.S. It is a geochemical disease affecting individuals exposed to volcanic-derived soil containing irritant particles that cause disruption of the lymphatics through granuloma formation after entering through the soles of the feet.[42]
Pathophysiology
Lymphatic system - normal physiology
- Roles of the lymphatic system: vary from maintenance of tissue fluid homeostasis by clearing interstitial fluid of water, lipids, and plasma proteins, to immune cell and antigen transport between the periphery and central lymphoid tissues.[43] See short video on lymphatic system in topic "Lymphedema - Introduction to the Lymphatic System"
- Differences compared to the cardiovascular system: Unlike true circulatory systems, the lymphatic system is an open-loop low-pressure circuit with no central pump. The lymphatic system is composed of multiple structures (Figure 2) [1]:
-
- Lymph nodes (Figure 3): contain lymphocytes and leukocytes that provide immune function and filter lymphatic fluid.
- Lymphatic vessels (Figure 3): with valves that provide unidirectional lymph fluid flow throughout lymphangions (i.e, segments that comprise a lymphatic vessel, the "lymph micro heart"). Lymph flow is facilitated by neighboring muscle contraction and arterial pulsations.
- Lymphatic capillaries (Figure 4): made up of lymphatic endothelial cells separated by gaps, allowing the passage of larger molecules that would not enter blood capillaries. The lymphatics, not the venules, are predominantly responsible for maintaining tissue fluid balance by absorbing interstitial fluid and ultimately returning it into the circulation.[5]
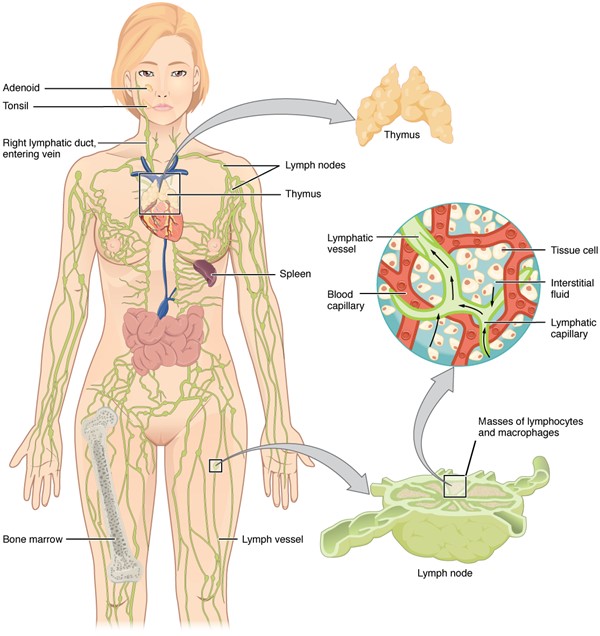
Figure 2: Anatomy of the lymphatic system, composed of lymph nodes, lymphatic collecting vessels and associated lymphoid organs
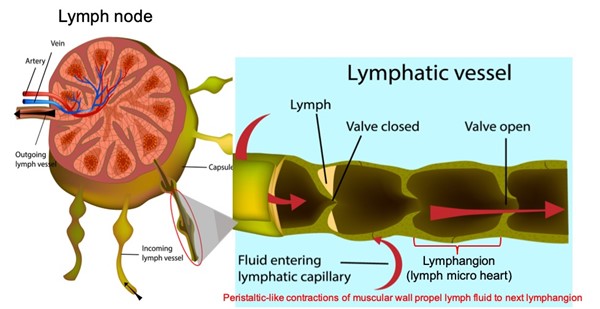
Figure 3: Lymph node and incoming lymph vessel, demonstrating how a lymphangion directs lymphatic flow
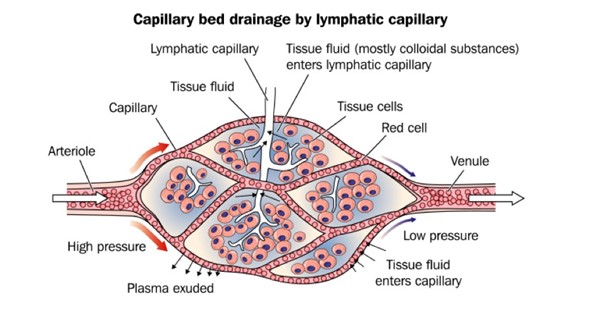
Figure 4. Lymphatic capillary (white capillary in the center) collects larger molecules that would not enter blood capillaries (red)
Pathophysiology of lymphedema
- Lymphedema is caused by lymphatic insufficiency, a condition leading to an imbalance between the rate of lymph production (lymphatic load) and the rate of fluid clearance through lymphatic vessels (lymphatic transport capacity) (Figure 5).[13]
- As lymphatics (and not venules) absorb interstitial fluid and return it to the venous system, experts believe that "lymphatic failure is responsible for all forms of peripheral edema", and that “all chronic edema indicates an inadequacy or failure of lymph drainage.”[5][44][45][46][47]
- Three types of lymphatic insufficiency leading to edema have been described [1][48]:
- Dynamic insufficiency: high-output failure, characterized by lymphatic system overload of microvascular filtrate reducing transport capacity (lymphatic system is normal, but there is too much lymph to be drained)
- Acute: infection, cardiac failure, nephrotic syndrome, trauma, early stages of CVI.[47]
- Chronic: post thrombotic syndrome, end-stage-kidney disease, stable cardiac heart failure with chronic lower extremity swelling, CVI with trophic changes.[47]
- Mechanical (static) insufficiency: low-output failure, characterized by an intrinsic abnormality causing decreased lymphatic transport (due to disruption or obstruction of the lymphatics).
- Disruption or obstruction of any part of the lymphatic system can result in lymphatic load exceeding the normal lymphatic transport capacity, with subsequent abnormal accumulation of interstitial fluid.[13] Causes include [47]:
- Iatrogenic: lymph node dissection, radiation, trauma
- Congenital: genetic abnormality
- Mobility dysfunction: neurological, loss of muscular activity
- Safety valve (combined) insufficiency: a combination of high and low-output failure.
- Consequences of lymphatic insufficiency and abnormal accumulation of interstitial fluid include the development of protein-rich tissue edema, local blood flow disruption, tissue fibrosis progressing to sclerosis, and predisposition to recurrent infection.[43]
- Lymphedema is chronic edema resulting from impairment, malformation or dysfuntion of the lymphatic system.[47] That is, lymphedema is considered a form of end-organ damage affecting the lymphatic system, characterized by fibrosis and sclerosis of lymphatic vessels and their lumens rendering them nonfunctional [49]. As a result of chronic interstitial fluid accumulation, chronic inflammatory pathways and adipocyte proliferation are upregulated, causing further physical damage to lymphatic vessels.
- At the cellular level, disruption/obstruction of lymphatics trigger inflammation and cellular proliferation ultimately leading to fibrosis and irreversible structural lymphatic injury. CD4+ T-lymphocytes, in particular the Th2 variety, drive this structural injury by leading to interstitial fibrosis, inhibiting the development of collateral lymphatic vessels, and inhibiting lymphatic pumping function [50]. Recent studies suggest that the upregulation of the protease chymase and TGF-ß1 are also involved in the development of lymphedema-associated fibrosis [51].

Figure 4: Pathogenesis of lymphedema from lymphatic injury and resulting tissue fluid buildup.
Conditions associated with lymphedema
- Certain states that increase interstitial fluid accumulation and edema can mimic lymphedema clinically but do not involve lymphatic dysfunction and are therefore not involved in its central pathogenesis. These include states of increased capillary permeability, decreased oncotic pressure, and venous hypertension.[13]
- It is important to note that lymphedema and edema of other origins are different entities but can have simultaneous/overlapping clinical manifestations:
-
- Lymphedema is characterized by high-protein edema, with protein concentrations of 1.0-5.5 g/mL which create a state of high oncotic pressure in the interstitium, driving the accumulation of additional water. In contrast, other forms of edema have protein concentrations below 1.0 g/mL.[1]
- It is common however, for lymphedema to be associated with other conditions:
-
- Phlebolymphedema: chronic lymphatic insufficiency associated with chronic venous insufficiency (CVI). See topic "Venous ulcers - Introduction and Assessment". This results in impairment to both clearance of the interstitial space from excess fluid and particles, and return of venous blood to the heart. Lymphedema therefore coexists with tissue edema driven by the increased oncotic pressure of protein-rich interstitial space [52].
-
- In the setting of mild to moderate chronic venous insufficiency, when the venous system becomes overwhelmed and interstitial fluid builds up, the lymphatic system can sustain this increase in interstitial pressure and is able to prevent the development of edema. However in severe chronic venous insufficiency, the lymphatic system can no longer compensate for the increase in interstitial pressure, and edema develops. This condition is known as phlebolymphatic insufficiency [53]. Lymphatic insufficiency, on the other hand, refers to conditions that lead to abnormal accumulation of interstitial fluid, without underlying chronic venous insufficiency.
- Chronic venous insufficiency is one of the most common etiologies of edema among patients seen in wound clinics.[2] Given the association between lymphedema and CVI, clinicians evaluating venous ulcers should have a high level of suspicion for associated undiagnosed lymphedema.
- Lipolymphedema: a more severe form of lipedema that has progressed to lymphedema. Lipedema is characterized by abnormal fatty deposition symmetrically and bilaterally in the lower extremities. It almost exclusively affects females with positive family history. Only in advanced disease states does lipedema involve tissue fibrosis progressing to sclerosis that is characteristic of lymphedema, at which point it is referred to lipolymphedema.[54]
- Chylous reflux: caused by injury to the thoracic duct or lymphatic trunks resulting in reflux of triglyceride-rich chylous fluid transported by the lymphatic system. [1]
Complications of chronic lymphedema
Lymphedema-associated skin changes can be classified into 5 categories [55]:
1. Skin changes directly caused by lymphedema
- Skin fissures and ulcers. See section 'Ulcers' below.
- Lymphorrhea: lymphatic fluid weeping through the skin
- Lymphedema rubra: blanchable erythema caused by vasodilation of tissue affected by lymphedema.
- Papillomas: benign skin and connective tissue tumors
- Dermal fibrosis: woody skin changes associated with venous stasis
- Nodular fibrosis: progression of fibrosis causing the development of nodules
- Elephantiasis nostras verrucosa (ENV): caused by fibroblast proliferation in the setting of chronic lymphostasis and inflammation. ENV is characterized by progressive dermal and subcutaneous fibrosis with hyperkeratosis, papillomatosis and verrucae formation [56]. Obesity, soft tissue infection and chronic venous insufficiency (phlebolymphedema) are risk factors for the development of ENV [57].
- Malignancy: malignant lymphatic transformation into lymphangiosarcoma is a rarer complication of chronic lymphedema. Cutaneous lymphangiosarcoma is referred to as Stewart-Treves syndrome and was originally observed in breast-cancer related lymphedema.[58] Squamous cell carcinoma, Kaposi sarcoma, B-cell lymphoma and histiocytomas have also been reported in patients with chronic lymphedema.[1]
2. Skin changes indirectly caused by lymphedema
- Skin infections. See section on 'Skin infections' below.
- Dermatitis
- Limb enlargement
- Pressure ulcers. See topic "Pressure Ulcers/Injuries - Introduction and Assessment"
3. Skin changes of mixed venous/lymphatic origin
- Phlebolymphedema: lymphatic insufficiency arising in the setting of severe chronic venous insufficiency, where the lymphatic system can no longer compensate for interstitial fluid accumulation. Phlebolymphedema is a distinct entity from lymphedema, which is caused by pure lymphatic insufficiency.
4. Skin changes associated with diseases causing lymphedema
- Nonhealing surgical wounds
- Radiation-induced injury. See topic "Radiation-induced Cutaneous Damage - Introduction and Assessment"
- Traumatic wounds
- Venous stasis ulcers. See topic "Venous ulcers - Introduction and Assessment".
5. Skin changes associated with treatment of lymphedema
- Nonhealing surgical wounds
- Compression stocking erythema/dermatitis
The two most common lymphedema-associated skin changes seen in wound clinics are ulcers and skin infections [2]:
Ulcers
- The predilection to inflammation, infection and parenchymal destruction caused by a dysfunctional lymphatic system ultimately weakens the skin and compromises its role as a protective barrier. This predisposes to skin fissures and ulcers resulting from lymphangiectasia, lymphorrhorea, inflammation and pustule formation, skin maceration, infection, papillomatosis and hyperkeratosis.[59]
- Chronic skin ulcers can occasionally be caused by lymphedema alone.[55] However, they are frequently of mixed origin and their presence should hint at the possibility of associated conditions such as [1]:
-
- Venous stasis ulcers can arise secondary to concomitant venous disease resulting in chronic venous hypertension. Venous hypertension decreases forward arterial blood flow, predisposing to venous stasis and tissue ischemia. The resulting decrease in oxygen and nutrient delivery to affected skin inhibits tissue healing and contributes to ulcer formation [1][60]. Clinicians seeing patients with venous stasis ulcers and skin changes characteristic of lymphedema, such as nodules and woody fibrosis, should maintain a high suspicion for phlebolymphedema. See topic "Venous ulcers - Introduction and Assessment".
- Pressure ulcers can arise secondary to physical pressure, friction or shear from compression garments and bandages used in the management of lymphedema. The pathophysiology of pressure ulcers, like venous stasis ulcers, is attributed to pressure injury resulting in skin and soft tissue ischemia.[1][60] See topic "Pressure Ulcers/Injuries - Introduction and Assessment"
Skin infections
- The structural damage observed in lymphedema manifests as injury to lymphatic endothelial cells resulting in destruction of the lymphatic vascular lumen by fibroblasts. Since the lymphatic system is normally responsible for the elimination of pathogens and clearance of tissue debris, this inflammatory process ultimately leads to increased susceptibility to injury by infectious pathogens and damage to the host’s parenchymal tissue [1].
- The skin is in direct contact with the environment and experiences frequent epidermal injury. This allows bacterial pathogens to easily enter the skin through abrasions, cuts and lesions.
- Because of the lymphatic system’s role in immune protection, lymphedema predisposes to infection through immune dysregulation, ultimately increasing the risk of repeated episodes of infection and inflammation of skin, underlying soft tissue, and lymphatics themselves [1].
- Cellulitis, erysipelas and lymphangitis are the most common forms of infection seen, with recurrent infections increasing the risk of lymphedema progression [61].
-
- Cellulitis and erysipelas are generally caused by infection with group A beta-hemolytic Streptococcus (S. pyogenes) and occasionally by Staphylococcus species. They are characterized by localized erythema, edema and tenderness with centrifugal expansion. In contrast with cellulitis, erysipelas is more frequently characterized by systemic symptoms, including fever, chills and generalized malaise [1].
- Lymphangitis presents as inflammatory erythematous tender streaks that follow the distribution of localized superficial lymphatics. It is also commonly associated with S. pyogenes [1].
- Necrotizing fasciitis is a rarer and potentially fatal infectious complication of lymphedema, characterized by rapidly spreading soft tissue infection with necrosis along the fascia. It presents with indurated edema that extends beyond the margins of the superficial lesion, with possible gas gangrene. A fever is commonly noted with rapid development of hemodynamic instability [1].
- Clinicians seeing patients with skin infection and skin changes characteristic of lymphedema, such as nodules and woody fibrosis, should maintain a high suspicion for phlebolymphedema.
ASSESSMENT
History
History taking is fundamental in the clinical diagnosis of lymphedema. An accurate history and physical exam can diagnose lymphedema in up to 90% of cases.[3] The non-exhaustive checklist below highlights relevant factors that should be assessed when taking history of a patient with lymphedema. Risk factors for development of lymphedema are indicated with the icon R [53]:
Chief Complaint and History of Present Illness
- Symptoms experienced, such as swelling, heaviness, pain
- Time of symptom onset and duration
- History of triggering event, such as penetrating trauma, infection, cancer-related surgery or radiation
- History of medical condition mimicking lymphedema, such as heart failure, hypothyroidism, hypoalbuminemia or nephrotic syndrome
- History of vascular disease mimicking lymphedema, such as chronic venous insufficiency or peripheral artery disease
- History of complications, such as skin infections and ulcers. See section on 'Complications' above
- Assess risk factors for development of lymphedema. See section ‘Risk factors’ above R
- Past treatment history
Surgery/Radiation
- History of oncological surgery, lymphadenectomy or lymph node sampling R
- History of radiation, particularly to the inguinal region or axilla R
Medications
- Adjuvant taxanes in the treatment of breast cancer-related lymphedema (BCRL) [29][33][34] R
- Medications that delay wound healing (in case patient has an ulcer) include: anticoagulants, antimicrobials (various antibiotic classes), anti-angiogenesis agents (eg, bevacizumab, aflibercept), antineoplastic drugs, anti-rheumatoid drugs (eg, methotrexate, aspirin/nonsteroidal anti-inflammatory drugs [NSAIDs]), colchicine (anti-gout drug), topical hydrogen peroxide, topical iodine, full-strength 0.5% Dakin’s solution (sodium hypochlorite), nicotine, steroids, and vasoconstrictors.[62][63]
Social History
- History of substance abuse, particularly smoking which impairs wound healing
- Diet, since obesity increases the risk for lymphedema and high-salt diet increases the risk for fluid retention R
- History of travel to areas endemic for filariasis or podoconiosis R
Family History
- Family history of lymphedema R
Review of Systems
- General:
- Nutritional status: obesity is a risk factor for lymphedema R
- HEENT
- Cardiovascular:
- Heart failure is a risk factor for lower extremity lymphedema R
- Peripheral arterial disease (PAD) may preclude treatment of lymphedema with standard compression therapy, as compression may worsen PAD symptoms
- Endocrine: diabetes is a risk factor for lower extremity lymphedema R , hypothyroidism can lead to edema mimicking lymphedema
- Gastro-intestinal/Genito-urinary: kidney disease is a risk factor for lower extremity lymphedema [32], hypoalbuminemia or nephrotic syndrome can lead to edema mimicking lymphedema
- Neurological: functional assessment, including activities of daily living and activity status
- Musculoeskeletal: functional assessment, including activities of daily living and activity status
- Skin: integumentary integrity
Patient's and caregivers' concerns
Patients with lymphedema report a poorer quality of life (QOL) on surveys compared to their non-diseased counterparts.[7] Common patients’ concerns include:
- The chronic and progressive nature of the disease
- Uncertainty about disease progression, prognosis and curative treatments
- Uncertainty regarding insurance coverage
- Cancer relapse in lymphedema cases secondary to malignancy (majority of cases in the U.S.)
Impact on patients’ QOL frequently stem from [7][1] :
- Feelings of decreased attractiveness due to the disfiguring nature of the disease
- Fear of stigmatization
- Chronic pain, numbness or tingling due to sensory nerve involvement
- Potential need to change profession due to impaired motor function and decreased mobility, particularly when the extremities are affected
Patient's and caregiver's concerns and psychosocial aspects should be assessed and taken in consideration when creating a treatment/prevention plan. Concerns and psychosocial aspects include but are not limited to:
- Values and goals of care of the individual and/or the individual’s significant others.
- Psychological health, behavior, and cognition.
- Pain:
Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"- To assess pain, validated pain scales are recommended (see 'Documentation' below), but they may not capture sufficient information to guide intervention. An initial pain assessment should include the following four elements:
- A detailed pain history including the character, intensity, triggering factors and duration of pressure ulcer pain (incorporate the individual's body language, non-verbal cues and words that describe pain)
- A physical examination that includes a neurological component;
- A psychosocial assessment; and
- An appropriate diagnostic work-up to determine the type and cause of the pain.
- Social and financial support systems
- Resources available to the individual
- Ability to adhere to a prevention and management plan
- Knowledge and belief about management of lymphedema
- Refusal of some aspects of care and treatment
We recommend use of Patient-Reported Outcome Tools to assess aspects above and measure impact of interventions. See 'Patient-reported outcomes (PRO) tools' below.
Physical Examination
A complete physical examination is essential to clinically assess lymphedema. Local exam of patients with lymphedema is described below.[3] A Lymphedema Assessment Form may be used for initial assessment and follow-up visits to monitor progress (See Lymphedema Assessment Form Template/Checklist).[64] When performing a local exam, it is important to use a consistent lymphedema measurement technique and location of measurement (i.e, fixed distance from the floor). Measurement variability should be reduced by picking a constant time throughout the day to perform local measurements. It is also essential to determine the current compression method used to ensure adherence and correct device operation.
Local Exam
- Location affected: extremities are the most common, followed by genitalia, head and neck, and abdomen.
- Swelling: presence of pitting or non/pitting edema, lymphadenopathy, degree of shape distortion, preferential swelling of distal extremity that progresses proximally and affects the dorsum of hands/feet.
- Skin changes and complications of lymphedema (Figures 6-11): color, pigmentation, temperature, dryness, fragility, dermatitis, ulcers, skin infections, lymphangiectasia, lymphorrhea. Examples of skin changes that can arise in advanced disease include: hyperkeratosis, peau d’orange, papillomatosis and nodular fibrosis, lymphangiosarcoma and elephantiasis nostras verrucosa. See lymphedema-associated skin changes in section 'Complications' above.
- Ulcers: features seen in chronic lymphedema leg ulcers include [59]:
- Lymphorrhea with superficial eczematous ulcerated lesion
- Established ulcerated lesions
- Ulceration with deep sinus tract in elephantiasis legs
- Ulcers developed after excision procedures
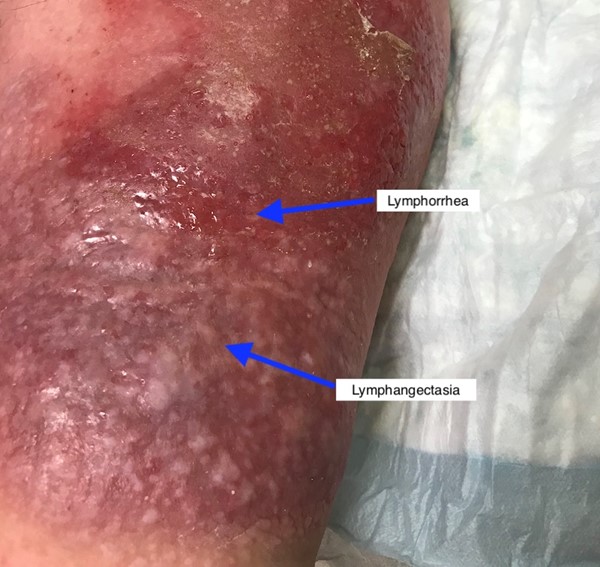
Fig. 6. Lymphedematous leg with lymphangectasia and lymphorrhea | 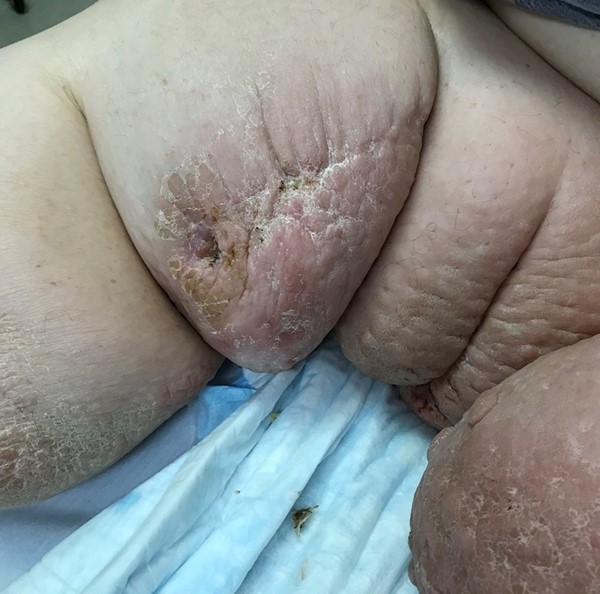
Fig. 7. Lymphedematous leg with hyperkeratosis, peau d'orange, and post-surgical scar | 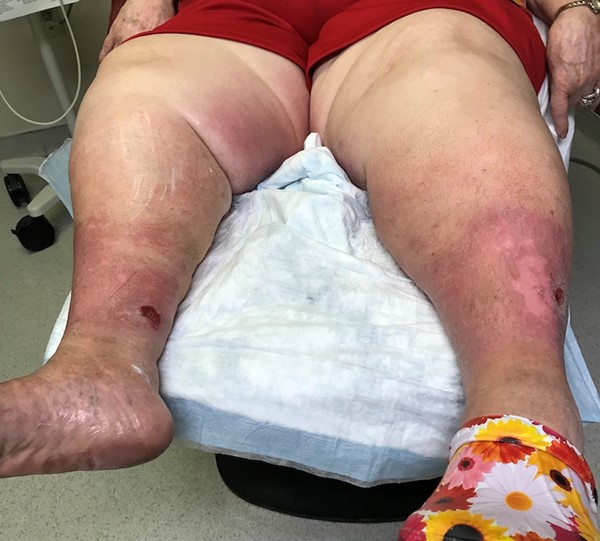
Fig. 8. Lymphedematous legs and ulcers |
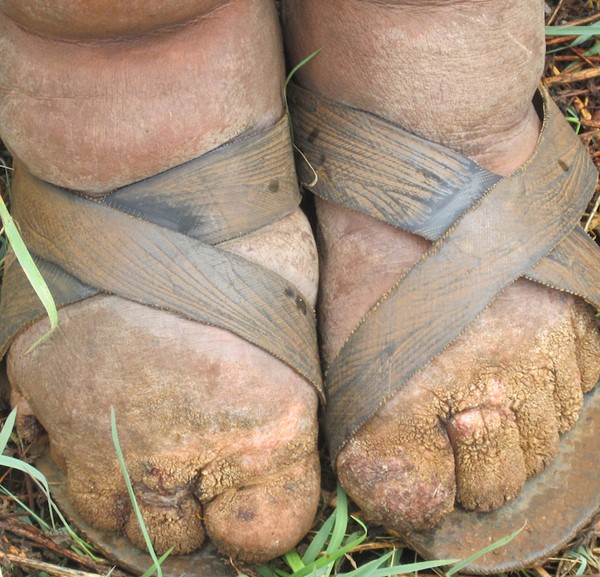
Fig. 9. Elephantiasis nostras verrucosa due to podoconiosis | 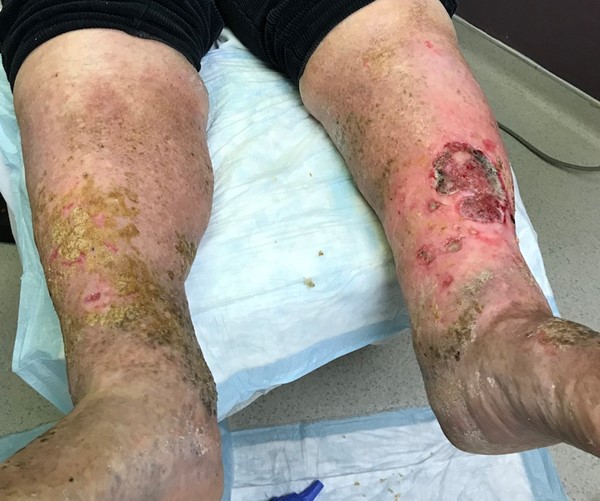
Fig. 10. Phlebolymphedema and ulcer | 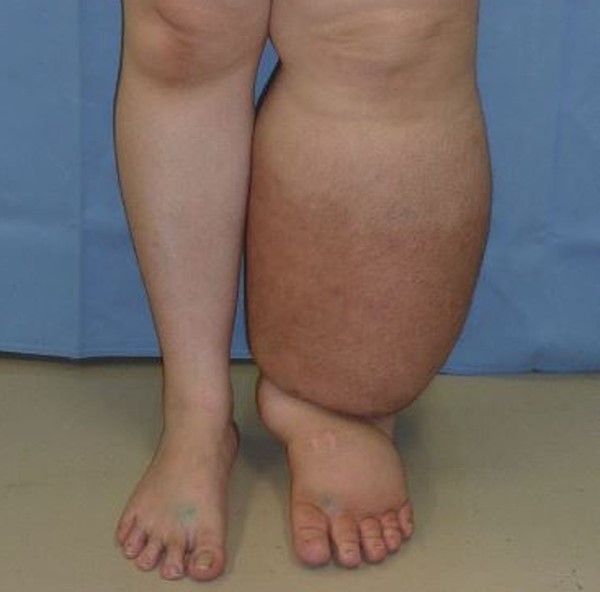
Fig. 11. Right lower extremity lymphedema
|
- Stemmer sign (Figure 12): moderately sensitive and specific for lymphedema [65]
-
- Grade I: thickened skinfolds that can be raised, applying pressure reduces the thickness
- Grade II: skinfolds can still be raised, applying pressure does not reduce thickness
- Grade III: skinfolds cannot be raised, smooth skin
- Grade IV: skinfolds cannot be raised, hyperkeratotic skin changes with box-shaped toes

Figure 12: Grade IV Stemmer sign. Inability to raise skinfolds between fingers and the presence of hyperkeratosis with box-shaped toes. [48]
- Sensory changes and pain assessment: deep bone pain, ulcer pain, arthritic pain.
- Lipedema assessment: diet-resistant bilateral fat deposits in legs or arms, sharp demarcation between normal and abnormal tissue (swelling stops at ankles or wrist), pain and bruising are frequent features. In pure lipedema, Stemmer sign is often negative, often painful on pinching.[64]
- Mobility and functional assessment: range of movement, ability to wear and remove garments, ability to hand grip and pincer grasp in upper extremity lymphedema, gait assessment in lower extremity lymphedema, effect on lymphedema on activities of daily living.
- Limb circumference measurements: expert committees recommend baseline and follow-up measurements of the affected areas (if limbs, ipsilateral and contralateral limb).[29][6] Measurements include:
- Circumference (in centimeters):
- A flexible, non-stretch measuring tape, which is the most used method, widely accessible and inexpensive.
- Limb volume (in milliliters) :
- Volume can be estimated by inputting the circumference value measured in centimeters in the truncated cone formula. It can be time-consuming and accuracy and reproducibility depend on clinician’s familiarity with the method and time of the day (needs to be measured at the same time to be meaningful, as swelling may increase throughout the day)
- Truncated cone formula: V = 1/12 x h x (C2 + Cc + c2), where V = volume, h = cone height (distance from proximal to distal circumference), C = proximal circumference, and c = distal circumference
- Water displacement volumetry: is another alternative for calculating arm or lower leg volumes. Practical limits include size of limb, isolating the affected area, measuring areas near the root of the limb, and hygiene issues.
- Perometry: measures limb volume with infrared light beams. It is quick, accurate, easy to reproduce and capable of identifying subclinical disease. However it is still costly and hand and foot are not included.
- Photography: recommended to monitor facial and truncal swelling, since there is no effective method to measure lymphedema of the head and neck, breast, abdomen or genitalia.[35]
- Vascular assessment: it is important to rule out signs and symptoms of PAD prior to application of compression therapy (a cornerstone in lymphedema management), as compression may worsen tissue ischemia.
- Vascular status is primarily determined by history and clinical examination:
- A detailed history of the presenting complaint including night time rest pain and intermittent claudication
- A clinical examination of the feet and lower limbs showing signs such as color changes (e.g. white, pale, especially upon elevation), delayed capillary refill, decreased peripheral pulses
- Bedside arterial non-invasive tests:
- Ankle-brachial index (ABI): Lymphedema best practices documents recommend performing ABI to rule out PAD prior to application of compression therapy. However, experts acknowledge that conducting ABI properly in lymphedema patients may be difficult, due to significant edema, larger limb size, pain, tissue fibrosis, etc. The British Lymphology Society no longer requires ABI measurement in lymphedema patients due to these reasons.[66] Instead, clinicians should rely on clinical assessment. If familiar with a handheld Doppler, clinicians can also utilize this modality at the point-of-care to support clinical findings. If clinicians do not have familiarity with handheld Doppler devices, patient can receive compression therapy as long as there are no significant cardiovascular risk factors and clinical signs or symptoms of PAD and provided the vascular status has been thoroughly assessed.[66]
- Handheld Doppler waveform: patients with bi and triphasic signals and normal clinical assessment can receive standard compression (>30 mmHg). Patients with monophasic signals should be referred to a vascular specialist for assessment. If asymptomatic, patients with monophasic signals can receive modified compression (10-20 mmHg) while waiting for vascular assessment as long as patients can be closely monitoring for any symptoms of PAD. Patients who are symptomatic should not receive compression therapy while waiting to be seen by a vascular specialist. See algorithm for vascular assessment with handheld Doppler waveform on "How to Select Adequate Compression Therapy Pressure Levels and Products"
- Documentation: if a ABI is not conducted, documentation must be provided to/from care team, demonstrating findings of the clinical assessment and rationale for not completing an ABI
Diagnosis
Diagnosis of lymphedema is mainly clinical, based on a comprehensive history and physical exam.[5] However, many diagnostic modalities are used to corroborate evidence and assess severity. Patient comorbidities, such as congestive heart failure, thyroid disease, hypertension and cerebrovascular disease should be taken under consideration when choosing the most appropriate diagnostic modality. [6][29]
For cases with unclear diagnosis of lymphedema
Referral to a lymphologist or lymphologic center is recommended for clarification of prognosis and treatment plan.
Imaging
Imaging modalities commonly used in case of diagnostic uncertainty include [29][67][68]:
- Lymphoscintigraphy: considered the gold imaging standard to confirm diagnosis and plan surgical intervention. It measures lymph flow in both central and peripheral lymphatics using a radio-labeled tracer. Since the diagnosis of lymphedema is mainly clinical, the test is not required in most patients. Lymphoscintigraphy might be indicated in patients who do not respond to standard conservative management of lymphedema.[69]
- Duplex ultrasonography: Initial test to rule out venous disease, useful to characterize underlying venous involvement in patients with lower limb edema. It allows for measurement of volumetric changes and structural soft tissue changes, but cannot be used to visualize lymphatic anatomy
- Other diagnostic tools include:
-
- Lymphangiography: popular before lymphoscintigraphy became the standard, requires accessing the lymphatics through a skin incision and therefore carries the risk for infection, inflammation and hypersensitivity reaction, still used for preoperative visualization of lymphatics.
- Computed tomography: useful for characterizing structural changes in the diagnosis of lymphedema and in response to treatment.
- Magnetic resonance imaging: similar uses as computed tomography with better soft tissue resolution. May provide complementary findings to lymphoscintigraphy.[70]
Genetic testing
Genetic testing is indicated in cases with a strong family history of lymphedema that might be associated with a specific underlying hereditary syndrome, or in cases where syndromic features might indicate the presence of a primary genetic cause. Genetic tests include: [71]
- FLT4 (Milroy disease)
- GJC2, VEGFC, PIEZO1 (inherited lymphedemas)
- FOXC2 (lymphedema-distichiasis syndrome)
- SOX18 (hypotrichosis-lymphedema-telangiectasia)
- CCBE1 (Hennekam syndrome)
- PTPN14 (choanal atresia-lymphedema)
- GJA1 (oculodentodigital syndrome)
- HGF (lymphedema-lymphangiectasia)
- PTPN11, SOS1 (Noonan syndrome)
- GATA2 (Emberger syndrome)
- Chromosomal aneuploidies
For early diagnosis of lymphedema in individuals at higher risk of developing secondary lymphedema (e.g, patients who undergo lymphadenectomy)
Expert committees recommend establishment of a surveillance plan, as early diagnosis leads to early treatment and may limit disease burden.[29][6]
Assessment of early fluid changes in the subclinical stage before lymphedema is clinically obvious can be accomplished using bioimpedance spectroscopy or tissue dielectric constant analysis [29] [6]:
- Bioimpedance spectroscopy: used to analyze the composition of fluid compartments of the body. Quick objective measure that is FDA-approved, can identify subclinical cases of lymphedema but has no role in established disease.
- Tissue dielectric constant: used to quantify localized tissue water in affected body part. Quick and portable, further guidelines needed to establish standard thresholds.
- As for standard thresholds, a study analyzing normal individuals proposed that a calf/forearm tissue dieletric constant ratio that exceeds 1.35 is suggestive of lower extremity lymphedema. The calf/forearm ratio could be used to track temporal changes and therapy-related improvements.[72]
Differential Diagnosis
Systemic and local conditions causing limb edema may mimic lymphedema:
Systemic causes of edema
Systemic causes of leg edema generally present with bilateral lower extremity edema and a characteristic constellation of symptoms originating from dysfunction of the target organ [1]:
- Congestive heart failure
- Cirrhosis, hypoalbuminemia
- Renal failure, nephrotic syndrome
- Protein-losing enteropathy
Local causes of edema
Local causes of leg edema presents with unilateral or bilateral leg involvement and might be more difficult to differentiate given the nonspecific symptoms exhibited [68][3]:
- Chronic venous insufficiency (phlebolymphedema) (Figure 13): the most common cause of lower extremity swelling in adults, generally bilateral, the risk increases with age, obesity, hypertension and a sedentary lifestyle, caused by valvular incompetence with subsequent reflux of pooled venous blood.
- Deep vein thrombosis: swollen, warm and tender calf in the setting of pro-coagulatory risk factors, such as recent surgery, immobility, malignancy, and thrombophilia.
- Post-thrombotic syndrome: complication of chronic deep venous thrombosis resulting from thrombus-induced valvular disruption.
- Postoperative complications following ipsilateral surgery: seen particularly in the case of arterial reconstructive surgery, may persist up to 3 months.
- Cellulitis (Figure 14): swollen, warm and tender calf in the setting of skin lesion acting as a site of pathogen entry, more common in immunocompromised population.
- Arthritis: swollen, warm and tender joint in the setting of rheumatologic disease.
- Baker’s cyst: cystic enlargement of the popliteal bursa, often in the setting of obesity and knee arthritis.
- Trauma: post-traumatic swelling can be mistaken for lymphedema, especially in the setting of a sprain or fracture.
- Venous malformation: venous malformations located within deep structures, such as muscle, bone or joints, can intermittently bleed and appear as extremity swelling. In chronic cases, venous malformation injury to deep structures can result in leg-length discrepancy, disuse atrophy, and degenerative arthritis.
- Lipedema (Figure 15): lipodystrophy affecting only the extremities but sparing the most distal parts (hands/feet), often bilateral, seen in females during periods of hormonal change, such as puberty, pregnancy and menopause. It can present with easy bruising and pain. Lipedema is associated with a lower risk of diabetes mellitus compared to obesity.[73] It can be inherited in an X-linked dominant or autosomal dominant fashion.[74] In late stages, the fat accumulation of lipedema can compress lymphatic vessels and contribute to lymphatic dysfunction, resulting in lipolymphedema.[75]
- Obesity (Figures 16-18): may present as asymmetric subcutaneous fat deposition resulting in leg enlargement, separate entity from lipedema. Frequently associated with lymphedema.
- Myxedema: dermatologic involvement in patients with moderate-to-severe thyroid disease, caused by mucopolysaccharide deposition in the dermis, classically affects the lower legs.
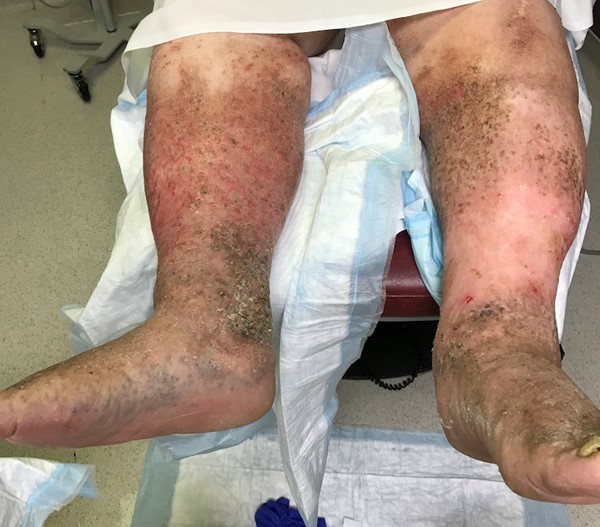
Fig. 13. Phlebolymphedema | 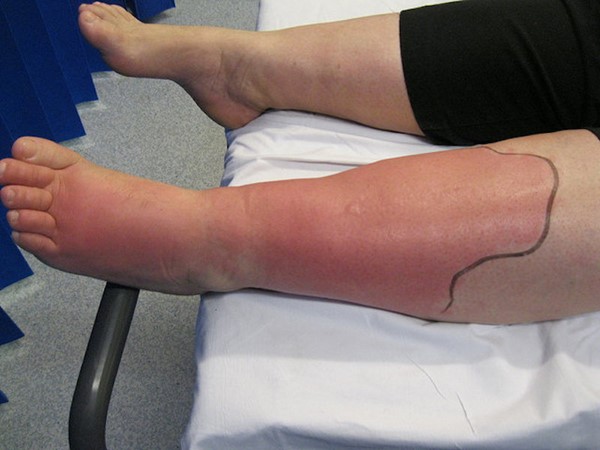
Fig. 14. Cellulitis on the left leg |
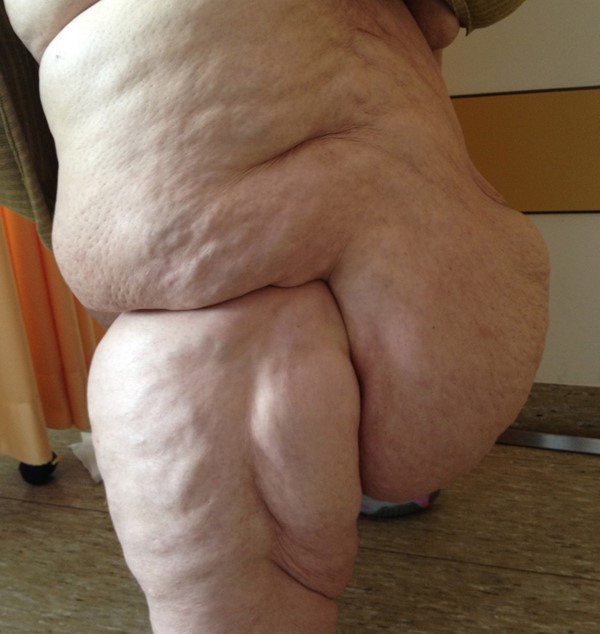
Fig. 15. Lipedema. Lateral aspect of right leg | 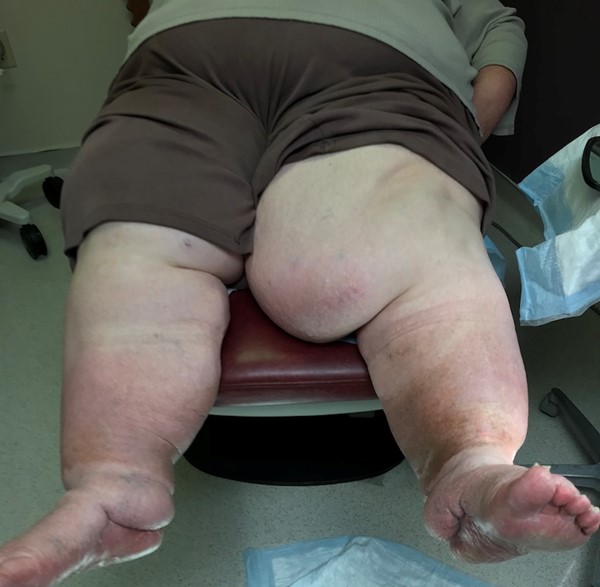
Fig. 16. Lymphedema associated with obesity
|
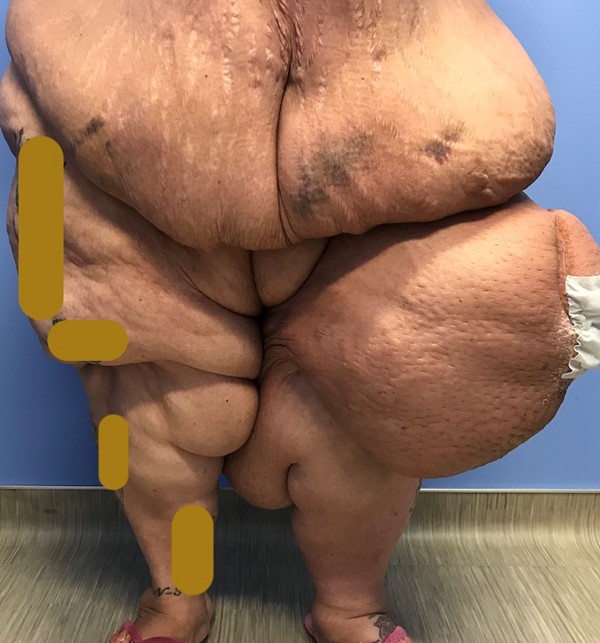
Fig. 17. Lymphedema associated with obesity, female patient | 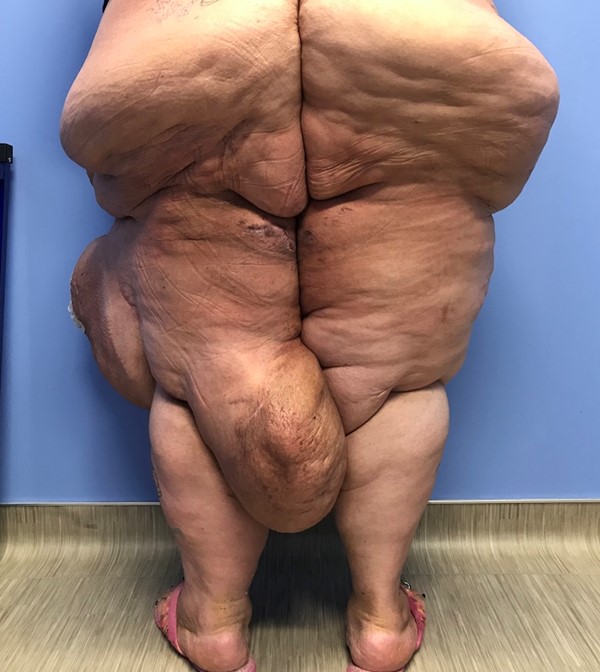
Fig. 18. Lymphedema associated with obesity, female patient |
Clinical Staging of Lymphedema
Multiple guidelines have been developed for the clinical staging of lymphedema. The most widely used by clinicians is by the International Society of Lymphology. The New Taiwan Lymphoscintigraphy classification has been gaining interest recently for incorporating objective measures into its staging assessment.
Lymphedema staging according to the 2016 consensus document of the International Society of Lymphology: [29]
- Stage 0: subclinical disease, swelling absent despite underlying impaired lymphatic transport
- Stage 1: tissue swelling present but reduced by limb elevation. Pitting may occur
- Stage 2: limb swelling partially reduced by elevation, pitting is manifest. Later in stage 2, limb may not pit due to excess subcutaneous fat or fibrosis
- Stage 3: lymphostatic elephantiasis with skin changes (e.g., acanthosis, thickening, further fibrosis, warty overgrowths) and recurrent infections
Lymphedema staging according to the New Taiwan Lymphoscintigraphy system: [76]
- Stage L0: normal lymphatic drainage, good visualization of proximal lymph nodes, good visualization of lymphatic ducts
- Stage P1: partial obstruction, good visualization of proximal lymph nodes, distal visualization of lymphatic ducts only
- Stage P2: partial obstruction, reduced visualization of proximal lymph nodes, distal visualization of lymphatic ducts only
- Stage P3: partial obstruction, no visualization of proximal lymph nodes, no visualization of lymphatic ducts but retained visualization of intermediate lymph nodes
- Stage T4: total obstruction, no visualization of proximal lymph nodes, engorged lymphatic ducts visualization with distal dermal backflow only
- Stage T5: total obstruction, no visualization of proximal lymph nodes, engorged lymphatic ducts visualization with complete dermal backflow
- Stage T6: total obstruction, no visualization of proximal lymph nodes, no visualization of lymphatic ducts without intermediate lymph node visualization
Assessment of lymphedema severity
Lymphedema is classified in stages (see 'Clinical Staging' above). Within each stage, severity can be assessed and classified according to simple volume differences of the affected part, that is, the change from baseline in swelling not due to weight change (i.e, if limbs, determined by measuring both limbs). Severity can be classified as follows [29]:
- Minimal: typically > 5 to < 20% increase in limb volume
- Moderate: 20-40% increase
- Severe: > 40% increase
- Some clinics prefer to use > 5 to < 10% as minimal and > 10 to < 20% as mild
Severity can also be assessed with factors such as extensiveness, occurrence of erysipelas attacks, inflammation, and other descriptors or complications [29].
Documentation
Documentation of lymphedema and its complications is important to monitor and assess adequacy of treatment plans.
Frequency of documentation
- In cases of lymphedema-associated ulcers, physical appearance should be documented weekly with focus on ulcer number, position, size, depth and drainage characteristics. More frequent documentation is recommended if ulcer appearance or symptoms worsen, such as onset of skin infection, dermatitis, maceration or non-healing wound.
- Postoperative surveillance of cancer survivors at risk of development of lymphedema should be documented regularly in 3-month intervals up to 12 months following surgery [77].
Documentation to support ICD-10 of lymphedema
- Findings that support the diagnosis of lymphedema, such as
-
- Cause (primary, secondary), risk factors
- Date of onset of symptoms and other pertinent history
- Objective findings that establish the severity of the condition
-
- Baseline and follow-up measurements of the ipsilateral and contralateral limb to assess for asymmetry recorded in measurement charts
- Severity staging using the International Society of Lymphedema guidelines
- Presence of lymphedema-specific findings:
-
- Positive Stemmer sign,
- Location affected and description of limb swelling (pitting/nonpitting), presence of dorsal foot swelling
- Fibrosis, papillomas, lymphatic blisters or weeping, recurrent infections
- Ulcer number, position, length, width and depth. For ulcers, tools that facilitate standardized assessment and documentation should be used whenever possible:
- Validated wound assessment tools: Bates-Jensen Wound Assessment Tool [78]
- Wound Reference Wound Prep&Dress Tool creates notes to help support medical necessity that can be copied and pasted to electronic medical records
- Wound imaging: digital photographs of PU/PI at the first consultation and periodically thereafter to document progress is helpful and ensures consistency of care among healthcare practitioners, facilitates telemedicine in remote areas, and illustrates improvement to the patient.
- Wound measurement: methods include manually measuring length and width (the longest length with the greatest width at right angles), manual tracing, digital photography, and software programs that calculate wound dimensions from a photograph of the lesion. Wound tracings that calculate the area via digital software are slightly better than linear measurement.[79]
- Findings pertaining to lymphedema therapy:
-
- Use of compression stockings
- Leg elevation
- Exercise
- History of adherence
Patient-reported outcome (PRO) Tools:
Validated instruments allow clinicians to obtain feedback on whether a given treatment plan is resulting in the best outcomes possible and is improving patient's quality of life (QOL). PRO assessment will continue to gain importance as health care systems transition from a volume-based reimbursement model to a value-based model.
- QOL tools validated for lymphedema patients: the 36-item Short Form (SF-36), ULL-27 [80], LYMQOL[81]
- Medicare Quality Payment Program, Improvement Activity "Promote Use of Patient-Reported Outcome Tools" suggests use of Wound-Quality of Life (QoL) and patient-reported Wound Outcome if patient has a wound. [82]
- Validated pain measurement tools include:
- Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up": Wong-Baker FACES scale; for adults - visual analog scale (VAS).
- Disability assessment: World Health Organization’s guidelines for the International Classification of Functioning, Disability, and Health (ICF) may be used to rate disability.[29]
CODING
ICD-10
(see codes in the Appendix)
Lymphedema ICD-10 code I98.0 includes:
- Elephantiasis (nonfilarial) NOS
- Lymphangiectasis
- Obliteration, lymphatic vessel
- Praecox lymphedema
- Secondary lymphedema
See other codes in the Appendix
If ulcer is present, code underlying condition first (e.g. lymphedema), then code ulcer as follows:
- Specify laterality
- Right, left or unspecified
- Specify ulcer severity:
- Limited to breakdown of skin
- With fat layer exposed
- With necrosis of muscle
- With necrosis of bone
- Unspecified severity
Diagnostic Related Group
ICD-10-CM I89.0 is grouped within Diagnostic Related Group(s) (MS-DRG v36.0):
- 606 Minor skin disorders with mcc
- 607 Minor skin disorders without mcc
APPENDIX
ICD-10
ICD-10 - lymphedema
- I89.0 Lymphedema, not elsewhere classified
- Compression of lymphatic vessel
- Dilatation of lymphatic vessel
- Edema due to lymphatic obstruction
- Edema, lymphatic
- Elephantiasis (nonfilarial), lymphangiectatic
- Elephantiasis (nonfilarial), lymphatic vessel
- Elephantiasis (nonfilarial), glandular
- Elephantiasis (nonfilarial), scrotum
- Elephantiasis (nonfilarial), streptococcal
- Elephantiasis (nonfilarial), telangiectodes
- Lymphangiectasis, postinfectional
- Lymphangiectasis, scrotum
- Lymphangiectatic elephantiasis
- Lymphectasia
- Lymphedema praecox
- Lymphedema secondary
- Obliteration of lymphatic vessel
- Obstruction of lymphatics
- Obstruction of thoracic duct
- Occlusion of lymph or lymphatic channel
- Occlusion of thoracic duct
- Q82.0 Hereditary lymphedema
- Chronic hereditary lymphedema
- Congenital elephantiasis
- Congenital lymphedema
- Familial lymphedema
- Hereditary lymphedema of the legs
- Idiopathic lymphedema
- Meige’s disease/syndrome
- Milroy’s disease/syndrome
- Nonne-Milroy-Meige disease/syndrome
- Pseudoelephantiasis neuroarthritica
- Trophedema
- Tropholymphedema
- I97.89 Other postprocedural complications and disorders of the circulatory system, not elsewhere classified (includes post surgical lymphedema)
- Complication of circulatory system, postprocedural, not elsewhere classified
- Complication from surgical procedure on elephantiasis
- Complication from surgical procedure on lymphedema
- Elephantiasis (nonfilarial), surgical
- Lymphedema (acquired), surgical, not elsewhere classified
- I97.2 Postmastectomy lymphedema syndrome
- Complication from surgical procedure on elephantiasis or lymphedema, postmastectomy
- Complication of lymphedema after mastectomy
- Elephantiasis (nonfilarial) due to mastectomy
- Elephantiasis (nonfilarial), postmastectomy
- Lymphatic edema due to mastectomy
- Lymphedema acquired postmastectomy
- Obliteration of lymphatic vessel due to mastectomy
- B74.0 Filariasis due to Wuchereria bancrofti
- N94.89 Female Genital Lymphedema
- N50.8 Scrotum-Genital Lymphedema
ICD-10 Associated conditions
- I87.2 Chronic Venous Insufficiency
- L03.XXX Cellulitis
- non-pressure ulcer
Red arrows indicate non-billable code, Green arrows are billable codes
 L97.2 Non-pressure chronic ulcer of calf
L97.2 Non-pressure chronic ulcer of calf
 L97.20 Non-pressure chronic ulcer of unspecified calf
L97.20 Non-pressure chronic ulcer of unspecified calf
 L97.201 …… limited to breakdown of skin
L97.201 …… limited to breakdown of skin
 L97.202 …… with fat layer exposed
L97.202 …… with fat layer exposed
 L97.203 …… with necrosis of muscle
L97.203 …… with necrosis of muscle
 L97.204 …… with necrosis of bone
L97.204 …… with necrosis of bone
 L97.209 …… with unspecified severity
L97.209 …… with unspecified severity
 L97.21 Non-pressure chronic ulcer of right calf
L97.21 Non-pressure chronic ulcer of right calf
 L97.211 …… limited to breakdown of skin
L97.211 …… limited to breakdown of skin
 L97.212 …… with fat layer exposed
L97.212 …… with fat layer exposed
 L97.213 …… with necrosis of muscle
L97.213 …… with necrosis of muscle
 L97.214 …… with necrosis of bone
L97.214 …… with necrosis of bone
 L97.219 …… with unspecified severity
L97.219 …… with unspecified severity
 L97.22 Non-pressure chronic ulcer of left calf
L97.22 Non-pressure chronic ulcer of left calf
 L97.221 …… limited to breakdown of skin
L97.221 …… limited to breakdown of skin
 L97.222 …… with fat layer exposed
L97.222 …… with fat layer exposed
 L97.223 …… with necrosis of muscle
L97.223 …… with necrosis of muscle
 L97.224 …… with necrosis of bone
L97.224 …… with necrosis of bone
 L97.229 …… with unspecified severity
L97.229 …… with unspecified severity