ABSTRACT
Pressure injuries/ulcers (PUs/PIs) are localized damage to the skin and/or underlying tissue, usually over a bony prominences or related to a medical or other devices, resulting from prolonged pressure or pressure in combination with shear. The lesion can present below intact skin or as an open ulcer, which may be painful. Synonyms for this condition include, bedsores, decubitus ulcers, pressure sores and many more.[1]
Pressure ulcers/injuries (PUs/PIs) represent a large burden to healthcare systems. It is estimated that PUs/PIs cost the U.S. between $9.1-11.6 billion a year.[2] Despite optimal care, not all PUs/PIs are avoidable.[3][4][5] See more information on unavoidable PUs/PIs (e.g, Kennedy Terminal Ulcer).
For a clinical pathway on PU/PI assessment, see 'Algorithm for Assessment of PU/PI'
History: Patients with PUs/PIs need a comprehensive assessment and history including risk factors for development, recurrence, delayed healing and complications related to a PU/PI, comorbidities, medication, nutritional, functional and psychological status, activities of daily living, presence of depression, alcohol consumption, and smoking. Structured assessment with tools (e.g. Braden, Norton and Waterlow scales), associated with clinical judgement, are recommended.
Physical Examination: Patients with PU/PI often present with important comorbidities that warrant a comprehensive examination. Focused examination includes assessment of: the rectal/perineal/genital area for presence of incontinence associated dermatitis, peripheral blood supply if ulcer is on lower extremity, neurological findings (spasticity, motor strength, sensation, etc), musculoeskeletal deformities, cutaneous abnormalities, presence of blanchable erythema (which may precede PU/PI), the PU/PI itself, signs of infection, and complications.
Diagnosis: The diagnosis of PU/PI is clinical based on information gathered during history and physical examination. Laboratory tests ordered upon initial assessment help establish a baseline and monitor any chronic underlying medical conditions and nutritional status which may be factors that impair wound healing.[6][7] Diagnostic tests may be considered for investigation of non-healing ulcers, complications (e.g. osteomyelitis) and suspected deep tissue injury. Emerging technologies show promising initial results in early detection of PU/PI.
Differential diagnoses: Diabetic Foot Ulcer, arterial ulcer, Venous ulcers, inflammatory ulcer (e.g. pyoderma gangrenosum), erythema that blanches on compression, moisture-associated dermatitis, skin tears, tape burns, excoriations. See topic "Pressure Ulcers/Injuries - Is it Really a Stage 2?"
Classification systems of PUs/PIs: The NPIAP classification is the most widely adopted classification system for pressure ulcers/injuries (PUs/PIs)[8], and was updated in 2016.[9] In the U.S., CMS allows nursing homes to adopt the NPIAP guidelines in their clinical practice and nursing documentation. See most common PU/PI classification systems in “Pressure Ulcers/Injuries - Classification/Staging”.
Ulcer prognosis (healability) assessment: Aims to determine whether the ulcer can be healed with active conservative management alone (healable) or not (non-healable) or if co-existing medical conditions, drugs or circumstances will likely impede wound healing (maintenance). Determination of ulcer healability helps creation of an adequate treatment plan.[10][11]
Documentation: At each dressing change observe the PU/PI for signs of deterioration/improvement to determine if a change in treatment is required. It is also important to explore and document any barriers to adherence to the care plan, including patient's or caregiver's refusal of a given intervention. Proper documentation should be completed at least once a week and more often as needed; adequate documentation helps track wound healing progress. Signs of ulcer improvement need to be documented to support medical necessity (as per Medicare requirements). As part of a patient-centered approach, it is recommended that clinicians adopt patient-reported outcome measures to assess patient’s Quality of Life (QOL), pain, and depression. See summary of Medicare, NPIAP and American Medical Association (AMA) recommendations for adequate documentation.
Treatment: See "Pressure Ulcers/Injuries - Treatment"
Prevention and Patient Education: See topic "Pressure Ulcers/Injuries - Prevention" and topic "Pressure Ulcers/Injuries -Coordination of Care"
When to refer to specialists:
- Nutritionist at initial evaluation.
- Physical therapist/occupational therapist for seating/functional evaluation, for postoperative rehabilitation if surgery is considered, and for some of the adjunctive therapies (e.g, electrical stimulation, ultraviolet C light).
- Wound specialist if PU/PI does not show signs of improvement in 2 - 4 weeks.
- Vascular specialist if PU/PI is in lower extremity and peripheral artery disease is suspected.
- Respective specialists if associated metabolic, hematologic, autoimmune, oncologic diseases are suspected.
- Nurse specialized in continence for assessment and recommendations on how to manage incontinence.
- Social work for economic and social support.
- Psychologist/ mental health professional for depression and other psychological issues.
- Surgeon, plastic surgeon: full thickness ulcers that are extensive or refractory, if important structures are exposed (e.g., vessels, nerve, bone, muscle, fascia) or osteomyelitis is suspected.
- Hospice if the patient is certified as terminally ill (prognosis of 6 months or less) by their attending provider (if they have one) and the hospice provider.[12]
ICD-10 Coding: See ''ICD-10 Coding'
Clinical Guidelines and Quality Measures: See "Pressure Ulcers/Injuries - Overview"
|
Algorithm for Assessment of PU/PI
|
INTRODUCTION
Overview
This topic provides an introduction and framework for assessment of pressure ulcers/injuries, including epidemiology, risk factors, etiology, pathophysiology, history, physical examination, diagnosis, differential diagnoses, documentation and ICD-10 coding. For clinical guidelines and quality measures, see "Pressure Ulcers/Injuries - Overview". For common classification systems with pictures and descriptions, see "Pressure Ulcers/Injuries - Classification/Staging". For management of pressure ulcers/injuries see "Pressure Ulcers/Injuries - Treatment". For prevention strategies, see "Pressure Ulcers/Injuries - Prevention". For best practices in care coordination, see "Pressure Ulcers/Injuries -Coordination of Care".
Background
-
Definition: Pressure injuries/ulcers (PUs/PIs) are localized damage to the skin and/or underlying tissue, usually over a bony prominences or related to a medical or other devices, resulting from prolonged pressure or pressure in combination with shear. The lesion can present below intact skin or as an open ulcer, which may be painful. Synonyms for this condition include, bedsores, decubitus ulcers, pressure sores and many more.[1]
- In the latest version of the Mortality and Morbidity Statistics provided by the WHO (ICD-11), the official terms is “EH90 Pressure ulceration” including pressure injury, pressure ulcer, and bedsore.[1]
- Relevance:
-
- In the United States approximately 2.5 million patients are affected by pressure ulcers/injuries (PUs/PIs), and each year 60,000 of them die from these ulcers.[13]
- PUs/PIs represent the second-most common hospital billing claim. Among Medicare beneficiaries, a PU/PI adds up to US$43,000 to each hospital stay.[13] Hospital length-of-stay is on average 20 days longer for patients with PU/PI.[14] It is estimated that PU/PI cost the U.S. between US$9.1-11.6 billion a year.[2] PU/PI treatment products (including high cost advanced wound care products), comprise a large part of these costs and was estimated to be about US$1.7 billion in North America in 2017.[15]
- PU/PI has been identified by the Centers for Medicare and Medicaid Services (CMS) as one of the 14 hospital-acquired conditions (HACs) that are reasonably preventable using evidence-based guidelines.[16] Along the many recent CMS “value-based purchasing” initiatives, an effort that ties payment to performance, in 2008 CMS decided not to reimburse hospitals for Stage 3 and 4 PU/PI and other preventable HACs.[17]
- Medicare, the National Pressure Injury Advisory Panel (NPIAP), and the Wound, Ostomy and Continence Nurses Society (WOCN) recognize that despite optimal care, not all PUs/PIs are avoidable [3][4][5][18]
-
- Avoidable PU/PI: is defined as a situation in which the person receiving care developed a PU/PI and the provider of care did not do one of the following: evaluate the person’s clinical condition and pressure ulcer risk factors; defined and implemented interventions that are consistent with individual needs, goals and professional/recognized standards of practice; monitor and evaluate the impact of the interventions; or revise the interventions as appropriate.
- Unavoidable PU/PI: means that the person receiving care developed a PU/PI even though the provider of care had evaluated the person's clinical condition and risk factors; defined and implemented interventions that are consistent with individual needs, goals, and professional/recognized standards of practice; monitored and evaluated the impact of the interventions; and revised the approaches as appropriate. Of note, adequate documentation of all aforementioned preventative measures is imperative for a PU/PI to be considered unavoidable.[18]
-
-
Table 1 displays consensus statements published by the NPIAP with examples of situations in which PU/PI may be unavoidable.[19]
- Examples of other unavoidable lesions at the end-of-life include:
-
-
Kennedy terminal ulcer (KTU)[20][21]: described as lesions that present as pear, butterfly or horseshoe-shaped red/yellow/black ulcer, often on the sacrum or coccyx, that may occur suddenly (Figures 1 and 2). Patients with these wounds sometimes die in a matter of hours and sometimes live for more than 6 weeks after presentation of the ulcer.[22] The literature is unclear as to whether KTU should be considered a PU/PI or a separate skin problem that also occurs over a bony prominence.[23] At least for long term facilities, Medicare considers KTUs as unavoidable "pressure ulcers that generally occur at the end-of-life".[5]
-
Trombley-Brennan terminal tissue injury [24]: A purple maroon discoloration that may appear suddenly on the body of a patient at the end of life. The patient will exhibit these skin changes on bony and non-bony prominences. Although, these injuries will never evolve into full thickness wounds with non viable tissue, they are frequently characterized by an increase in surface area. No drainage will be noted and linear and mirror images may appear on lower extremities. There are no complaints of discomfort with these areas.
- Skin changes at life's end (SCALE): the SCALE concept represents the loss of skin integrity in patients at the end-of-life from any of a number of factors, including but not limited to equipment or devices, incontinence, chemical irritants, chronic exposure to body fluids, skin tears, pressure, shear, friction, and/or infections.[23] SCALE may be due to an unavoidable PU/PI.
- Of note, the term "skin failure" is used as unifying term for skin changes at the end-of-life such as KTU, SCALE and Trombley-Brennan terminal tissue injuries, and for other acute states that compromise skin integrity.[23][25] For details on how to differentiate among PU/PI and skin failure, and other conditions, see topic "Skin Failure - Introduction and Assessment".
- Despite the establishment and implementation of prevention guidelines, avoidable PUs/PIs continue to occur.[13] One of the likely reasons stems from the fact that PU/PI management is hampered by wide variations in clinical conditions under which patients are hospitalized and receive care. Clinicians often stage other types of wounds such as incontinence associated dermatitis as PU/PI, and may use different interpretations of classification staging, preventing accurate clinical staging and reliability among observers.
|
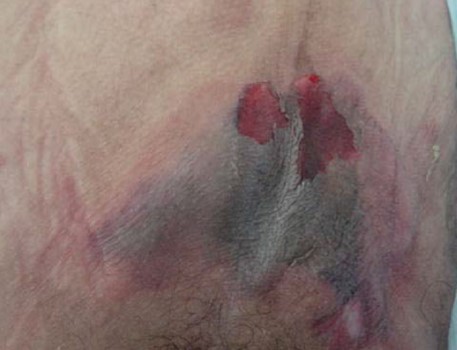 Fig. 1. Kennedy Terminal Ulcer - horseshoe-shaped Fig. 1. Kennedy Terminal Ulcer - horseshoe-shaped
|
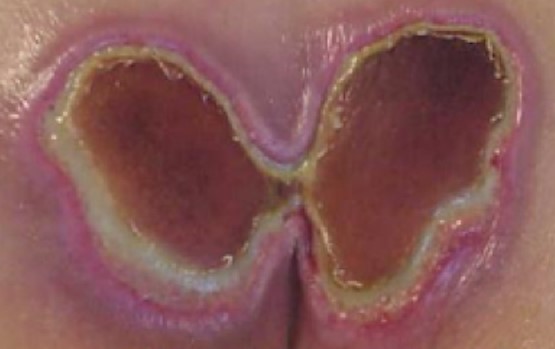 Fig. 2. Kennedy Terminal Ulcer - butterfly-shaped Fig. 2. Kennedy Terminal Ulcer - butterfly-shaped
|
Table 1. NPIAP consensus statements on increased likelihood of unavoidable PUs/PIs[19]
- When an individual’s cardiopulmonary status is significantly altered and recovery to baseline does not occur within minutes
- When an individual is repositioned and alterations in hemodynamic stability require ongoing vasopressor support
- When sustained head-of-bed of greater than 30° elevation is medically necessary
- Septic shock and/or systemic inflammatory response syndrome
- Extensive body edema
- Severe burn injury
- In hemodynamically unstable or critically ill/critically injured individuals, when management of life-threatening conditions must take precedence over skin-preservation interventions
- When life-sustaining, vascular access, or other medical devices preclude turning and/or repositioning
- An unstable pelvic fracture or spinal cord injury that precludes turning
- Terminally ill individuals who become immobile
- Individuals with malnutrition in combination with multiple comorbidities
- Individuals with cachexia
- When it is medically contraindicated to adjust, relocate or pad underneath a therapeutic medical device
- In patients with COVID-19: when anatomical locations are subjected to pressure and/or shear stress and:
- Microvascular occlusions from COVID-19 increase the magnitude and severity of non-modifiable risks (e.g. need for vasopressors, medical devices) to a level that preventive interventions are not able to mitigate, despite reasonable efforts at prevention;
- Multi-organ dysfunction from critical illness ultimately impair cutaneous function and integrity
- All efforts to provide evidence-based preventative care are attempted, within the context of a healthcare system that may be at crisis capacity
|
Epidemiology
Incidence:
Hospital discharge data from 210 academic medical centers in the USA show decreased incidence rates of PU/PI from
11.8/1000 cases in 2008 to 0.8/1000 cases in 2012.[14][26]
Prevalence
Data from a sample of 918,621 patients in the USA showed that the overall prevalence of PU/PI at all facilities decreased from 13.5% (2006) to 9.3% (2015). Overall prevalence decreased at each type of care setting as well, and is shown below [26]:
- Acute care: from 13.3% (2006) to 8.8% (2015)
- Long-term acute care (LTAC): from 32.9% (2006) to 28.8% (2015)
- Long term care (LTC) (e.g, skilled nursing facilities): from 12.1% (2006) to 11.3% (2015)
- Rehabilitation facilities: from 16.3% (2006) to 11.0% (2015)
However, in general the prevalence of PU/PI continues to be higher among patients receiving palliative care than in the general population. A systematic review with 63,118 patients from different countries, different types of care settings and different data collection periods, found that the overall prevalence was 12.4%.[27] When analyzing prevalence by care settings, authors found the following differences:
- Acute care: 13.8%–19%
- Long term care (LTC) (e.g, skilled nursing facilities): prevalence was 6.9%–16.2% (depending on whether patients had short or long stays)
- Home health: 10.2%–11%
Among patients with COVID-19 requiring intensive care, the prevalence of PU/PI has been shown to be three times greater than that among patients without COVID-19 who also needed intensive care.[28][29]
- Among critically ill COVID-19 patients admitted to an intensive care unit in China, 46 out of 109 patients (42.2%) developed PU/PI despite preventative measures. The median interval from intubation to the presence of pressure injury was 8 days.[30]
- International data suggest that up to 57% of patients with COVID-19 ventilated in the prone position develop a PU/PI. Ventilation in the prone position is indicated for patients with severe acute respiratory distress syndrome. Up to 28% of patients admitted to the ICU with confirmed infection due to severe COVID-19 are cared for in the prone position.[31]
Among hospitalized children, the pooled prevalence of PU/PI for 4,639 children has been shown to be 12.2%.[32]
Risk Factors
Risk factors for incidence:
The two major risk factors in the development of pressure ulcer/injury are magnitude and time of applied mechanical loads and their mode of action (compression/shear) and the tolerance of the individual.[33]
By co-morbidity
Specific patient populations are more prone to developing PU/PI [34]
- Patients with hip fractures (8.8 - 55% develop PU/PI) [35][36]
-
- Increased risk associated with age older than 81 years and placing the limb in a foam rubber splint. [37]
- Patients with neurological conditions (acquired or congenital) that affect mobility:
-
- Spinal cord injuries (33 - 95% develop PU/PI) [38][39]
-
- Risk factors for patients with spinal cord injuries include decreased sensation, immobility, urinary and bowel incontinence, a history of previous pressure injury, and tobacco use.[34]
- Spina bifida (19% develop PU/PI) [40]
-
- Risk factors for patients with spina bifida include wheelchair use, urinary incontinence, shunt presence, above the knee orthopedic surgery, recent surgery, and male sex.[40]
- Elderly patients with immobility and/or cachexia (muscle wasting).[34]
- Lower extremity trauma resulting in bone or soft-tissue injury and subsequent fixation with casting: associated with PU/PI under the cast or on the heels.[34]
- Patients with COVID-19 requiring intensive care [28][29], especially those ventilated in the prone position.[31]
By anatomical location
- Generally, sacrum is often the most common site (28 - 36% of PUs/PIs), followed by the heel (23 - 30% of PUs/PIs) and the ischium (17 - 20%).[34]
- Among patients with COVID-19 requiring intensive care, a study (n=46 patients) reported that the sacrum (89.1%) was the most common location, followed by the face (23.9%) due to the prone position ventilation adopted to optimize oxygenation, heels (21.7%) and hips (8.7%).[30]
- Among patients with spinal cord injuries, location depends on the phases following spinal injury. For instance, in the acute post injury phase patients may be kept in a supine position for long periods of time for stabilization of the spinal shock and the spine. Thus, areas such as sacrum, ischium, scalp, spinous process and elbow may be more affected. Once patients are back to their lives in their communities and are sitting in wheelchairs, PUs/PIs in the sacral, ischial, heel, trochanteric and elbow areas are most common.[41][6]
- Among hospitalized children, the most affected body sites have been shown to be the occiput, ears, and nose.[32]
By care setting
-
Long term care (LTC)(e.g., skilled nursing facilities): Stage 2-4 PUs/PIs are significantly associated with Stage 1 non-blanchable erythema [42][43], a lower Braden score, and pain in the pressure area.[43] In LTCs, 80 percent of PU/PIs develop within two weeks of admission and 96 percent develop within three weeks of admission.[44]
-
Surgical departments (inpatient hospital): Significant risk factors include: age = 75 years, female gender, ASA = 3 (physical status classification system by the American Society of Anesthesiologists to assess the fitness of patients before surgery, a healthy patient is ASA 1), body mass index < 23, preoperative Braden score = 14, anemia, respiratory disease, and hypertension.[45][46] As for surgery duration, it has been reported that surgeries longer than 4 hours [47] and 6:15 hours [48]on a standard OR table increase risk of PU/PI.
-
Critical care units (inpatient hospital): Significant risk factors include older age, altered mobility, poor perfusion, or use of vasopressor.[49]
-
Inpatients hospital, overall: strongest risk factors have been reported to be prior PU/PI, prior diabetic foot ulcer, and malnutrition.[50]
Risk assessment tools:
-
1CClinical guidelines recommend performing a PU/PI risk assessment upon the patient's admission to a healthcare setting, and on a regularly scheduled basis to identify individuals at risk of developing a PU/PI (Grade 1C).[9][51][52]
-
-
Rationale: Currently there is no reliable evidence to suggest that the use of structured PU/PI risk assessment tools reduces the incidence of pressure ulcers compared to clinical judgement alone.[53][54] However, due to the burden and impact of PU/PI development on both the individual and the health service, and the potential differences in clinical judgement due to gaps in healthcare professionals' knowledge of wound care and PU/PI[55], clinical guidelines [9][51][52] recommend use of structured risk assessment tools to identify those at potential risk so that individualized preventive interventions can be planned and initiated.[9] Use of structured risk assessment tools are also relevant for documentation standardization and medical legal purposes.
- To improve documentation and care coordination, clinicians at each institution should receive training and utilize the same validated risk assessment tool.
-
Validated risk assessment scales for adults include: Braden, Waterlow, and Norton[56]:
-
-
Braden Scale for Predicting Pressure Sore Risk: is the most commonly used in North America and has 6 subscales (sensory/perception, moisture, activity, mobility, nutrition, and friction/shear) [57]
-
Waterlow Scale: most frequently used scale in the U.K.[58][59], has 9 subscales: (BMI, continence, skin type, mobility, appetite, tissue malnutrition, neurological deficit, major surgery/trauma, and medications).[58] The higher the score, the higher the risk of developing PU/PI.
-
The Norton Scale was developed in the 1960's and has 5 subscales (physical condition, mental condition, activity, mobility, and incontinence).[60] Total score ranges from 5-20. A lower Norton score indicates higher levels of risk for PU/PI development. Generally, a score of 14 or less indicates at-risk status.
- Validated tools for children include:
- Braden Q Scale is a validated, adjusted version for children under 8 years of age [61]
- Braden QD scale: revised, simplified version is the "Braden QD Scale", for immobility-related and device-related pressure injuries in the pediatric acute care environment.[62]
- For more details, see section 'Structured Risk and Skin Assessment' in topic "Pressure Ulcers/Injuries - Prevention".
-
Etiology
According to the NPIAP, PU/PI occurs as a result of intense and/or prolonged pressure or pressure in combination with shear. The tolerance of soft tissue for pressure and shear may also be affected by microclimate, nutrition, perfusion, co-morbidities and condition of the soft tissue.[9]
-
Pressure: is defined as the “force per unit area exerted perpendicular to the skin surface.” [56][63]
-
Shear: is the “force per unit area exerted in parallel to the skin surface.”[56][63]
- Friction: is the term used to describe all phenomena related to the interface properties of surfaces sliding with respect to each other.[33]
- Since 2009, the NPIAP has disregarded "friction" as a direct factor in PU/PI development. Instead, NPIAP notes that friction is rather one of the causes of shear strain in the tissue, which in turn increases risk of tissue breakdown and lead to PU/PI.[64]
Pathophysiology
- According to a recent PU/PI conceptual framework, PUs/PIs develop as a result of an imbalance between an individual's tolerance to external mechanical loads and mechanical loads that exceed this tolerance.[63][65]
-
- An individual's ability to tolerate mechanical loads is linked to factors such as morphology and mechanical properties of the affected tissue. These, in turn, are affected by patients' characteristics, health status and exposure to specific risk factors (i.e, skin status, poor perfusion, poor nutrition, diabetes, moisture).
-
- PUs/PIs occur over predictable pressure points where bony protuberances are more likely to compress tissues when the patient is in contact with hard surfaces.[65]
- External mechanical loads causing PU/PI present in the form of pressure or shear, with variable duration and intensity. Risk factors that affect loading exposure include immobility and poor sensory perception/response.
- When the damage threshold of an individual is reached, a series of events occur and result in tissue damage[63]:
-
-
Tissue deformation: may cause immediate muscle damage in susceptible areas/individuals.[66][67] Figure 3 illustrates the PU/PI damage cascade that includes the sequential damage associated with direct deformation, inflammatory response, and ischemia.[68] Over short periods of loading exposure, the level of deformation is the most important factor in the damage process for muscle tissue, while over prolonged exposure periods, ischemia and reperfusion gradually become the dominant factor.[69][65]
-
- The exact length of time needed for a PU/PI to develop depends on the loading exposure and the individual's tolerance to it. Cell damage due to direct deformation damage to cells can happen in the order of tens of minutes.[66][67]
-
Inflammatory response-related damage: cell death due to tissue deformation leads to local inflammatory reactions with edema and increased interstitial pressure. This additional tissue deformation leads to more cell distortion and cellular damage.[68]
-
Localized ischemia/ hypoxia: classically thought of as the dominant etiological factor in PU/PI development. External mechanical loads cause blood vessels to compress and nutrients cannot be delivered, leading to local hypoxia and tissue damage.[63] To decrease local blood flow, the applied external mechanical pressure must be higher than the normal arterial capillary pressure of 32 mmHg. Duration of external pressure also contributes towards local changes resulting in tissue ischemia and necrosis.[6]
-
Reperfusion injury: as local pressure is relieved, blood vessels become patent again and blood is delivered to previously ischemic tissues. Reperfusion leads to release of harmful oxygen free radicals further damaging local tissues.[70]
-
Impaired lymphatic drainage: external mechanical loads occlude lymph vessels as well as blood vessels, resulting in an accumulation of waste products, contributing to PU/PI development. [71]
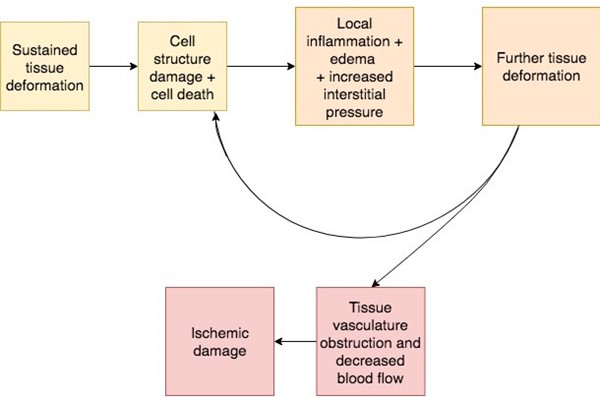
Fig. 3. PU/PI damage cascade with direct deformation, inflammatory response, and ischemia [68]
- Among patients with severe COVID-19: cytokine storm, metabolic acidosis and micro-thrombosis caused by COVID-19 may not only contribute to the risk of PU/PIs by augmenting or accelerating the PU/PI damage at the different phases of its evolution (Figure 3), but can also induce overall pain. This pain caused by COVID-19 may mask a localized discomfort or pain sensation caused by excessive pressure and prevent timely diagnosis of a new PU/PI.[28]
ASSESSMENT
This section focuses on PU/PI-specific elements that need to be included in a comprehensive assessment of patients with PU/PI, based on recommendations from the NPIAP [33], Wound and Ostomy Continence Nurses (WOCN)[51] and other relevant professional associations. For risk assessment of patients without an active PU/PI, see topic on "Pressure Ulcers/Injuries - Prevention".
- Patients with PUs/PIs need a comprehensive initial assessment to guide development of the most appropriate management plan and monitor wound healing progress. The initial assessment should include a complete history and physical examination to assess factors that affect healing, vascular supply if ulcer is in lower extremity, nutrition, pain, risk for development of new ulcers, psychological condition, social and financial support, functional capacity, ability to adhere to prevention/treatment plans.[33]
- Primary goals of assessment are:
-
- To identify underlying causes of the PU/PI
- To identify risk factors for development of PU/PI or that may impede healing (if healing is a realistic goal)
- To identify pertinent patient's/caregivers' concerns
- To rule out other types of ulcers, which may require different treatments. See 'Differential Diagnoses'
- To classify the PU/PI
- To determine healability, that is, if the PU/PI has potential to heal with conservative interventions only. See 'Ulcer Healability'.
- See Algorithm for Assessment of Pressure Ulcers/Injuries below (Algorithm 1)
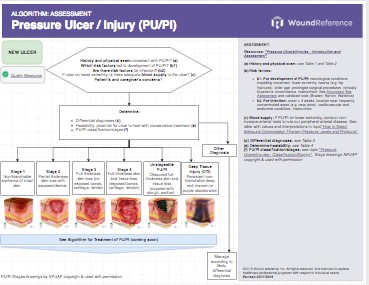
Algorithm 1. Algorithm for Assessment of Pressure Ulcers/Injuries (click this link to enlarge)
History
The non-exhaustive checklist below highlights relevant factors that should be assessed when taking history of a patient with a PU/PI. Risk factors for development of PU/PI are indicated with the icon , risk factors for infection are indicated with
Chief Complaint and History of Present Illness
- Setting in which the patient developed PU/PI (inpatient, outpatient, etc): certain risk factors are more relevant in specific care settings.[51] See 'Risk Factors' above. Unavoidable PUs/PIs are highly prevalent (~30%) among residents of specialized inpatient palliative care units upon death.[23]
- Age, gender: female gender, age greater than 75 years old [45][46]
- Onset of ulcer: if present for more than 4 weeks, higher risk for infection [33] Unavoidable PU/PI at the end-of-life are observed in persons in the last hours/days/weeks of their life.[23]
- Location of ulcer:
- Prolonged pressure points : for instance, patients who sit for too long in one position may be more prone to a PU/PI over the ischial tuberosity. Slouching in a chair may predispose an at-risk individual to PU/PI of the spine, scapula or elbow.[5] Prolonged pressure can also be caused by medical devices [5], including nasogastric or nasoenteric tubes pressing against the nasal vestibule, exterior nose and nasal mucosa.[72] See section 'Preventative Measures for Medical Device-related PU/PI' in topic "Pressure Ulcers/Injuries - Prevention""
- Some areas are likely to be repetitively contaminated (e.g., near the anus) [33]
- Pain: can indicate infection and/or ischemia [33]
- Presence of other PU/PIs [33]
- History of previous of PU/PI [50][33]
- Prior treatment/prevention:
-
- Individual's functional capacity, particularly in regard to repositioning, posture and the need for assistive equipment and personnel.[33]
- Previous employment of pressure relieving and redistributing maneuvers/devices.[33]
-
- Type and manufacturer, date of purchase, fit, usage patterns
- Past surgical history related to the PU/PI (e.g., debridement, flaps, etc) and complications
Medications
- Medications that impair wound healing include anticoagulants, antimicrobials (various antibiotic classes), anti-angiogenesis agents (eg, bevacizumab, aflibercept), antineoplastic drugs, anti-rheumatoid drugs (eg, methotrexate, aspirin/nonsteroidal anti-inflammatory drugs [NSAIDs]), colchicine (anti-gout drug), topical hydrogen peroxide, topical iodine, full-strength 0.5% Dakin’s solution (sodium hypochlorite), nicotine, steroids, and vasoconstrictors.[73][74][75]
Social History
- Smoking, drugs, alcohol may directly or indirectly impair wound healing [73][34]
- Social and caregivers' support, support at home
Review of Systems
- General:
-
- Nutrition status: malnutrition and hydration deficits [5][33]
- Fever and malaise may indicate PU/PI infection
- Anemia [45][46]
- Cachexia [34]
- History of cancer
- Immunosuppression: auto-immune disease, immunosuppressants [33]
- HEENT: visual field deficits can impair safe self-mobility for individuals with neurological deficits
- Cardiovascular: hypertension/hypotension, hemodynamic instability, poor diffuse or localized blood perfusion (e.g. generalized atherosclerosis or lower extremity arterial insufficiency), or use of vasopressor infusion [5][33][45][46][46][49]
- Endocrine: thyroid disease or diabetes mellitus [5][33]
- Gastro-intestinal/Genito-urinary
-
- Fecal/urine incontinence : both urine and feces irritate skin and make it more susceptible to breakdown and moisture-related skin damage[5]
-
- Frequency of evacuation, use of containment/collection supplies (e.g., incontinence underwear, etc)
- Use of urine collection devices (e.g, catheters)
- Diarrhea or constipation, frequency
- Urinary tract infections
- End-stage renal disease [5]
- Neurological:
-
- Cognitive impairment [5]
-
Spinal cord injuries [38], any other neurological conditions such as epilepsy, brain tumor, muscular dystrophy, spina bifida, etc. Note date of onset, level of of spinal cord damage and resulting neurological changes (sensory, autonomic, motor)
- Musculoskeletal:
-
- Immobility [33][50]
- Hip fractures [35][36]
- Skin: assess use of topical medication, creams and ointments, and presence of rashes and other dermatological conditions
Structured Risk Assessment
-
1CClinical guidelines recommend performing a PU/PI risk assessment upon the patient's admission to a healthcare setting, and on a regularly scheduled basis (Grade 1C).[9][51][52]
-
In the United States, the most used structured risk assessment tool for people over 8 years of age is the “Braden Scale for Predicting Pressure Sore Risk” tool.[57][56] A specific version for home care exists: "Braden Scale for Predicting Pressure Sore Risk in Home Care". The tool has 6 categories, namely: Sensory perception, Moisture, Activity, Mobility, Nutrition, Friction/shear. To use the tool:
-
- Assess each of the 6 categories and select the description for each category that best describes the person's condition
- Assign a score for each of the 6 subscales from 1 to 4 according to descriptions on the tool (except for friction/shear, which is scored from 1 to 3)
- Calculate the total score, which will be between 6 and 23 points. The lower the score, the greater the risk for skin breakdown. Patients scoring 18 or less are considered to be at risk.
-
- At risk: 15 – 18
- Moderate risk: 13 – 14
- High risk: 10 – 12
- Very high risk: 9 or less
- Risk factors not captured by the Braden Scale, for instance advanced age, prolonged intensive care unit (ICU) stay, hemodynamic instability, and others can increase risk for PU/PI development. When those risks are detected, they should be indicated on the medical records.
- For details and clinical examples see section 'Structured Risk Assessment' in topic "Pressure Ulcers/Injuries - Prevention".
Patient's and caregivers' concerns
Patient's and caregiver's concerns and psychosocial aspects should be assessed and taken in consideration when creating a treatment/prevention plan. Concerns and psychosocial aspects include but are not limited to:
- Values and goals of care of the individual and/or the individual’s significant others.[33]
- Psychological health, behavior, and cognition.[33]
- Pain related to PU/PI.[33]
-
Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"- To assess pain, validated pain scales are recommended (see 'Documentation' below), but they may not capture sufficient information to guide intervention. An initial pain assessment should include the following four elements [33]:
-
- A detailed pain history including the character, intensity, triggering factors and duration of pressure ulcer pain (incorporate the individual's body language, non-verbal cues and words that describe pain)
- A physical examination that includes a neurological component;
- A psychosocial assessment; and
- An appropriate diagnostic work-up to determine the type and cause of the pain.
- Social and financial support systems [33]
-
- Resources available to the individual (e.g. pressure redistribution support surfaces)
- Ability to adhere to a prevention and management plan [33]
-
- Knowledge and belief about prevention and treatment of PUs/PIs [33]
- Resident refusal of some aspects of care and treatment [5]
We recommend use of Patient-Reported Outcome Tools to assess aspects above and measure impact of interventions. See 'Patient-reported outcomes (PRO) tools' below.
Nutritional Screening and Assessment
- 1BFor all individuals at risk of a PU/PI, clinical guidelines recommend that clinicians conduct a nutritional screening using a validated tool (Grade 1B).[33][76]
- Rationale: evidence shows that individuals identified as malnourished or at risk for malnutrition through nutritional screening are more likely to be at risk of developing a PU/PI. [33][77][78] Studies suggest that nutritional screening leads to early implementation of nutritional interventions, which may translate into up to 50% reduction in PU/PI rates, decreased length of hospital stay, and decreased healthcare costs.[79]
- Nutritional screenings may be completed by any qualified member of the health care team upon admission to the facility, or at first visit in community settings.[33] Guidelines suggest re-screening patients if the ulcer fails to show signs of progress, or whenever there is a change in the individual's condition.[33]
- Commonly used screening tools to assess risk for malnutrition in adults include the Nestlé MNA ® [80], MUST [80], NRS [81] and SNAQ [82], SCREEN© [83] and CNST [84], all of which have been explored in validation studies.[33]
- Medicare Quality Payment Program, Quality Measure:
- "Process Measure: Nutritional Screening and Intervention Plan in Patients with Chronic Wounds and Ulcers"
- "Patient Reported Nutritional Assessment and Intervention Plan in Patients with Wounds and Ulcers"
- "Preventative Care and Screening: Body Mass Index (BMI) Screening and Follow-Up"
- 1CFor individuals at risk of a PU/PI who are screened to be at risk of malnutrition, and for all adults with a PU/PI, clinical guidelines recommend that a comprehensive nutrition assessment be conducted by a registered dietitian/nutritionist in collaboration with an interprofessional nutrition team (Grade 1C).[33][51][51][85]
- A history of food intake, appetite and unintended weight change should be determined. For food intake, a complete food log covering 2-3 days is better than an incomplete one covering a longer period of time. It is important that a registered dietitian be involved in the evaluation.[6] See section 'Nutritional Assessment' in topic "How to Screen, Assess and Manage Nutrition in Patients with Wounds".
- To identify malnutrition, the Academy of Nutrition and Dietetics and the American Society for Parenteral and Enteral Nutrition recommend presence of two or more of these six parameters [86][85]:
- Insufficient energy intake
- Weight loss
- Loss of muscle mass
- Loss of subcutaneous fat
- Localized or generalized fluid accumulation that may mask weight loss
- Decreased functional status measured by hand grip
- Laboratory tests for nutritional assessment: The NPUIP recommends against relying solely on laboratory tests such as albumin or pre-albumin as indicators as malnutrition.[33][87] These laboratory tests may be altered by factors such as hydration, presence of infection, acute stress and, thus should be considered in addition to other assessment parameters.[51][85]
- Other laboratory values may be helpful in determining if malnutrition is related to starvation, chronic disease or acute disease/injury include: HbA1c, blood glucose, complete blood count, C-reactive protein[85][88] See section 'Biochemical Data, Medical Tests, and Procedures' in topic "How to Screen, Assess and Manage Nutrition in Patients with Wounds".
Functional, Equipment and Seating Evaluations
Functional, equipment and seating evaluations are important for individuals with PU/PI in order to identify and address factors contributing towards development of PU/PI and delayed healing. Upon initial assessment, the following should be recorded:
- Level of activity in the last 24h and need for assistance (number of transfers/day, number of hours/day in bed or sitting)
- Need for mobility aids or caregiver's assistance in ambulating, moving in bed/chair, transfer from one surface to another, sitting, etc
- Posture, joint function, neurological conditions, bowel and bladder control
- Pressure redistribution/offloading devices in use (bed, chair, wheelchair, cushions - type, manufacturer, date purchased)
Physical Therapy/ Occupational Therapy Evaluation
Physical therapists (PT) or occupational therapists (OT) conduct comprehensive functional, equipment and seating evaluations. Therapists' usual practice for patients with PU/PI include determining causation of the PU/PI (by performing evaluations related to seating/positioning/patient transfer techniques, etc), conducting direct wound care, teaching muscle strengthening exercises and educating family and caregivers on repositioning.[89][6]
As part of the evaluation, therapists typically conduct inspection of support surfaces and pressure redistribution devices, including assessment of:
-
Adequate use, wear and tear of support surfaces, pressure redistribution devices: effectiveness of a device can be made through careful assessment of skin over the weight-bearing bony prominences.[90]
-
Patient's clothing material and fit: may not stretch enough to allow support surfaces to be effective.[90]
-
Interface pressure measurement: interface pressure, that is, the pressure between the support surface (seat or mattress) and the skin,[90] is often assessed subjectively using "hand checks", but there is no evidence to support this method.[91] Instead, clinicians may opt to use computerized sensors to objectively measure interface pressure.[90]
-
-
Hand check: clinician slides a hand, palm side up, at the interface of the overlay and the mattress just under the patient’s sacrum. If the clinician can feel the patient’s sacrum resting in the palm of their hand or is not able to feel at least 1” of the inflated surface between the palm of the hand and the patient’s sacrum, the patient is said to be “bottoming out”, and the support surface needs to be replaced.[91]
-
Computerized sensors for interface pressure measurement (IPM) (e.g.,VU, CONFORMat™) : can be handheld, single pad sensors or larger pressure-mapping devices. IPM is recorded at all bony prominences in all positions for each patient, and serves to evaluate the IPM of bony prominences of the same person on different surfaces, and to help clinicians choose the safest surfaces for the individual.[90]
Physical Examination
A focused physical examination that includes the following is recommended [33]:
Rectum/Genitalia/Pelvic:
- Incontinence associated dermatitis (IAD): may be difficult to differentiate from partial thickness PU/PI. IAD commonly occurs in the area where incontinence brief or underpad has been used [5] (Figures 4 and 5) Consultation with nurse specialized in continence for assessment and manage of incontinence is recommended.
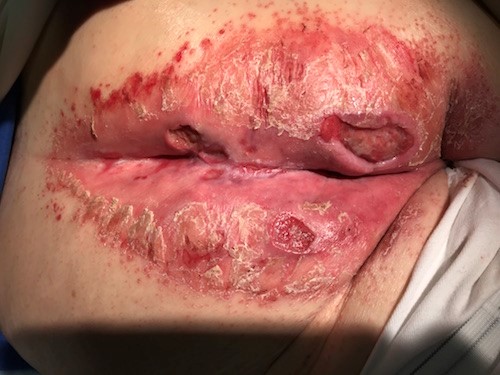 Fig. 4. Incontinence associated dermatitis and stage 2 PUs/PIs in the gluteal region Fig. 4. Incontinence associated dermatitis and stage 2 PUs/PIs in the gluteal region
|
 Fig. 5. Incontinence associated dermatitis treated with waterproof barrier film, stage 2 PUs/PIs also showing signs of improvement Fig. 5. Incontinence associated dermatitis treated with waterproof barrier film, stage 2 PUs/PIs also showing signs of improvement
|
Extremities, including pulses:
- If ulcer on lower extremity: check skin color, cyanosis, temperature, peripheral pulses, capillary refill time. Non-invasive arterial testing (e.g. ankle-brachial index or toe pressure) is recommended to rule out associated peripheral arterial disease as an etiology.[33] See table with values and interpretations of non-invasive arterial tests in section 'Noninvasive arterial tests' in topic "Arterial Ulcers - Introduction and Assessment".
Neurologic:
- Mental Status
- Motor strength, pathological reflexes and spasticity
- Sensation (light touch, pin prick, vibration and position)
- Cerebellar function, observed ambulation
- Bowel and bladder control
Musculoskeletal
- Musculoskeletal deformities, skin contractures: may prevent adequate positioning, limit mobility and result in pressure points where PU/PI may develop
Skin
- Comprehensive, head-to-toe skin assessment. See section 'Comprehensive Skin Assessment' in topic "Pressure Ulcers/Injuries - Prevention".
- Local edema [19] or lymphedema may increase risk for PU/PI [65]
- Scars, erythema, ecchymosis, dermatitis
- Thin friable skin with poor turgor: vulnerable to skin tears and PU/PI [90]
- Signs of abuse or neglect [92]
Ulcer/injury exam
Validated wound assessment tools may be used:
- Pressure Ulcer Scale for Healing (PUSH) Tool [93]
-
Bates-Jensen Wound Assessment Tool [94]
The following should be assessed:
- Ulcer location, and category/stage. See section 'Classification/Staging' and topic " Pressure Ulcers/Injuries - Classification/Staging"
- Presence of erythema: if erythema is present, differentiate between blanchable (i.e., due to normal reactive hyperemia or inflammation) and non-blanchable (i.e., Stage 1 PU/PI). Blanchable erythema is seen when a skin area that is reddened turns pale as a result of applying light pressure with a finger or plastic disc and preventing blood flow to the region. As the pressure is released, skin turns red again.[33] Non-blanchable erythema is visible skin redness that persists with the application of pressure. It indicates structural damage to the capillary bed/microcirculation. (Figure 6)
-
- Darkly pigmented skin: for darkly pigmented skin, a blanching response may not be visible. Other signs of PU/PI such as discoloration compared to the surrounding area, pain, and firmness should be prioritized.
- In patients with dark skin, pressure injuries (PIs) are often detected at later stages than in those with lighter skin.[95] To improve early detection and prevention, the implementation of advanced skin assessment methods and visual augmentation tools in clinical practice is recommended.[95] For details, refer to sections 'Comprehensive Skin Assessment' in topic "Pressure Ulcers/Injuries - Prevention" and 'Infrared thermography' and 'Ultrasound and subepidermal moisture (SEM) measurement' below.
-
Practice tips: it is important to note that blanchable erythema or changes in sensation, temperature, or firmness may precede visual changes [9] and represent an opportunity to implement offloading and pressure redistribution strategies before further skin damage occurs.
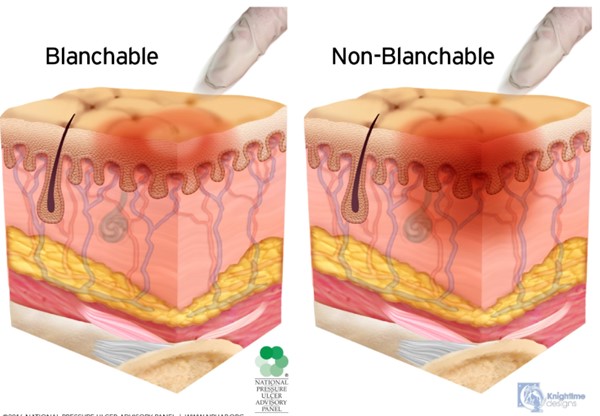
Fig. 6. Blanchable erythema (normal reactive hyperemia) and non-blanchable erythema (Stage 1 PU/PI) (click to enlarge) NPIAP copyright & used with permission
- Presence of deep tissue injury (DTI): Intact or non-intact skin with localized area of persistent non-blanchable deep red, maroon, purple discoloration or epidermal separation revealing a dark wound bed or blood filled blister. Pain and temperature change often precede skin color changes.
- According to the NPIAP [33]:
- The area with DTI may be preceded by tissue that is painful, firm, mushy, boggy, warmer or cooler as compared to adjacent tissue.
- DTI may be difficult to detect in individuals with dark skin tones.
- Evolution may include a thin blister over a dark wound bed. The wound may further evolve and become covered by thin eschar.
- Evolution may be rapid exposing additional layers of tissue even with treatment.
- DTIs may resolve without tissue loss or may evolve rapidly to reveal the actual extent of tissue injury (Figures 7 and 8). Evolution of DTIs to full-thickness lesions has been reported to occur in ~10 to 70% of the cases.[96]
- In one retrospective study, 1% of the DTIs healed, 4% remained purple and intact, 20% evolved to partial thickness ulcers, and 71% evolved to full thickness ulcers.[97]
- Another retrospective study evaluated 45 DTIs at initial visit and on average 6 days later (range from 1 to 20 days). All DTIs received preventive care. Final assessment showed that 5% healed, 48% remained the same, 26% of the DTIs evolved into full-thickness lesions, and 17% evolved into unstageable pressure ulcers.[98]
- A retrospective study evaluated 128 DTIs at initial visit and on average 6 days later (range 1 day to 14 weeks).[96] The final assessment showed that 85 sDTIs (66.4%) completely resolved or were progressing toward resolution, 31 (24.2%) remained unchanged (still described as purple-maroon discoloration or a blood-filled blister), and 12 (9.3%) evolved to to full-thickness tissue loss.
|
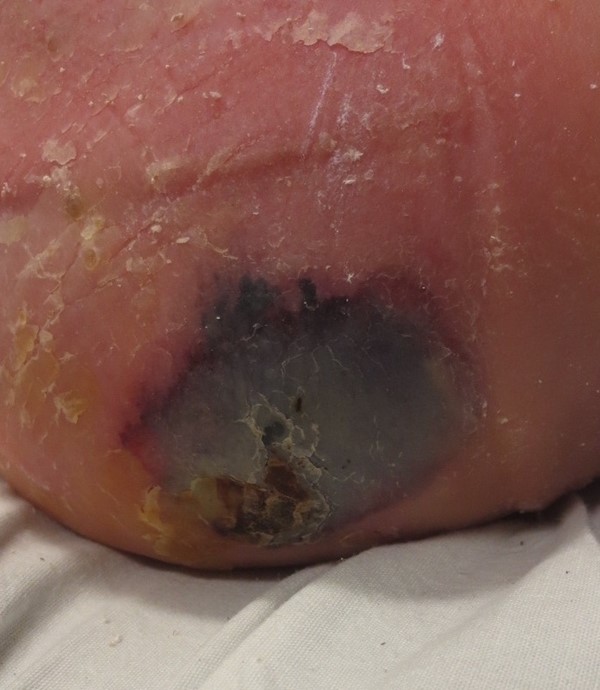
Fig. 7. Deep tissue injury on the heel
|
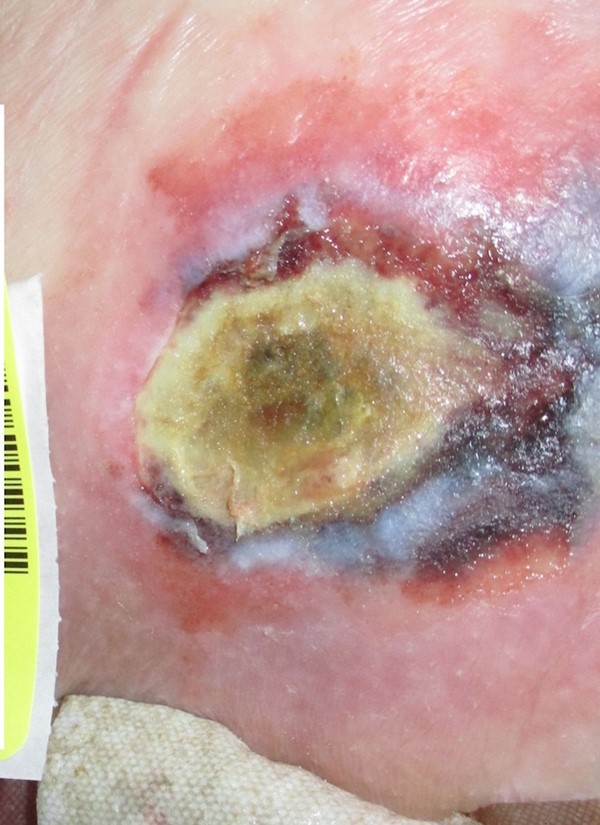
Fig. 8. Evolving deep tissue injury
|
- Ulcer size: depth, width and length
- Wound bed: necrosis, fibrin, granulation, etc
- Wound edges: hyperbole, sinus tracts, undermining, hypopigmentation of re-epithelialized tissue (Figure 9)
- Exudate: type and quantity
- Odor
- Periwound skin:
-
- Redness, warmth, induration or hardness, swelling, and any obvious signs of clinical infection.[99]
- Blisters, skin discoloration, induration may indicate pending PU/PI development
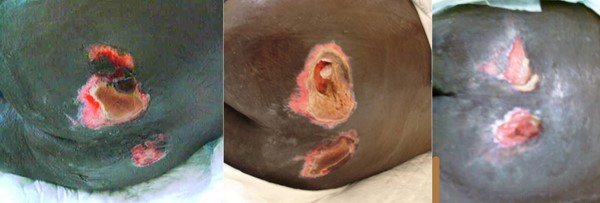
Fig. 9. Healing stage 4 pressure ulcers/injuries on the buttocks on a darkly pigmented patient. Note the hypopigmentation of the re-epithelialized tissue at the wound margins on the rightmost picture. By Tânia Santos, RN, MSc
Infection associated with PU/PI
The International Wound Infection Institute consensus updated the stages of clinical infection to contamination, colonization, local infection, spreading infection and systemic infection.[100] For more information on the wound infection continuum, see section 'Relevance' in topic "Wound Culture - Swabs, Biopsies, Needle Aspiration".
- By default, chronic wounds are contaminated by several types of bacteria.[101][102] It is generally recognized that wound infection gradually increases over time with respect to the number and virulence of microorganisms.[33] Table 2 shows signs and symptoms associated with stages of the wound infection continuum (based on the International Wound Infection Institute).[100]
BIOFILM
- Bacterial biofilms are estimated to be present in ~78.2% of chronic wounds, including in infected PUs/PIs.[33][103]
- Biofilms play important roles in chronic inflammation and may ultimately lead to delayed wound healing.[33] For more information on biofilm, see section 'Impaired Wound Healing - Role of Biofilm' in topic "Principles of Wound Healing".
- Signs of biofilm in a PU/PI include [33]:
- Failure to heal, recalcitrance or delayed healing despite appropriate antibiotic therapy
- Increased exudate
- Poor granulation or friable hypergranulation
- Low level erythema and/or low level chronic inflammation
- Secondary signs of infection
Local infection
- Classical clinical signs of local infection are shown in Table 2.[33]
- Infection in a PU/PI may present itself through subtle signs, as shown in Table 2 below.[33][100]
- For PU/PI in individuals with darkly pigmented skin, prioritize assessment of the following characteristics when assessing infection: skin heat, skin tenderness, change in tissue consistency, and pain.[33]
- Inexpensive, pen/pocket sized, no-touch surface infrared thermometry devices can be used to predict wound-related deep and surrounding infection. This device may be used on patients of any skin color, but may be especially useful for darkly pigmented skin. A 3 degree Fahrenheit increase in temperature of the periwound skin compared to skin of the contralateral limb, associated with 2 of the following criteria (Os probing to bone, new breakdown and necrotic tissue, erythema flare, exudate or smell) is predictive of deep or surrounding skin infection.[104]
- If ulcer probes to the bone, or has not shown signs of improvement despite standard therapy, evaluate the individual for osteomyelitis. See section 'Diagnosis - Infection associated with PU/PI' below.
- Ischial ulcers may also present with ischial bursitis. Anatomically, there is no bursa in the ischial area, but sliding forces can lead to an acquired bursa that can become infected and impair healing of an ischial PU/PI. Ischial bursitis may also occur in the absence of a PU/PI.[6][105]
Spreading infection
- See signs of spreading infection in PUs/PIs in Table 2 below.
systemic infection
- See signs of systemic infection in PUs/PIs in Table 2 below.
Table 2: Signs and symptoms associated with stages of the wound infection continuum (based on the International Wound Infection Institute) [100]
| Local infection, covert (subtle) signs | Local infection, overt (classic) signs
| Spreading infection | Systemic infection |
- Wound healing:
- Lack of signs of healing for two weeks
- Wound characteristics:
- Friable granulation tissue, bleeding;
- Hypergranulation
- Malodor;
- Increased pain in the ulcer;
- Increased heat in the tissue around the ulcer;
- Increased drainage from the wound;
- An ominous change in the nature of the wound drainage (e.g., new onset of bloody drainage, purulent drainage);
- Increased necrotic tissue in the wound bed; and/or
- Pocketing or bridging in the wound bed
| - Wound healing:
- Delayed wound healing beyond expectations
- Wound characteristics:
- Erythema
- Local warmth
- Swelling
- Purulent discharge
- New or increasing pain
- Increasing malodor
| - Wound characteristics:
- Extending in duration +/- erythema
- Lymphangitis
- Crepitus
- Wound breakdown/ dehiscence with or without satellite lesions
- Systemic signs/symptoms:
- Malaise/lethargy or nonspecific general deterioration
- Loss of appetite
- Inflammation, swelling of lymph glands
| - Systemic signs/symptoms:
- Severe sepsis
- Septic shock
- Organ failure
- Death
|
Complications
- Infection: acute and chronic osteomyelitis are the most common complication.[6] Soft tissue infections are also common, and abscesses can be formed when deep tissue injury (DTI) tracts become walled off [34] Infected ischial or sacral ulcers can further complicate and result in necrotizing fasciitis due to descending infection from the ulcer into the thigh compartment, or Fournier's gangrene of the scrotum and perineum.[6][106]
- Acute sepsis, amyloidosis, heterotopic ossification, septic joint, perineal and urethral fistula, squamous cell carcinoma changes in PU/PI.[6]
- Ulcer recurrence [34]
Diagnosis
Pressure ulcer/injury
Diagnosis of PU/PI is clinical, based on information gathered during history and physical examination. Laboratory tests ordered upon initial assessment help establish a baseline and monitor any chronic underlying medical conditions, as well as the patient’s nutritional status, which may be factors that impair wound healing.[6][7] Diagnostic tests can be considered when investigating non-healing ulcers and/or complications.
For all patients with PU/PI:
-
Laboratory tests [6]:
-
- White blood cells and differential white blood count (WBC)
- C-reactive protein or erythrocyte sedimentation rate (ESR): nonspecific, but may indicate acute/chronic bone infection
- Complete metabolic panel: to assess liver, kidney function, nutritional status. Tests include albumin, blood urea nitrogen, calcium, carbon dioxide, chloride, creatinine, glucose, potassium, sodium, total bilirubin and protein, and liver enzymes (alanine aminotransferase, alkaline phosphatase, and aspartate aminotransferase)
- Coagulation panel: may indicate liver dysfunction, nutritional deficiency, bleeding disorders thus abnormal values should be investigated/corrected in patients undergoing surgery. Tests include activated partial thromboplastin time (APTT), prothrombin time/International Normalized Ratio (PT/INR), thrombin time (TT)
- Urinalysis and culture/sensitivity
-
Radiographs: X-rays may be useful to detect bone changes that may lead to PU/PI and to rule out other pathology.[107] If indicated, consider plain X-rays of the pelvis in the antero-posterior (AP) position, and bilateral hip X-ray in the lateral position: X-rays detect ectopic bone or bone changes that may lead to PU/PI, and air within the PU/PI, which helps assess extent of the lesion beyond physical examination.[6][108] Advantages include simplicity, minimal need for patient cooperation, less costly. Common findings are listed below [6]:
-
- Ischial area:
-
- Free air or fistulous canals, representing ulcer extensions
- Heterotopic ossification secondary to recurrent ulceration and infection
- Destruction of ischial tuberosity seen secondary to recurrent infection
- Osteitis: irregular surface of the ischium secondary to calcification and chronic infection
- Trochanteric and hip area:
-
- Free air secondary to the ulcer or trochanteric bursa
- Heterotopic ossification
- Subluxation and rotation of the hip joint frequently seen in spinal cord injured patients leads to pressure on greater trochanter when sitting
- Fracture of intertrochanteric region in insensate patients
- Necrosis of the head of the femur secondary to infection, arthritic changes, congenital abnormality seen in spina bifida
For suspected deep tissue injury (DTI)
-
Ultrasound: clinicians may consider ultrasound of the tissue overlying the ischial tuberosity.[39] The best diagnostic method has not been determined, but a some non-randomized clinical studies have found that ultrasound may allow early detection of subclinical deep tissue injury before signs are evident on the skin, and may allow prediction of ulcers that are likely to progress. Studies suggest discontinuous fascia and heterogeneous hypoechoic areas are more reliable predictors of future progression of pressure ulcers than other findings.[109][110]
-
Infrared thermography: thermography is a noninvasive, noncontact method for measuring temperature differences between injured and adjacent skin (warm tissue is well perfused, cold tissue is not). [111] If resources and training are available, the NPIAP suggests use of an infrared thermographic imaging device or an infrared thermometer to help identify a Stage 1 PU/PI or suspected DTI.[33][112][113][114][115][116][117]
- A retrospective study (n=85) showed that pressure-related intact discolored areas of skin (e.g., Stage 1 PU or DTI) that are on average 1.2 degrees C cooler than adjacent skin are more prone to evolving to necrosis.[112]
- Infrared thermography may be especially useful in assessing darkly pigmented skin, as the visible color of the skin has no impact on the infrared radiation captured by thermography.[111]
For patients at risk of developing PU/PI (prediction of PU/PI development)
-
Infrared thermography: if resources and training are available, the NPIAP suggests use of an infrared thermographic imaging device or an infrared thermometer to help assess temperature of skin and soft tissue. Low certainty evidence indicates that cooler temperature in the center of an area of skin discoloration is predictive of PU/PI development (see 'Summary of Evidence' below).[33][112][113][114][115][116][117]
-
Ultrasound and subepidermal moisture (SEM) measurement: SEM is a noninvasive portable skin assessment device used in adjunct to clinical judgement, that assesses changes in SEM during the initial inflammatory phase of wound healing. Prolonged mechanical loading blocks blood and lymph flow, waste products accumulate in the cell niche and interstitial space and increases SEM.[118]
- If resources and training are available, the NPIAP suggests use of a SEM device as an adjunct to routine clinical skin assessment, especially when assessing darkly pigmented skin.[33]
- Low certainty evidence generated by retrospective and prospective cohort studies indicate that ultrasound and SEM may be promising in the detection and prediction of early tissue damage and PU/PI presence.[119][120][118] SEM values increase with increasing tissue damage, and ultrasound can detect pockets of fluid/edema which may be correlated with different levels of tissue damage. Higher quality clinical trials are needed to confirm these findings.[121]
- Current literature indicates that there are still significant knowledge gaps in clinical practice preventing widespread use of SEM devices to help detect impending PUs/PIs. To promote adoption, training, guidelines, infection control and device availability need to be considered.[122][123]
Infection associated with PU/PI
For suspected soft tissue infection or no healing signs after 2 weeks of debridement and pressure relief:
-
Tissue biopsy or quantitative validated swab technique (e.g., Levine) for culture, obtained after debridement:
- Tissue biopsy: microbial loads greater than 105 of any organism per gram of wound tissue or presence of any level of beta-hemolytic streptococcus is typically considered an indicator of infection of chronic wounds.[85] However, obtaining tissue biopsies may be resource intensive and most geographic and clinical settings have limited or no access to this diagnostic modality.[33] Therefore, an acceptable alternative to quantitative culture of biopsied tissue is a semi-quantitative wound swab.[33]
- Wound swab: for sample policies and procedures on how to collect a wound swab, see topic "How to Collect a Wound Swab (Levine Technique) for Culture".
- Chronic wound fluid protein analysis: differential expression levels of specific wound proteins assayed by mass spectrometry and multiplexed microassays are predictive of wound healing.[33][124]
For suspected osteomyelitis:
-
Bone biopsy for culture and histology: invasive, but it is the gold standard for definitive diagnosis of osteomyelitis. The sensitivity (ability to correctly identify osteomyelitis) and specificity (ability to correctly identify absence of osteomyelitis) of noninvasive tests for diagnosing osteomyelitis are not as high as direct bone biopsy and are not as useful in determining treatment.[85] Some disadvantages include the need for invasive sampling and possible cross-contamination with pathogens present in exudate and skin.[125]
- Imaging modalities (Table 3): noninvasive tests such as magnetic resonance imaging (MRI), 3 phase-bone scan (scintigraphy), ultrasound, computed tomography (CT) scans.
Table 3. Diagnostic imaging studies for osteomyelitis
| Imaging modality |
Sensitivity |
Specificity |
Comments |
| Radiographs |
14 - 54% [107]
|
68 - 70% [107]
|
Useful to rule out other pathology, relatively cheap [126][107]
|
| Magnetic resonance imaging (MRI) |
78 - 100% [107][127]
|
60 - 90% [107]
|
- Imaging modality with the greatest sensitivity for osteomyelitis. MRI Allows for early detection, helps differentiate between soft tissue and bone infection and assess extent of infection.[126][107] Might be useful especially in preempting early surgical intervention.[128]
- T1 and T2 images should be ordered. Intravenous gadolinium contrast is not needed to diagnose osteomyelitis, as it does not increase the proportion of actual acute osteomyelitis that are correctly identified as such (sensitivity). [127] However, contrast increases the proportion of actual non-acute osteomyelitis cases that are correctly identified as not having the condition (specificity). [127] Contrast is useful in chronic osteomyelitis, as inflamed areas can be better visualized.[129]
- Costly and requires patient cooperation [128]
|
3 phase-bone scan (scintigraphy)
|
~85% [126]
|
~25% [126]
|
- May be used if MRI is contraindicated.[128] It reliably excludes bony infection limited to bone [130]
- Does not detect adjacent soft tissue infection or the presence of orthopedic devices, and loses specificity if there is local trauma or recent surgery, as these mimic osteomyelitis.[131] It is relatively expensive and requires use of radionuclide.
|
| Computed tomography (CT) |
67% [126]
|
50% [126]
|
- Generally not used in osteomyelitis evaluation. Requires the use of radiation and is limited in its ability to predominantly show bone changes.
|
| Ultrasound |
n/a |
n/a |
- Not helpful for visualizing bone changes associated with osteomyelitis, nor can it detect associated surrounding soft-tissue infection, but it can be useful in the detection of fluid collection in joint or soft tissue.[128]
|
Differential Diagnosis
- Stage 1 (non-blancheable erythema of intact skin)
- Superficial burn (assess history of exposure)
- Cellulitis (suspect if located on a non-pressure area)
- Post-injury inflammation
- Deep tissue pressure injury
- Non-palpable erythema that blanches on compression
- Stage 2 (partial thickness skin loss with exposed dermis)
- Moisture-associated skin damage (MASD) including incontinence-associated dermatitis (IAD), intertriginous dermatitis (ITD), medical adhesive-related skin injury (MARSI) or traumatic wounds (skin tears, tape burns, excoriations)
- Herpes zoster
- See topic "Pressure Ulcers/Injuries - Is it Really a Stage 2?"
- Stage 3 (full-thickness skin loss) or Stage 4 (full-thickness skin and tissue loss)
- Burns, necrotizing fasciitis, calciphylaxis (may have history of rapid development of eschar)
- Infection (abscess)
- Diabetic Foot Ulcer, Venous ulcers, arterial ulcer, Inflammatory Ulcers (e.g. Pyoderma Gangrenosum)
- Unstageable PU/PI (obscured full-thickness skin and tissue loss)
- Arterial ulcer, calciphylaxis
- Deep tissue pressure injury [132]
- Stage 2 pressure ulcers, incontinence-associated dermatitis, skin tears, bruising, hematoma, venous engorgement, arterial insufficiency, necrotising fasciitis and terminal skin ulcers. (See NPIAP poster "Deep Tissue Pressure Injury or an Imposter?").
- COVID-19 dermatologic changes: skin changes due to COVID-19-related hypercoagulable state may mimic deep tissue injury.[133]
- According to the NPIAP, if the lesion is on a non-pressure bearing surface and cannot be attributed to pressure and/or shear, the lesion should not be classified as pressure injury.
- If the lesion is on a pressure bearing surface or has been subjected to shear, further investigation needs to be carried out. History is important, such as timing and development of the lesions, presence of factors leading to deep tissue injury, and implementation of adequate preventative interventions. In order to rule out concomitant deep tissue injury, the NPIAP recommends palpating the lesions to detect differences in tissue consistency and temperature.
Table 4 below illustrates selected conditions that may be mistaken for a PU/PI. To make an appropriate patient assessment and avoid misdiagnosis, it is essential to evaluate risk factors and clinical presentation, in addition to wound appearance.[134]
Table 4. Selected Differential Diagnosis of Pressure Ulcers/injuries Stages 2, 3 and 4 [134]
|
Differential diagnosis
|
Risk factors
|
Clinical symptoms
|
Wound appearance
|
Workup
|
| Pressure ulcer/injury |
Reduced mobility, poor circulation, malnutrition, fecal and urinary incontinence |
Gradually progressive wound over pressure points: coccyx, ischium, spinous processes, heels, ankles, elbows, iliac crest, trochanters |
- Stage 2: exposed dermis, erythema, moist
- Stage 3: exposed subcutaneous adipose tissue, rolled edges, tunneling
- Stage 4: exposed fascia, muscles, bone
|
Clinical diagnosis: rule out differential diagnoses |
|
Arthropod/spider bite
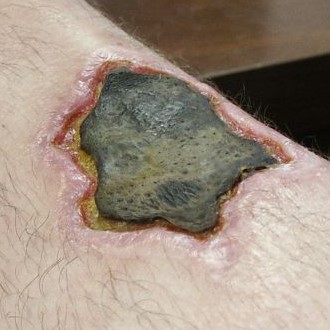
|
Geographical location |
Sharp pain, burning, itching, fever, nausea, vomiting, malaise |
Puncture wound with edema; necrosis into ulcer possible |
Clinical diagnosis |
|
Calciphylaxis
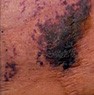
|
End-stage renal disease, dialysis, cirrhosis, multiple myeloma, polyneuropathy, warfarin usage, obesity, diabetes |
Painful lesions (thighs, abdomen); hyperphosphatemia, hypercalcemia, hyperparathyroidism |
Violaceous or erythematous subcutaneous nodules and plaques; ulcerations, necrosis, eschar possible |
Skin biopsy: microvascular calcifications |
| Coagulopathies |
Hypercoagulable states: trauma, pregnancy, Factor V Leiden, prothrombin mutation, sickle cell disease, OCP use |
Wide range of symptoms |
Wide range of presentations |
Clinical diagnosis |
|
End-of-life skin failure (Kennedy Terminal Ulcer)
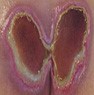
|
Towards end of life |
Rapid decline, deterioration, change in existing ulcer |
Dark discoloured patch, irregular borders (horseshoe/butterfly shape); necrosis possible |
Clinical diagnosis |
|
Epidermolysis bullosa
|
Family history |
Wide range of symptoms |
Fragile skin, blisters, erosions |
Skin biopsy |
| Genital herpes of gluteal region and fold |
Sexual activity |
Painful vesicles or ulcers; systemic symptoms: fever, lymphadenopathy, muscle aches |
Vesicles, ulcerations |
PCR; Cultures |
| Graft-versus-host disease |
Recent bone marrow transplant |
Abdominal cramping, diarrhea |
Maculopapular rash spreading from face/palms/soles to trunk |
Skin biopsy (to rule out other causes) |
|
Hidradenitis suppurativa
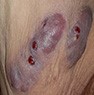
|
Obesity, smoking, family history |
Intertriginous areas, painful lesions; prodromal burning, warmth, pruritus, hyperhidrosis |
Deep nodules, draining tracts, fibrotic scarring |
Clinical diagnosis |
|
Marjolin ulcer
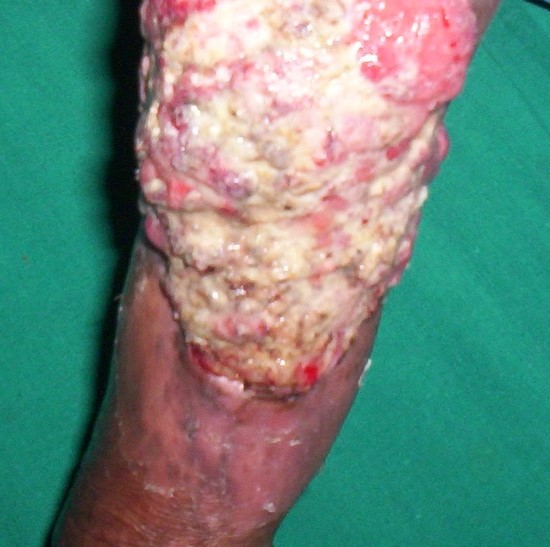
|
Nonhealing burn scars or chronic pressure ulcers |
|
Nodular ulcer, indurated margins, tendency to bleed |
Biopsies at varying depths; MRI |
|
Necrotizing fasciitis
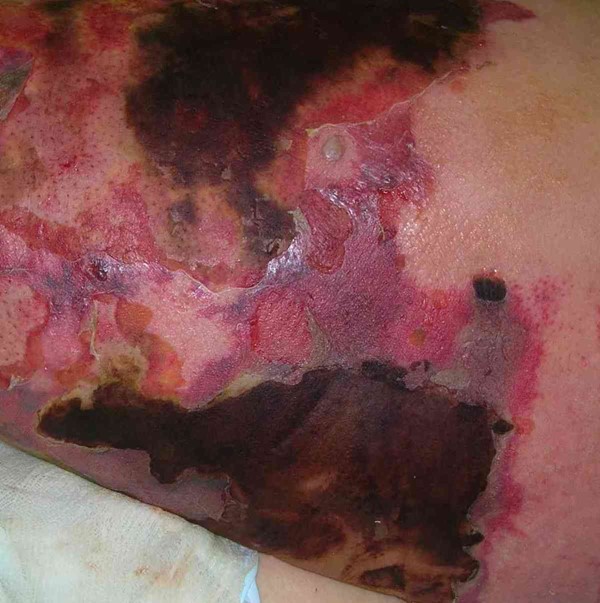
|
History of trauma or penetrating injury |
Pain disproportionate to wound appearance, rapidly progressive, foul smelling tan purulence; late findings: sepsis, multiorgan failure |
Black/grey necrotic tissue, rapidly expanding, subcutaneous emphysema and crepitus |
-
Clinical diagnosis for surgical debridement;
-
Ultrasound if hemodynamically unstable;
-
CT scan: unorganised adipose tissue, fascia thickening, gas transecting fascia planes
|
|
Pyoderma gangrenosum
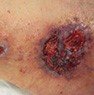
|
Older age (>65 years), autoimmune disease (IBD, RA, etc.), traumatic event |
Often found on lower extremities |
Violet border, peripheral macular erythema |
Biopsy: neutrophil infiltrates |
|
Radiation wounds
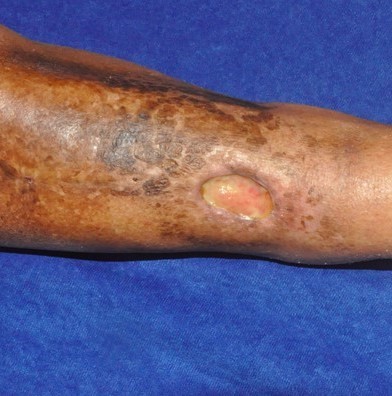
|
Obesity, diabetes, poor nutrition, prior radiation, connective tissue disorders, concurrent chemotherapy |
Itching, erythema, decreased sweating |
Ranges from erythema to necrotic ulcers |
Clinical diagnosis |
| Stevens–Johnson syndrome |
Drug reaction (allopurinol, carbamazepine, phenytoin, TMP-SMX, cephalosporins) |
Fever, pharyngitis, malaise, cough prior to lesions |
"Widespread erythematous macules → confluent erythema; flaccid bullae → ulcerations" |
Clinical diagnosis |
| Surgical wounds after pilonidal cyst excision |
Sedentary lifestyle, male sex, obesity, excessive body hair |
Painful lesion in gluteal cleft region |
Midline abscess or infected sinus with tracts |
Clinical diagnosis |
| Trauma and enterocutaneous fistula |
Abdominal surgery, trauma, IBD, malignancy, radiation therapy, diverticulitis |
Signs of infection, abdominal pain |
Wound with continuous drainage |
CT scan: visualize fistula tract and potential abscesses |
| Vascular disease of gluteal muscle and iliac disease |
Coronary artery disease, hypertension, obesity, smoking history, sedentary lifestyle |
Painful lesions |
Punched out lesions, shiny skin (possible necrosis), purple discolouration, pallor |
Ankle Brachial index; Duplex ultrasound |
| Vasculitis |
Drug reactions (sulfa, NSAIDs, penicillin), infection, neoplasm, inflammatory disease (lupus, RA, Sjögren’s syndrome) |
Often on lower extremities |
Wide range of presentations |
Clinical diagnosis |
Classification/ Staging
Please refer to topic "Pressure Ulcers/Injuries - Classification/Staging" for pictures, complete definitions, comparison and pictures of the NPIAP and Medicare MDS Classification/Stages. Guidance on PU/PI staging is provided as well.
The NPIAP classification is the most widely adopted classification for pressure ulcers/injuries (PUs/PIs)[8], and was updated in 2016.[9] In the US, CMS allows nursing homes to adopt the NPIAP guidelines in their clinical practice and nursing documentation. However, CMS requires its adapted version of the NPIAP guidelines to be used when entering information in the Minimum Data Set (MDS) system, used by nursing homes. Stage definitions of this MDS version do not perfectly correlate with each stage as described by NPIAP. For coding purposes, the MDS should be coded according to the definitions stated in the Medicare MDS 3.0 RAI Manual.[135] Briefly, the NPIAP staging is as follows:
- Stage 1 Pressure Injury: Non-blancheable erythema of intact skin
- Stage 2 Pressure Injury: Partial-thickness skin loss with exposed dermis
- Stage 3 Pressure Injury: Full-thickness skin loss
- Stage 4 Pressure Injury: Full-thickness skin and tissue loss
- Unstageable Pressure Injury: Obscured full-thickness skin and tissue loss
- Deep Tissue Pressure Injury
Ulcer Healability (Prognosis)
Categorization of wound healability (i.e., healable, maintenance, or non-healable) is of particular importance. [56] This designation defines for the clinician, patient, and family an expected course of action, plan of care, and healing rate. As a prerequisite to setting realistic treatment objectives, wounds are differentiated as:
- Healable wound: the cause is corrected, there is enough blood supply to heal; moist interactive healing
- Maintenance wound: the wound could heal, but the cause is not corrected due to patient unwillingness to adhere to treatment or a lack of required system resources
- Non-healable wound: the patient is ill or may have negative protein balance or inadequate blood supply that is not bypassable or dilatable
If patient has any of the conditions below, consider a non-healing program. The ulcer will likely not heal with conservative treatment only:
- Co-morbidities that impede healing:
- Ulcer is malignant tumor,
- Major organ failure
- Blood supply to a PU/PI located in the lower extremity is inadequate (as evidenced by ABI, Doppler, TCOM, other exams)
If patient has any of the conditions that impede wound healing below, consider a maintenance-healing program until element impeding healing is mitigated:
Co-morbidities:
- Uncontrolled diabetes
- Immunosuppression
- Obesity: BMI > 40
- Inadequate nutrition (abnormal serum protein, unintended weight loss)
Drugs and interventions :
- Steroids, immunosuppressants
- Chemotherapy/ radiation
Lifestyle:
- Regular smoking, illicit drugs, alcohol abuse
- Financial or resource constraints
Documentation
- Structured risk assessment should be documented initially and at subsequent evaluations. (See 'Risk Assessment')
- The American Medical Association (AMA) recommends that to support diagnosis of PU/PI, clinicians document location, ulcer size, depth (full thickness, partial thickness), wound characteristics (i.e., wound bed tissue type - eschar, fibrin, slough, granulation tissue, exudate, odor, sinus tracts, undermining), infection and appropriate staging (see 'Classification/Staging' above). Codes for PU/PI include location, laterality and severity (as defined as PU/PI stages), so only one code is needed to describe the patient's condition.[136]
-
- Documentation of PU/PI assessment should be done initially, then at least weekly.[33] The NPIAP recommends a 2 week period to evaluate progress towards healing and determine if any changes in the care plan are needed..[33] At long term care facilities, Medicare recommends plan reassessment if PU/PI does not show signs of improvement despite 2-4 weeks of adequate care.[5] At each dressing change, observe the PU/PI for signs of deterioration/improvement to determine if a change in treatment is required..[33] It is also important to explore and document any barriers to adherence to care plan, including patient's or caregiver's refusal of a given intervention.
- Documenting signs of improvement to support medical necessity (Medicare):
-
- Reimbursement for wound care services on a continuing basis for a particular wound in a patient requires documentation in the patient's record that the wound is improving in response to the wound care being provided.
- It is not medically reasonable or necessary to continue a given type of wound care if evidence of wound improvement cannot be shown.
- Medicare expects the wound-care treatment plan to be modified in the event that appropriate healing is not achieved.
- Such evidence must be documented with each date of service provided.
- Evidence of improvement includes measurable changes (decreases) of some of the following:
-
- Drainage
- Inflammation
- Swelling
- Pain
- Wound dimensions (diameter, depth)
- Tissue type: necrotic tissue/slough
- Tools that facilitate standardized assessment and documentation should be used whenever possible:
-
- Validated wound assessment tools:
-
- Pressure Ulcer Scale for Healing (PUSH) Tool [93]
-
Bates-Jensen Wound Assessment Tool [94]
- Wound Reference Wound Prep&Dress Tool creates notes to help support medical necessity that can be copied and pasted to electronic medical records
- Wound imaging: digital photographs of PU/PI at the first consultation and periodically thereafter to document progress is helpful and ensures consistency of care among healthcare practitioners, facilitates telemedicine in remote areas, and illustrates improvement to the patient.
-
Wound measurement: methods include manually measuring length and width (the longest length with the greatest width at right angles), manual tracing, digital photography, and software programs that calculate wound dimensions from a photograph of the lesion. Wound tracings that calculate the area via digital software are slightly better than linear measurement.[137]
-
Documenting adequate practice to prevent/manage PU/PI at long term care facilities (nursing homes) that accept Medicare: to avoid letters of non-compliance, facilities are required to document [5]:
-
- Provision of preventive care, consistent with professional standards of practice, to residents who may be at risk for development of pressure injuries; or
- Provision of treatment, consistent with professional standards of practice, to an existing pressure injury; or
- Interventions to prove that a resident did not develop an avoidable PU/PI.
- NOTE: To cite non-compliance (F686), it is not necessary to prove that a PU/PI developed. F686 can be cited when it has been determined that the provider failed to implement interventions to prevent the development of a PU/PI for a resident identified at risk.
- The plan of care and patient response should be clearly documented and reflected in the entire medical record.[23] It is critical that clinicians follow their organization's documentation policy, whether this is charting by exception or real time documentation of events and care.
-
Patient reported outcome (PRO) Tools: Validated instruments allow clinicians to obtain feedback on whether a given treatment plan is resulting in the best outcomes possible and is improving patient's quality of life (QOL). PRO assessment will continue to gain importance as health care systems transition from a volume-based reimbursement model to a value-based model.[138]
-
-
QOL tools validated for PU/PI patients: the 36-item Short Form (SF-36) [139][140], PU-QOL instrument, [141], SCI-QOL Pressure Ulcers scale for spinal cord injured patients with PU/PI.[142]
-
-
- Medicare Quality Payment Program, Improvement Activity "Promote Use of Patient-Reported Outcome Tools" suggests use of Wound-Quality of Life (QoL) and patient-reported Wound Outcome.[143]
-
Validated pain measurement tools include: for neonates up to 6 months - CRIES (Crying; Requires O2 for Saturation > 95%; Increasing vital signs; Expression; Sleepless)[33]; for children 2 months to 7 years of age - FLACC (Face, Leg, Activity, Cry, and Consolability)[33], and the Wong-Baker FACES scale; for adults - visual analog scale (VAS). The Braden Scale has a subscale that measures sensory perception/pain.
-
- Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"
CODING
ICD-10
See all PU/PI ICD-10 codes in the Appendix - ICD-10 Coding
- Code first any associated gangrene (I96)
- Identify site of PU/PI
-
- Ankle
- Back
-
- Lower (except sacral region)
- Sacral region (includes coccyx)
- Upper
- Unspecified part
- Buttock
- Contiguous site of back, buttock, hip
- Elbow
- Head (including face)
- Heel
- Hip
- Other specified site
- Unspecified site
- Specify laterality (does not apply to sacral region, contiguous site of back, buttock, and hip; or other unspecified)
-
- Specify PU/PI stage (see 'Classification/Staging' above)
-
- Stage 1
- Stage 2
- Stage 3
- Stage 4
- Unstageable (based on clinical documentation that stage cannot be clinically determined because ulcer anatomical depth cannot be visualized - it should not be confused with unspecified, which is when no documentation regarding the stage of PU/PI exists)
- Unspecified stage
Unstageable pressure ulcers
Assignment of the code for unstageable pressure ulcer (L89.--0) should be based on the clinical documentation. These codes are used for pressure ulcers whose stage cannot be clinically determined (e.g., the ulcer is covered by eschar or has been treated with a skin or muscle graft). This code should not be confused with the codes for unspecified stage (L89.--9). When there is no documentation regarding the stage of the pressure ulcer, assign the appropriate code for unspecified stage (L89.-- 9). If during an encounter, the stage of an unstageable pressure ulcer is revealed after debridement, assign only the code for the stage revealed following debridement.
Coding Guidance for "Present on Admission" PU/PI in Acute Care Setting
According to the CMS ICD-10 Official Guidelines for Coding and Reporting [144]:
- If a patient is admitted to an inpatient hospital with a PU/PI at one stage and it progresses to a higher stage, two separate codes should be assigned: one code for the site and stage of the ulcer on admission and a second code for the same ulcer site and the highest stage reported during the stay. The indicator "present on admission" (POA) should be used by coders for each PU/PI diagnosis.
- For ulcers that were present on admission but healed at the time of discharge, coders should assign the code for the site and stage of the pressure ulcer at the time of admission.
- Coders should not report pressure ulcers that are completely healed at the time of admission.
Background
- In 2008, CMS introduced a claim-level payment penalty that denied increased reimbursement for discharge records that contained a documented hospital-acquired condition (HAC).[145][146] PU/PIs Stages 3 and 4 are one of the 14 HACs subject to payment penalty. However, this list of reportable events excludes a PU/PI of lower stage that progressed to Stage 3 or 4, if the lower stage was recognized upon admission.[147]
- Under the 2008 CMS HAC payment provision, hospitals are required to report a corresponding POA indicator for each PU/PI diagnosis. Possible values for the POA indicator include among others [145][148]:
- "Y", indicating that the diagnosis was present at the time of admission
- "N", indicating that the diagnosis was not present a the time of admission (i.e., and thus considered a HAC).
- Current coding guidelines for the POA indicator allow HACs to be coded as POA for conditions diagnosed during the admission that were clearly present but not diagnosed until after the admission occurred.[144] See other POA Criteria in Appendix I of the CDC ICD-10-CM Official Guidelines for Coding and Reporting.[144] Of note, under the current CMS HAC-POA program, hospitals have financial incentives to mischaracterize clinical conditions (e.g., document a PU/PI as stage 2 rather than stage 3, if acquired during the hospitalization, or document a PU/PI as stage 3 rather than stage 2, if POA).
- Despite these coding guidelines issued by CMS, CDC, Coding Clinic, reports [144][147][149] show that there are variances in interpretation among acute care facilities, so clinicians might want to confirm local practice with their specific coding/billing department.
Medicare MDS (long term care facilities)
For MDS coding purposes, PUs/PIs should be coded according to the definitions stated in the Medicare MDS 3.0 RAI Manual.[5] See stage definitions in 'Classification/Staging' above. For complete guidance, click on reference to refer to Section M of Chapter 3, in 'Long-Term Care Facility Resident Assessment Instrument 3.0 User’s Manual'.[5]
Kennedy Terminal Ulcer
There is no specific ICD-10 for Kennedy Terminal Ulcer (KTU). KTUs are considered to be PUs/PIs that generally occur at the end-of-life, and thus are coded and staged as PUs/PIs.[5] CMS recognizes the that PUs/PIs at the end-of-life may be unavoidable.[5] To be considered unavoidable and consistent with regulatory requirements, it is important that clinicians properly document that the the facility has implemented appropriate efforts to stabilize the resident’s condition (or indicted why the condition cannot or should not be stabilized) and has provided care to prevent or treat existing PU/PIs (including pertinent, routine, lesser aggressive approaches, such as, cleaning, turning, repositioning).[5]
APPENDIX
ICD-10 Coding
Code First any associated gangrene (I96)
- Red boxes indicate non-billable code, Green boxes are billable codes
L89 Pressure ulcer- L89.0 Pressure ulcer of elbow
- L89.00 Pressure ulcer of unspecified elbow
- L89.000 …… unstageable
- L89.001 …… stage 1
- L89.002 …… stage 2
- L89.003 …… stage 3
- L89.004 …… stage 4
- L89.006 Pressure-induced deep tissue damage of unspecified elbow
- L89.009 …… unspecified stage
- L89.01 Pressure ulcer of right elbow
- L89.010 …… unstageable
- L89.011 …… stage 1
- L89.012 …… stage 2
- L89.013 …… stage 3
- L89.014 …… stage 4
- L89.016 Pressure-induced deep tissue damage of right elbow
- L89.019 …… unspecified stage
- L89.02 Pressure ulcer of left elbow
- L89.020 …… unstageable
- L89.021 …… stage 1
- L89.022 …… stage 2
- L89.023 …… stage 3
- L89.024 …… stage 4
- L89.026 Pressure-induced deep tissue damage of left elbow
- L89.029 …… unspecified stage
- L89.1 Pressure ulcer of back
- L89.10 Pressure ulcer of unspecified part of back
- L89.100 …… unstageable
- L89.101 …… stage 1
- L89.102 …… stage 2
- L89.103 …… stage 3
- L89.104 …… stage 4
- L89.106 Pressure-induced deep tissue damage of unspecified part of back
- L89.109 …… unspecified stage
- L89.11 Pressure ulcer of right upper back
- L89.110 …… unstageable
- L89.111 …… stage 1
- L89.112 …… stage 2
- L89.113 …… stage 3
- L89.114 …… stage 4
- L89.116 Pressure-induced deep tissue damage of right upper back
- L89.119 …… unspecified stage
- L89.12 Pressure ulcer of left upper back
- L89.120 …… unstageable
- L89.121 …… stage 1
- L89.122 …… stage 2
- L89.123 …… stage 3
- L89.124 …… stage 4
- L89.126 Pressure-induced deep tissue damage of left upper back
- L89.129 …… unspecified stage
- L89.13 Pressure ulcer of right lower back
- L89.130 …… unstageable
- L89.131 …… stage 1
- L89.132 …… stage 2
- L89.133 …… stage 3
- L89.134 …… stage 4
- L89.136 Pressure-induced deep tissue damage of right lower back
- L89.139 …… unspecified stage
- L89.14 Pressure ulcer of left lower back
- L89.140 …… unstageable
- L89.141 …… stage 1
- L89.142 …… stage 2
- L89.143 …… stage 3
- L89.144 …… stage 4
- L89.146 Pressure-induced deep tissue damage of left lower back
- L89.149 …… unspecified stage
- L89.15 Pressure ulcer of sacral region
- L89.150 …… unstageable
- L89.151 …… stage 1
- L89.152 …… stage 2
- L89.153 …… stage 3
- L89.154 …… stage 4
- L89.156 Pressure-induced deep tissue damage of sacral region
- L89.159 …… unspecified stage
- L89.2 Pressure ulcer of hip
- L89.20 Pressure ulcer of unspecified hip
- L89.200 …… unstageable
- L89.201 …… stage 1
- L89.202 …… stage 2
- L89.203 …… stage 3
- L89.204 …… stage 4
- L89.206 Pressure-induced deep tissue damage of unspecified hip
- L89.209 …… unspecified stage
- L89.21 Pressure ulcer of right hip
- L89.210 …… unstageable
- L89.211 …… stage 1
- L89.212 …… stage 2
- L89.213 …… stage 3
- L89.214 …… stage 4
- L89.216 Pressure-induced deep tissue damage of right hip
- L89.219 …… unspecified stage
- L89.22 Pressure ulcer of left hip
- L89.220 …… unstageable
- L89.221 …… stage 1
- L89.222 …… stage 2
- L89.223 …… stage 3
- L89.224 …… stage 4
- L89.226 Pressure-induced deep tissue damage of left hip
- L89.229 …… unspecified stage
- L89.3 Pressure ulcer of buttock
- L89.30 Pressure ulcer of unspecified buttock
- L89.300 …… unstageable
- L89.301 …… stage 1
- L89.302 …… stage 2
- L89.303 …… stage 3
- L89.304 …… stage 4
- L89.306 Pressure-induced deep tissue damage of unspecified buttock
- L89.309 …… unspecified stage
- L89.31 Pressure ulcer of right buttock
- L89.310 …… unstageable
- L89.311 …… stage 1
- L89.312 …… stage 2
- L89.313 …… stage 3
- L89.314 …… stage 4
- L89.316 Pressure-induced deep tissue damage of right buttock
- L89.319 …… unspecified stage
- L89.32 Pressure ulcer of left buttock
- L89.320 …… unstageable
- L89.321 …… stage 1
- L89.322 …… stage 2
- L89.323 …… stage 3
- L89.324 …… stage 4
- L89.326 Pressure-induced deep tissue damage of left buttock
- L89.329 …… unspecified stage
- L89.4 Pressure ulcer of contiguous site of back, buttock and hip
- L89.40 …… unspecified stage
- L89.41 …… stage 1
- L89.42 …… stage 2
- L89.43 …… stage 3
- L89.44 …… stage 4
- L89.45 …… unstageable
- L89.46 Pressure-induced deep tissue damage of contiguous site of back, buttock and hip
- L89.5 Pressure ulcer of ankle
- L89.50 Pressure ulcer of unspecified ankle
- L89.500 …… unstageable
- L89.501 …… stage 1
- L89.502 …… stage 2
- L89.503 …… stage 3
- L89.504 …… stage 4
- L89.506 Pressure-induced deep tissue damage of unspecified ankle
- L89.509 …… unspecified stage
- L89.51 Pressure ulcer of right ankle
- L89.510 …… unstageable
- L89.511 …… stage 1
- L89.512 …… stage 2
- L89.513 …… stage 3
- L89.514 …… stage 4
- L89.516 Pressure-induced deep tissue damage of right ankle
- L89.519 …… unspecified stage
- L89.52 Pressure ulcer of left ankle
- L89.520 …… unstageable
- L89.521 …… stage 1
- L89.522 …… stage 2
- L89.523 …… stage 3
- L89.524 …… stage 4
- L89.526 Pressure-induced deep tissue damage of left ankle
- L89.529 …… unspecified stage
- L89.6 Pressure ulcer of heel
- L89.60 Pressure ulcer of unspecified heel
- L89.600 …… unstageable
- L89.601 …… stage 1
- L89.602 …… stage 2
- L89.603 …… stage 3
- L89.604 …… stage 4
- L89.606 Pressure-induced deep tissue damage of unspecified heel
- L89.609 …… unspecified stage
- L89.61 Pressure ulcer of right heel
- L89.610 …… unstageable
- L89.611 …… stage 1
- L89.612 …… stage 2
- L89.613 …… stage 3
- L89.614 …… stage 4
- L89.616 Pressure-induced deep tissue damage of right heel
- L89.619 …… unspecified stage
- L89.62 Pressure ulcer of left heel
- L89.620 …… unstageable
- L89.621 …… stage 1
- L89.622 …… stage 2
- L89.623 …… stage 3
- L89.624 …… stage 4
- L89.626 Pressure-induced deep tissue damage of left heel
- L89.629 …… unspecified stage
- L89.8 Pressure ulcer of other site
- L89.81 Pressure ulcer of head
- L89.810 …… unstageable
- L89.811 …… stage 1
- L89.812 …… stage 2
- L89.813 …… stage 3
- L89.814 …… stage 4
- L89.816 Pressure-induced deep tissue damage of head
- L89.819 …… unspecified stage
- L89.89 Pressure ulcer of other site
- L89.890 …… unstageable
- L89.891 …… stage 1
- L89.892 …… stage 2
- L89.893 …… stage 3
- L89.894 …… stage 4
- L89.896 Pressure-induced deep tissue damage of other site
- L89.899 …… unspecified stage
- L89.9 Pressure ulcer of unspecified site
- L89.90 …… unspecified stage
- L89.91 …… stage 1
- L89.92 …… stage 2
- L89.93 …… stage 3
- L89.94 …… stage 4
- L89.95 …… unstageable
- L89.96 Pressure-induced deep tissue damage of unspecified site
Summary of Evidence
SOE - Infrared devices
(back to text)
Prospective cohorts and retrospective studies
- A study has shown that the use of temperature differentials as measured by an infrared imaging system was not helpful in detecting patients at high risk for PU/PI among a critically ill patient sample.[113] Near infrared spectroscopy (NIRS) has been shown to be unreliable in the examination of the influence of external pressure on oxygenation of the soft tissues in the sacral area.[114]
- On the other hand, other studies have found infrared devices to be helpful: a small case series showed that alternate light source can help detect tissue trauma and areas at risk of PU/PI but not readily visible by naked eye.[115]A prospective cohort study showed that diffuse correlation spectroscopy (DCS) and diffuse near-infrared spectroscopy (DNIRS) can help predict the development of PU/PI among spinal cord injury subjects with only nonbleachable redness [116].
- In 2011, Judy et al. published a prospective cohort study that found infrared thermography to be helpful in detecting which high risk individuals are more likely to develop PU/PI. [117]
- In 2012, Farid et al. analyzed data from 85 individuals with skin discoloration due to pressure but no skin breaks, where skin temperature readings were part of the evaluation. These readings were taken from discolored areas and nearby healthy skin using a handheld infrared device. Initially, about 65% of these individuals had cooler temperatures in the discolored areas compared to the surrounding skin. Those with lower temperatures in the discolored regions (on average 1.2 degrees C cooler than adjacent skin) were significantly more likely to experience skin necrosis within a week.[112]
- In 2016, Cox et al. conducted a study in skilled nursing homes (n=67 participants) to explore how well infrared thermography can predict skin and tissue health outcomes. They tracked skin color changes for 1 to 2 weeks and found that at the two-week mark, almost half of the areas with changed skin color had healed, but about a third had turned into necrotic tissue. Areas that were initially cooler at the center were much more likely to develop necrosis within a week. These results indicate that infrared thermography can detect potential deep tissue injuries early on. However, the study's findings are less certain because of the broad range of possible outcomes and the fact that the study mainly included Caucasian participants, which may limit how well these results apply to other groups.[150]
REVISION UPDATES
| Date | Description |
| 11/5/23 | Updated to version 3 (Sections on epidemiology, assessment) |
| 7/29/23 | Updated 'Coding' section |
| 10/28/21 | Updated sections on end-of-life PU/PI, and unavoidable ulcers.
|
| 10/9/19 | Updated coding section with new ICD-10 codes effective 10/1/19
|
5/27/19
| Updated sections on end-of-life PU/PI, infection assessment tools and Kennedy Terminal Ulcer coding
|
| 5/14/19 | Updated sections on differential diagnoses and on PU/PI detection tools |