The following hyperbaric medicine treatment protocol is based upon the recommendations of the Hyperbaric Oxygen Committee of the Undersea and Hyperbaric Medical Society. Clinical protocols and/or practice guidelines are systematically developed statements that help physicians, other practitioners, case managers and clients make decisions about appropriate health care for specific clinical circumstances.
Protocols allow health providers to offer evidence-based, appropriate, standardized diagnostic treatment and care services to patients undergoing hyperbaric oxygen therapy (HBOT). Evidence-based medicine offers clinicians a way to achieve improved quality, improved patient satisfaction, and reduced costs. Utilization Review should be initiated when clinical decisions result in deviation from or modification of treatment protocols. This includes any course of treatment at or above the recognized threshold limits.
|
HBO request requirements checklist | Sample Physician Order | $ ICD-10 Crosswalk | Treatment Table | Emergent Indication
|
| Background |
Carbon monoxide (CO) can cause hypoxic stress to tissues (e.g., heart or brain) leading to injury. Common sources of CO include gasoline engines, carbon-fueled appliances, and propane heating and cooking sources. CO binds preferentially to hemoglobin in place of oxygen, leading to tissue hypoxia. The blood level of carbon monoxide has not correlated well with resultant persistent neurological deficits. Although there are no clinical studies demonstrating improved outcomes using normobaric oxygen administration of supplemental oxygen has long been the cornerstone of therapy for patients suffering CO poisoning.
An intense inflammatory response has been documented in cases of CO poisoning. This may have some bearing on the persistent neurologic deficits that are seen clinically. The application of HBOT is of significance in improving the cognition of delayed encephalopathy patients. HBOT accelerates the elimination of carboxyhemoglobin (COHb) and positively influences the inflammatory responses triggered by carbon monoxide poisoning.[2][3] HBOT may improve delayed encephalopathy after CO poisoning by mobilizing circulating stem cells with up-regulation of brain-derived neurotrophic factor.[2] |
Goals of HBOT
|
- Accelerate dissociation of CO from the hemoglobin molecule (at a much greater rate than that achievable by breathing pure oxygen at ambient atmospheric pressure)
- Reduce brain inflammation and injury (by enhancing tissue oxygenation, inhibiting brain lipid peroxidation, decreasing microvascular injury, and improving mitochondrial oxidative processes)
|
Diagnosis
|
- Carbon monoxide poisoning is a clinical diagnosis based on the presence of symptoms and a documented CO exposure (i.e., history of inhalation of CO or elevated CO levels).
- The two organ systems most susceptible to injury from CO are the cardiovascular and central nervous system
- With non-lethal exposures to CO, clinical symptoms include headaches, nausea, vomiting, dizziness, general malaise, altered mental status, chest pain, and/or shortness of breath
- Symptoms are not well correlated with blood levels of CO except in high concentrations with unconsciousness and death
- CO exposure can be confirmed by COHb, but the COHb level is not predictive as a risk factor for CO mediated delayed morbidity or mortality
- A COHb level of at least 3-4% in non-smokers, or 10% in smokers, indicates likely exogenous CO exposure
|
HBOT Criteria
|
- Signs of serious poisoning (transient or prolonged unconsciousness, neurologic signs, cardiovascular dysfunction, or severe acidosis), regardless of COHb level
- Prognosis is poorer for patients with underlying cardiovascular disease, are older than 60 years of age, have suffered any interval of unconsciousness due to the CO poisoning, or demonstrate severe acidosis.
- In patients with less severe symptoms, COHb levels between 25% and 30% or >15% in pregnant women warrants referral to HBOT
- Abnormal neuropsychological testing should warrant HBOT referral
- There are some facilities that take all patients referred with a documented exposure to CO and any sign/symptom, regardless of COHb level
|
| Evaluation | Administration of 100% supplemental oxygen utilizing a non-rebreather mask during the evaluation is recommended. - Focused comprehensive history if obtainable (source, time exposed, how much time has elapsed since the patient has left the toxic environment, symptoms)
- Physical examination:
- Neurological: serial neurologic exams and neuropsychometric testing to assess progress, and to detect signs of developing residual neurological deficits. An initial low Glasgow coma scale score (<9) in patients with early CO poisoning has been shown to be associated with the occurrence of delayed neurologic sequelae.[4]
- Cardiovascular: tachycardia, tachypnea, hypotension
- Laboratory Tests
- Arterial Blood Gas (ABG) with Carboxyhemoglobin (COHb)
- Complete Blood Count (CBC)
- Comprehensive Metabolic Panel (CMP)
- Lactate
- Cardiac enzymes (Creatinine Phosphokinase (CPK) & Troponin (If your hospital has an MI laboratory protocol, use that set of blood tests.)
- Chest x-ray
- Electrocardiogram (ECG)
- Imaging: brain computed tomography (CT) or magnetic resonance imaging (MRI) may show signs of cerebral infarction secondary to hypoxia or ischemia.
- Drugs of abuse screen (serum and urine) if suspected suicide attempt
- For childbearing-age women with potential CO poisoning: pregnancy test is indicated (HBOT is the treatment of choice for CO poisoning in pregnant women)
- For pregnant patient: consult obstetrician for evaluation and fetal heart rate monitoring. HBOT should be considered for COHb levels of 15-20% and signs of fetal distress. Fetal hemoglobin has a much higher affinity for CO than adult hemoglobin.
* For patients with a history of smoke inhalation, altered mental status, or soot in the mouth or mucous membranes, consider measuring blood cyanide levels to rule out CO poisoning complicated by cyanide poisoning (include lactate level, ABG, CO level and methemoglobin level). Cyanide toxicity is common when plastics are combusting. |
Treatment
|
For all patients
- The optimal benefit from HBOT occurs in those treated with the least delay between removal from CO source to treatment.
- Patients may require ventilatory support, blood pressure management, and ECG monitoring
- Soft restraints needed for unconscious patients.
- Institute/maintain intravenous fluid therapy, as indicated.
- If your facility cannot treat critically ill HBOT patients, consider referring to a nearby facility that has critical care support.
- If patient is unable to receive HBOT therapy, treat with 100% oxygen by non-rebreather. With administration of normobaric oxygen, the COHb half life is 74 ± 25 minutes.
- Repeat neuropsychometric screening following each HBOT treatment if possible/indicated.
For adult, non-pregnant patients: - Protocol 1: For patients with suspicion on neurologic or cardiologic involvement, repeated treatment should be considered, as it may yield a better outcome than a single treatment.
-
- Descend to 3.0 ATA with oxygen breathing for 60 minutes at pressure, then administer 5-minute air breaks at the 25 & 55 minutes of oxygen breathing. After 60 minutes at pressure, ascend to 2.0 ATA for additional 60 minutes of oxygen breathing. Administer a 5 minute air break after 25 minutes of oxygen breathing (See the Weaver protocol, table 4b).
- Repeat HBOT Treatments 2 & 3:
-
- Descend to 2.0 ATA oxygen for a total time of 90 minutes oxygen breathing, with two 5-minute air breaks. Treatments 2 & 3 should be given at 6 hour intervals. 2 additional treatments may be given where residual symptoms persist.
- Three treatments in 24 hours completes the Weaver protocol.
- Internal peer review required after 5 treatments.
- Protocol 2
- Initial compression to 2.8 ATA, then 2 ATA for 120 minutes, without further HBOT
For pregnant and pediatric patients
- Initial HBOT Treatment
-
- This encompasses the same treatment profile of the Weaver protocol. Internal peer review required after 5 treatments.
|
Follow-Up
|
- All discharged patients should be warned of possible delayed neurological complications and given discharge instructions on what to do if these occur.
- Up to 30% of patients with CO poisoning exhibit some degree of cognitive decline that appears up to 240 days after exposure, ranging from subtle impairments that are only detectable on neuropsychometric testing to frank dementia. Findings commonly observed include disorientation and deficits in attention, concentration, executive function, visual-spatial skills, verbal fluency, speed of information processing, and memory.[5]
- Patients receiving HBOT after developing delayed neurological complications have been shown to have a higher improvement rate (72.7%) compared to those who did not receive HBOT (25.5%).[6]
- Patients who received more than three sessions of HBOT during the acute stage of CO poisoning have been shown to have an even higher rate of improvement (81.8% vs. 27.5%)[6]
- If discharged to home:
-
- Educate patient regarding the need for assessment of the home for CO level and source. Do not return to the location until the source of poisoning has been corrected.
- In 24-hours: telephone follow-up
- In 6 weeks: repeat medical and neuropsychometric screening
|
Treatment Threshold
|
Utilization review after 3 treatments in 24 hours, after 5 total treatments and when exceeding 10 treatments.
|
Coding
|
Refer to the ICD-10 Guideline for the appropriate ICD-10 code
|
Comments
|
- Consider emergent needle myringotomies if patient unconscious, or obtunded and cannot manage their own airway or intubated.
- If diabetes mellitus is present, blood glucose should be checked within an hour prior to and immediately post-HBOT.
- Consider intubation for patient with oropharyngeal burns for airway management.
|
Primary Sources: Whelan and Kindwall [7]
, Weaver [8][9][10],
NBS [11],
Huang [12]
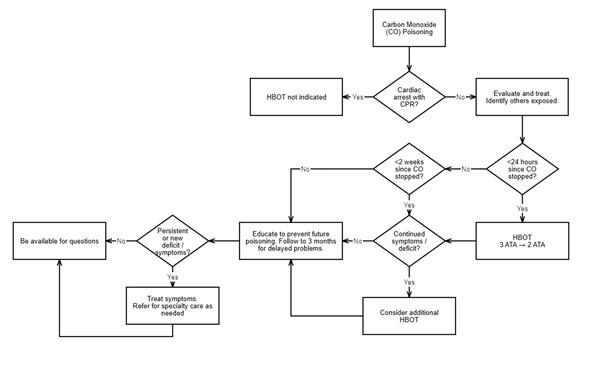
Figure 1. Flowchart for Carbon Monoxide Poisoning Treatment
Details of management are described in the text
History
- Document history of CO exposure (i.e., history of inhalation of CO or elevated CO levels) and symptoms. Diagnosis is clinical.
Below is a suggested history format for patients with acute carbon monoxide poisoning:
"Mrs. Jones was in her state of usual health until earlier today. She states that she felt 'unwell' this morning with vague symptoms of nausea, persistent frontal headache, and some spatial confusion/dizziness. She noted that when she went outside for a walk, she began to feel somewhat better. After returning home, she remembered hearing an alarm chirp early in the morning. She ignored it due to feeling unwell and noting that the batteries needed changing in the smoke alarms. As the day progressed, she felt worse and presented to the emergency room. Part of that workup was a venous carbon monoxide level of 22%.
She denies chest pain or discomfort. She states that she feels 'foggy' in thinking, and that has not gone away with 1 hour of high-flow O2 breathing in the emergency room. In addition, laboratory findings of pregnancy with an ultrasound confirming 6 month fetal development were noted. The hyperbaric medicine department was called to evaluate this patient based on the above.
We asked the fire department to assess her home environment for carbon monoxide. They found levels at the 400 ppm range. These levels were higher in her utility room."
[NOTE: Because she had a documented exposure to carbon monoxide, expressed symptoms linked to that exposure, and is pregnant, this is a person who should be treated with hyperbaric oxygen as soon as possible. Fetal hemoglobin has a higher affinity for carbon monoxide than adult hemoglobin, sometimes 10 - 15% higher.]
Physical Examination
- In addition to cursory system evaluation, physical examination should focus on neurological deficits. These should be noted clearly, and include slow mentation, difficulty with dates/times, cerebellar dysfunction, and others as required:
-
- Cerebellar dysfunction can be tested by performing heel-shin, finger-to-nose, Romberg, and/or rapid alternating movements.
- Cognitive dysfunction can quickly be determined by spelling "world" backward, and/or serial 7 test.
Laboratory Testing
Since the heart receives significant blood flow, we recommend that a rule-out myocardial infarction (MI) protocol be initiated while still in the emergency room. Small upticks in troponin levels with/without acute EKG changes are frequently seen. Of note, these quickly return to normal after hyperbaric oxygen therapy.
Impression
1. Acute Carbon Monoxide Poisoning Codes (ICD-10 T series)
Hyperbaric Plan
"We follow the evidence-based protocol demonstrated effective by Dr. Weaver in his October 3, 2002 New England Journal of Medicine article. The patient will be treated initially with a 3.0 ATA carbon monoxide treatment table. This will be followed by 2 - 2.4 ATA treatments, with 90 minutes of oxygen breathing during a 120 minute treatment. All 3 treatments should be completed in a 24 hour timespan."
Risk and Benefit of Hyperbaric Oxygen Therapy
- See risk and benefit statement in topic "Documentation HBO: Risks and Benefits"
Indication for Hyperbaric Oxygen Therapy (HBOT)
"The toxicity of carbon monoxide is based on a number of pathophysiological mechanisms. Carbon monoxide can cause hypoxic stress to tissues (e.g., heart or brain) leading to injury. The blood level of carbon monoxide has not correlated well with resultant neurological injuries. Administration of supplemental oxygen has long been the cornerstone of therapy for patients suffering carbon monoxide poisoning. Oxygen inhalation hastens the dissociation of CO from the hemoglobin molecule, as well as providing enhanced tissue oxygenation. Hyperbaric oxygen causes this dissociation to occur at a much greater rate than that achievable by breathing pure oxygen at ambient atmospheric pressure.
Several studies have shown that CO causes impaired leukocyte adhesion and injured microvasculature. This is thought to be secondary to the acute hypoxic state followed by a reperfusion injury. Alternatively, another theory suggests that CO initiates an inflammatory cascade resulting in permanent cell/tissue death. Benefits of hyperbaric oxygen include improved mitochondrial oxidative processes, inhibiting lipid peroxidation, and decreasing microvascular injury. The best clinical study to date has been performed by Lindell Weaver, MD, et. al. from LDS Hospital in Salt Lake City, Utah. Treatment of CO poisoned patients follows a 3 treatment protocol and has been shown to dramatically decrease post-exposure neurological deficits.
Sample Order
-
See Sample Physician Order
CLINICAL EVIDENCE AND RECOMMENDATIONS
Hyperbaric Oxygen Therapy for Acute Carbon Monoxide Poisoning
-
1BFor patients with acute CO poisoning who were removed from the CO source within 6 hours of exposure, we recommend HBOT (i.e., Weaver protocol) rather than normobaric oxygen therapy, in order to decrease neuropsychological sequelae (Grade 1B)
-
-
Rationale: CO poisoning is much more than a toxic gas ingestion. It is believed that CO poisoning potentiates central nervous system injury (CNS) through an unknown but presumably inflammatory lipid peroxidation pathway.[13][14] It is fair to assume that if CO poisoning were solely a matter of toxic gas ingestion, then the literature supporting the use of HBOT as quickly as possible after being removed from the poison gas source would make little scientific sense. Patients would be able to recover and be free of CNS sequelae with any oxygen breathing source (i.e., normobaric oxygen). However, the toxic gas belief only goes so far, and it does not explain a relatively high existence of permanent (or long-lasting) post-poisoning neurocognitive deficits. In the senior author's experience, many patients exhibit post-poisoning neurocognitive deficits, even when treated with appropriate HBOT as quickly as possible after being removed from the toxic environment. Moderate certainty evidence (evidence level B) supports primary HBOT for patients with fewer than 6 hours of CO exposure in order to reduce CNS sequelae.[15][16][13][13][17] Patients who received HBOT within 6 hours of CO exposure have been shown to have a better 6-month neurocognitive prognosis than those treated within 6-24 hours.[17] Use of HBOT in this context has been demonstrated to be cost-effective.[13] See 'Summary of Evidence' below. For protocol details, see 'Treatment Protocol' above.
-
Medicare coverage: Medicare covers HBOT as primary therapy for patients with acute CO poisoning.[1]
CODING
See specific ICD-10 coding for CO poisoning in "ICD-10 Coding"
APPENDIX
Summary of Evidence
(Back to text)
We reviewed the clinical guidelines, systematic reviews, meta-analyses and clinical trials summarized below. Applying the GRADE framework to the combined body of evidence, we found that:
- The recommendation to treat CO patients with HBOT to prevent neurocognitive deficits is supported by moderate certainty evidence (level B) derived from randomized controlled trials (RCTs) and from a recent, favorable meta-analysis.[15][16][13] RCTs whose results showed no difference in outcomes between patients treated with or without HBOT may lead to controversial recommendations.[18][19][20] However, careful analysis of these RCTs uncovers suboptimal experimental design that compromises certainty of results. For instance, authors recruited only "mildly impaired" patients, patients who had been removed from the CO source more than 6 hours prior to HBOT, and most significantly, used only a 2 ATA treatment table.[20] The latter goes against historical treatment profiles published by the US Air Force and US Navy. To prevent an outburst of the inflammatory cascade, inhibition of degranulation of WBCs and prevention of ischemia-reperfusion injury are needed. Full elucidation of this mechanism is beyond the scope of this topic, but suffice it to say that pressures in the 2.5 - 2.8 ATA range are required. Pressures less than this range will not work reliably. [13] Nevertheless, well-designed RCTs with favorable results provide enough evidence to support routine use HBOT in order to decrease neuropsychological sequelae post CO poisoning.
- Systematic reviews and meta-analyses:
- A 2018 favorable meta-analysis found benefit by treating CO patients with HBOT.[21] Both reduction of chronic neurocognitive defects and cost-effectiveness favored treatment with HBOT.
- Randomized controlled trials:
-
A 2011 RCT by Annane et al (n=385 patients) found no benefit in use of HBOT to decrease post CO poisoning CNS sequelae, possibly due to the fact that only 2 ATA oxygen breathing was used.[18]
- A 2002 double-blind RCT (n=152 patients) with an intention-to-treat that followed the CONSORT recommendations, used equal proportions randomization; sham control, and had specific and precise neuropsychological testing.[15] The RCT evaluated the effect of HBOT on cognitive sequelae due to CO poisoning, and concluded that 3 hyperbaric-oxygen treatments within a 24-hour period appeared to reduce the risk of cognitive sequelae 6 weeks and 12 months after CO poisoning. At a pre-determined point in the process, this study was terminated because the data clearly showed there were less neurocognitive deficits in the treatment group than the sham group. Unfortunately, the paper is now criticized and the conclusions minimized due to the early conclusion of this study. It is unfortunate, however continuing the study until completion would have been unthinkable.
- A 1999 RCT for this indication was performed by colleagues in Australia.[19] While the authors are respected in pulmonary, hyperbaric, and intensive care medicine, this study committed a number of errors that make its conclusions suspect. The randomization process used cluster randomization rather than individual; this was not an intention-to-treat study; follow up rate was less than 50%; the neuropsychological testing was vague and generalized, unable to sort out depression (high number of suicidal patients enrolled) and cognitive dysfunction; the control group was treated with 3 to 6 days of hospitalization on high-flow oxygen for the entire time (no study has ever done this); and there was no sham arm to the study. With this number of methodological errors, it is quite possible to reach an untenable, and unsupportable, conclusion.
-
A 1995 RCT by Ducasse et al (n=26) found statistically significant benefit in using HBOT to prevent post CO poisoning CNS sequelae in patients with less than 6 hours of CO exposure, treated with 2.5-2.8 ATA, compared with normobaric oxygen.[16]
-
A 1995 RCT by Thom et al (n=60 patients) found statistically significant benefit in using HBOT to prevent post CO poisoning CNS sequelae in patients with less than 6 hours of CO exposure, treated with 2.5-2.8 ATA, compared with normobaric oxygen. [13]
-
A 1989 RCT by Raphael et al (n=629) found no statistical benefit between CO poisoning patients treated with HBOT and with normobaric oxygen therapy. Most patients recruited for this study had been exposed to CO for more than 6 hours and were treated with only 2 ATA, factors that negatively affected results and conclusions of the study.[20]
Observational study
- A 2020 observational study by Nakajima et al. evaluated HBOT in adult patients with CO poisoning (n = 2034), comparing with standard care without HBOT (n = 4701). Authors found that while HBOT did not significantly reduce in-hospital mortality, it was significantly associated with improved consciousness levels and activities of daily living at discharge. This beneficial effect was observed even in patients with non-severe CO poisoning. [22]
CATEGORY A CONTINUING EDUCATION CREDIT
This topic has been reviewed and approved by the National Board of Diving and Hyperbaric Medical Technology (NBDHMT) for one (1) Category A Credit.
To claim the credit:
- 1. Read the topic
- 2. Answer the examination and course critique questions.
- Take the quiz via CliniPaths by Wound Reference
- To receive a certificate, a passing score of 70% is required
- 3. After passing, click the provided link to access your certificate. If prompted, sign into your WoundReference account.
For more information on Category A continuing education credits see blog post " Hyperbaric Certification and Continuing Education for Technicians & Nurses".
REVISION UPDATES
| Date | Comments |
| 10/31/24 | Expanded the 'Treatment Protocol' section |
| 1/08/20 | Added section 'Category A Continuing Education Credit'
|