ABSTRACT
Annually in the United States, over half a million patients will present to emergency department with burns, and 10% of those require hospitalization. Burns are injuries to the skin caused by various agents or mechanisms, including thermal, electrical, chemical, and less common causes like radiation exposure. Thermal burns result from contact with hot liquids, solids, or gases, leading to tissue damage. The physiologic effects of burns are manifested by tissue damage and subsequent cell death.
This topic provides an evidence-based review and framework for assessment of acute burns, including epidemiology, risk factors, etiology, pathophysiology, first aid, primary and secondary assessment, transfer criteria, burns classification, documentation and ICD-10 coding.
First Aid: The first responder should remove the subject from all potential burning sources including heat/flame, live electrical sources, and chemicals, with careful consideration of his/her own personal safety. See other steps recommended by Clinical Guidelines in this section.
Initial Care: The initial assessment of patients with acute burns must be expedited using a systematic protocol of care such as the Advanced Burn Life Support (ABLS) and Emergency Management of Severe Burns (EMSB). This systematic approach involves the assessment of the patient, resulting in a plan for subsequent management and transfer to a specialized burn center if necessary. See details on primary and secondary assessment, and assessment of inhalation injury in this section.
Classification: Burn injuries are frequently classified according to the depth of tissue destruction and extent of the injury measured in body surface area percentage. See classification systems in this section.
Burn center referral criteria: See guidelines for referral to specialized burn units, as recommended by the American Burns Association
Documentation: Ensure treatment documentation meets medical necessity criteria. See recommendations for documentation in this section
Treatment: See topic "Acute Burns - Treatment".
ICD-10 Coding: See section 'ICD-10 Coding'
INTRODUCTION
Overview
This topic provides a review and framework for assessment of acute burns, including epidemiology, risk factors, etiology, pathophysiology, first aid, primary and secondary assessment, transfer criteria, burns classification, documentation and ICD-10 coding. For clinical guidelines and quality measures, see "Acute Burns - Overview". For management of acute burns see "Acute Burns - Treatment".
Background
- Definition: Burns are cutaneous injuries caused by various agents or mechanisms, including thermal (further subdivided into liquid, solid and gaseous agents), electric, chemical, and other rarer tissue insults such as radiation. The physiologic effects of burns are manifested by tissue damage and subsequent cell death.
-
Relevance: Annually in the United States, over half a million patients will present to emergency department with burns, and 10% of those require hospitalization.[1]
Epidemiology
Prevalence and incidence data below were extracted from the American Burns Association National Burn Repository reports of 2015 and 2017 [1][2]:
Prevalence
- By age:
-
- Pediatric (ages 1-15 years): 23.5%
- Adult (ages 20 to 59 years): 55%
- Geriatric (age 60 years or older): 15%
- By gender:
-
- By ethnicity:
-
- Caucasian: 59%
- African-American: 20%
- Hispanic: 14%
- Other: 7%
Incidence
- In the U.S. in 2016, there were 486,000 documented burn injuries (includes individuals that presented for and received medical treatment).
Mortality
- Survival Rate: 96.8%, on average.[1][2] Rates differ depending on burn depth and extent. More than 67% of the reported total burn sizes were less than 10% total body surface area and these cases had a mortality rate of 0.6%.[1]
- The mortality rate from severe burn injuries is much lower than it was in the 1960s due to advances in surgical management and the advent of intensive care. Apart from the establishment of specialized burns centers, the survival rate after severe burns improved between 1970 and 1980. The evolution of knowledge about burns has heavily evolved in all aspects of care, including the initial response at the accident site, diversity of the response to trauma, clinical and surgical management, understanding of the risk factors for complications and sequelae, as well as prevention including educational measures and campaigns aimed toward high risk populations.[3]
Etiology
- By reported cause upon admission to healthcare facility:
-
- Fire/Flame: 3%
- Scald: 34%
- Contact: 9%
- Electrical: 4%
- Sources can be either of low voltage, such as contact with a home electrical outlet, or of high voltage, which is most common in the workplace.[4]
- Chemical: 3%
- Chemical agents include caustic acids (lab chemicals such as hydrochloric acid, battery acid), caustic bases (bleach, drain cleaner), and even fertilizer.[4]
- Other: 7%
- By place of occurrence:
-
- Home: 73%
- Occupational: 8%
- Street/Highway: 5%
- Recreational/Sport: 5%
- Other: 9%
- A large proportion of accidents leading to burn injuries occur at home, with children being victims in nearly half of all cases [1][2]; in this population, a common burn mechanism is direct contact with hot liquids (scald); abuse or neglect should be investigated. The geriatric population also has a high risk for liquid thermal burns due to their lessened ability to react coupled with other common physical manifestations of aging, such as diminished visual acuity. In adults, burns occur mostly at home or place of work.[1][2]
Pathophysiology
Thermal injury
- Tissue trauma from burn injury results in local and systemic inflammatory responses. The local response depends on the intensity of the heat or level of causticity of the agent in contact with the tissues, making the depth of the burn directly proportional to the duration of contact between the agent and the tissues, as well as the amount of heat released by the agent.
- Local inflammatory response was first described by Jackson in 1947 as being comprised of three zones (Figure 1)[5]:
-
- The zone of coagulation occurs at the point of the greatest thermal injury, which is generally at the most central area; this site has undergone irreversible damage due to the denaturation of tissue proteins.
- The zone of stasis surrounds the central zone and is characterized by diminished tissue perfusion; the process of volume resuscitation post burn injury is to increase perfusion in this area and prevent progression of the damage.
- The zone of hyperemia is the most peripheral zone, and represents an area of reactivity characterized by increased tissue perfusion. The chance of tissue recovery in the first 24 hours is likely, except in cases of prolonged hypoperfusion or severe sepsis. “Conversion” of burns, as it is commonly referred to, or apparent progression of injury to deeper tissue levels, can occur over this time period.
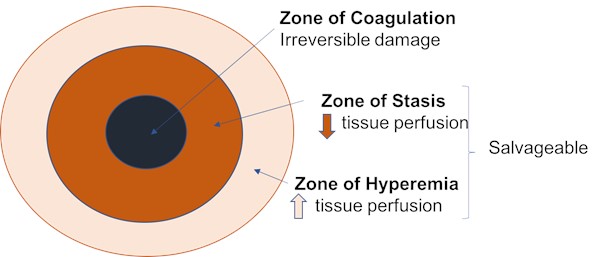
Figure 1. Pathophysiology of burns - Jackson's zones
-
- Systemic inflammatory response occurs when the burn injury exceeds 20% of the TBSA (total body surface area). The release of pro-inflammatory cytokines and other inflammatory mediators in the anatomical site of the burn injury leads to hypovolemic and distributive shock, or “burn shock”, which is characterized by increased capillary permeability and systemic vascular resistance, small vessel hydrostatic pressure, third spacing, decreased cardiac output, and hypovolemia. Edema forms rapidly in the interstitial space during the first eight hours after initial injury, and continues to develop, though at a slower rate, for at least 18 hours.
-
- Fluid resuscitation with crystalloids is required to reverse the evolution of shock. Volume required for resuscitation can be estimated by the calculated TBSA and the weight of the patient. The rate of fluid infusion must be titrated every hour, with adjustments based on the adequacy of the physiological response including the presence of adequate diuresis and normal arterial blood pressure. See "Fluid Resuscitation" section in "Acute Burns - Treatment" for available calculation methods.
Electrical injury
- The pathophysiology of electrical burn depends on the voltage, current flow, and tissue resistance. Electrical burns can damage tissue by the local generation of heat and the direct action of the passage of the current itself through the tissue. Heat leads to coagulative cell necrosis and the electrical current causes cell membrane disruption leading to tissue loss and death.[6]
- Fatal complications include severe acute renal failure secondary to massive rhabdomyolysis and myoglobinuria, sepsis and disseminated intravascular coagulation.[6]
Chemical injury
- The severity and clinical appearance of chemical burns are directly correlated with the amount, concentration of the agent, viscosity, location and duration of contact and penetrability. The pathophysiology of a chemical injury varies according to the type of chemical, which can be acid or alkaline, and organic or inorganic [7]:
- Acids denature cellular proteins leading to coagulative necrosis. On the skin, substances with a pH of less than 2 cause coagulative necrosis upon contact with the skin; which often results in an eschar that prevents further penetration of the acid.[7][8][9]
- Alkaline substances denature proteins and causes saponification of lipids (present in the cell membrane); the combined effect allows alkaline substances to cause liquefactive necrosis and penetrate into the submucosa and muscularis, which often results in more severe injuries than those caused by acids.[9]
- Organic substances differ from inorganic chemicals by the presence of carbon. Organic agents typically dissolve the lipid membrane of cells which disrupt physiological processes.
- Inorganic solutions cause injury by denaturation of physiological proteins. Inorganic solutions form salts and directly bind to skin.[8]
- In addition, as chemical reactions are exothermic, thermal burns often occur simultaneously.
Systemic effects
- Almost every organ system is affected by severe burns, causing inflammation, hypermetabolism, muscle wasting, insulin resistance and metabolic changes known to remain for several years following injury.[10][11]
- Two distinct physiological phases have been identified after severe burns: emergent or 'ebb' phase, and 'flow' or hypermetabolic phase.[10][11]
- Emergent or 'ebb' phase: the first phase, which peaks at 12 hours after burn injury, can last up to 72 hours and is marked by decreased tissue perfusion, decreased metabolism and edema.
- Primary goals during this phase involves restoring and preserving tissue perfusion to avoid ischemia from hypovolemic and cellular shock, and restoring or preserving normothermia.[10][11]
- Adequate resuscitation is essential during this phase. Refer to section "Fluid Resuscitation" section in "Acute Burns - Treatment".
- Hypothermia may occur in people with severe burns and can lead to serious complications such as shock, multisystem organ failure, and death.[11] For people with more than 10% of the body surface burned, the core body temperature may reset up to 2oC above normal temperature readings.[11] That is, if their core body temperature is within the normal range of equal to or less than 36.5oC, they may be experiencing hypothermia and is therefore at risk of further harm and associated complications.[11] Risk factors for hypothermia include younger or older age, female gender, larger percentage of the body burned.[11] Targeted temperature management with adjuncts such as application of warming devices and prewarmed intravenous fluids in the early stages of hospital admission and during surgery is essential to survival.[11]
- 'Flow' or hypermetabolic phase: the second phase phase, which starts approximately 3 days after the burn injury and lasts more than a year, is characterized by a decrease in vascular permeability, increased heart rate, and decreased peripheral vascular resistance resulting in an increase in cardiac output and increased metabolic phase.[10][11]
ASSESSMENT
First Aid
First aid starts with the first responder at the site of the accident and ends when Initial Care starts at a healthcare facility.[12] First aid steps recommended by the International Society for Burn Injuries (ISBI), are outlined below [12]:
- The first responder should remove the subject from all potential burning sources including heat/flame, live electrical sources, and chemicals, paying attention to his/her own personal safety.
- Removing the subject from the source of the flame is more important than putting out the fire.
- If the person's clothes are on fire let the person lie down or sit down then pour water liberally, as wet clothes will not catch fire.
- For heat/flame injuries, cool down the burn wound with clean cold running water for 15-20 minutes. After cooling the site of injury, the patient should be kept warm.
- For chemical burns, identify the agent. Ensure personal and bystanders' safety and remove and dispose of all contaminated clothing. Irrigate area with running clean water for up to 45 minutes. If possible, call poison control (while awaiting first responders) to determine if there is an antidote or or action that can be performed to reverse to diminish the chemical reaction causing the burn.
- For electrical burns, ensure personal and bystanders' safety and separate the victim from the source using non-conductive material. Begin cardiopulmonary resuscitation if needed, and cool the burns
- Transfer the victim to the nearest medical or burn facility. During transport:
- Elevate limbs during transportation to limit edema
- If suspicion of inhalation injury, position individual between lying and sitting (e.g., with the head of bed at 45 degrees)
- If trained personnel available, initiate primary assessment (ABCDE) as described below
Initial Care
The initial assessment of burn patients must be expedited using a systematic protocol of care such as the Advanced Burn Life Support (ABLS) and Emergency Management of Severe Burns (EMSB).[3] This systematic approach involves the primary and secondary assessment of the patient, resulting in a plan for subsequent management and transfer to a specialized burn center if necessary.[13]
Primary assessment
The initial assessment of a patient with acute burn follows the sequence below:
- Airway
- Breathing and Respiration
- Circulation
- Disability (Neurologic status)
- Exposure/environmental control: remove all the patient’s clothes completely but prevent hypothermia
Airway
- Protecting the airway in the patient with severe burns should be prioritized, and clinicians should always have a high index of suspicion for associated inhalation injury. Early endotracheal intubation is often indicated in patients with symptoms of inhalation injury or signs of burns on the face, in the nares, oral cavity, or oropharynx.[14] See section ‘Assessment of inhalation injury’ below.
Breathing and respiration
- After ensuring airway patency, auscultate lung fields to assess the adequacy of the patient’s ventilation and oxygenation. Early recognition of circumferential full thickness injury in the thoracic or cervical region is imperative for the timely performance of escharotomy to prevent respiratory compromise.[15]
Circulation
- Cardiac monitoring, continuous pulse oximetry, and measurement of arterial blood pressure must be performed for patients with major burns (see definition of 'major burn' in section 'Classification of Acute Burn Injuries - By severity' below ) in order to monitor the circulatory status of the patient. Volume resuscitation with crystalloids must be commenced based on the weight of the patient and the extent of burns, using a validated tool for calculation. [15] See "Fluid Resuscitation" section in "Acute Burns - Treatment"
Disability (Neurologic status)
- If there is a possibility of concomitant trauma, hypoxia related or unrelated to inhalation injury, drug use, or a preexisting neurological condition, neurological status should be assessed, and appropriate specialties consulted including orthopedics and neurology.[15]
Exposure
- The patient’s clothes must be removed completely, including accessories such as watches, rings, necklaces, including anything that may cause a tourniquet effect or prevent complete assessment. This process must occur in a warm environment in order to avoid hypothermia and associated sequelae.[15]
Burn Total Body Surface Area Calculation (TBSA)
- During the initial examination, Total Body Surface Area percentage (TBSA %) and depth of the burns will be estimated in order to best guide overall care, including accurate fluid resuscitation.[15] The extent of the burns may be calculated with:
-
- Lund-Browder tables: is the most accurate method for estimating TBSA for both adults and children and can be particularly useful in children since there is a great difference in proportions between the head and the lower limbs.
- The rule of nines: the body is divided into anatomical regions, and each area receives a value that is equal to nine or a multiple thereof.
- Patient’s palm: In cases where only one part of the body is burned, the rule of the patient’s palm may be used; the palm of the hand, including the fingers, corresponds to about 1% of the TBSA.[3]
- When calculating TBSA, clinicians should exclude areas with superficial (first degree) burns.[16] See section ‘Classification of Burn Injuries’ below.
- Patients with partial thickness burn injury that exceeds 20% of the TBSA experience “burn shock”, as described above, which requires carefully calculated volume resuscitation.[3][15] Fluid overload is as physiologically compromising to the patient as inadequate fluid resuscitation. Fluid overload can result in compartment syndrome in the extremities and abdomen as well as acute respiratory distress, whereas, inadequate fluid resuscitation may further prolong states of shock, cause the zone of coagulation to extend, and potentially lead to organ failure [14][15]. See "Fluid Resuscitation" section in "Acute Burns - Treatment"
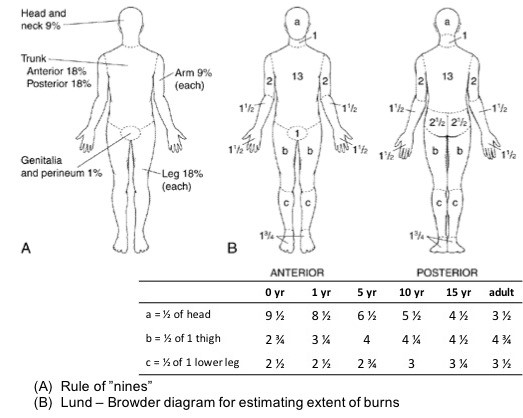
Figure 2. Calculating the Total Body Surface Area with the "Rule of Nines" and "Lund-Browder" tables
Assessment of inhalation injury
- In spite of the great advances that have been made in the management of burns in recent decades, patients with concomitant inhalation injury continue to suffer increased morbidity and mortality. For this reason, efforts at developing new therapeutic approaches for improving outcomes in these patients are crucial.
- The diagnosis of inhalation injury is made based on [3]:
-
- History of :
-
- Exposure to fumes in an enclosed space with incomplete combustion of materials
- Decreased level of consciousness during the physical examination
- Presence of soot in the buccal or nasal cavity
- Presence of burns of the face
- Signs of :
-
- Hoarseness, carbon eschars, wheezing, and dyspnea
- Bronchoscopy: A systematic review concluded that bronchoscopy is a useful tool for identifying inhalation injury (moderate certainty evidence, level B) [17]
- Chest X-rays or measured normal oxygenation do not exclude inhalation injury.
- The three main classes of inhalation injury are: supraglottic, infraglottic and systemic.
-
- The main physiologic presentation following infraglottic inhalation injury is increase in bronchial blood flow; the mucosa becomes hyperemic, edematous, produces copious mucous secretions, and transudation of plasma occurs in the airways, which alters the motility of local cilia.[18]
- Currently, there is no consensus on the diagnosis or grading of inhalation injury. Approaches differ by geographic location, from conservative management to advanced therapies based on nebulization with various pharmacologic agents.
- Treatment options for these three subtypes differ based on the patient presentation and symptoms.
- Systemic effects of inhalation injury may occur [14][19][20]:
-
- Indirectly by hypoxia or hypercapnia, which result from pulmonary function deficits and the systemic effects of pro-inflammatory cytokines
- Directly by toxicity from metabolic byproducts such as carbon monoxide and cyanide. Both of these agents with may cause nonspecific clinical symptoms, including circulatory collapse. Carbon monoxide and cyanide poisoning must be treated with oxygen and hydroxocobalamin respectively. See protocol for treatment of carbon monoxide poisoning with hyperbaric oxygen therapy in topic "Acute Carbon Monoxide Poisoning".
Secondary Assessment
- During this phase, assess the need of an indwelling urinary catheter, nasogastric tube, and other monitoring devices or lines.
- Collect urine drug screens and serum specimens to assess for ingestion of or exposure to other substances that may have contributed to injury or changes in neurological status.
- Tetanus immunization status must be investigated and if there is no definitive record of current vaccination, or if the vaccination is outdated, the patient must be immunized as soon as possible.[3]
- This systematic process simplifies identification of the major contributors to mortality from extensive burns in the first 24 hours, without waste of time or resources.[3]
Classification of acute burn injuries
Burn injuries are frequently classified according to the depth and extent of the injury:
By depth
The preferred terminology referring to the depth of burns that used the word “degree” has been changed to “thickness”. The latter, more descriptive system, is recommended, although both terms continue to be used.[16] Table 1 below describes the types of burns by depth of injury:
Table 1. Classification of burn injuries by depth
| Depth | Description | Injury characteristics
| Healing process |
|
Superficial (first degree)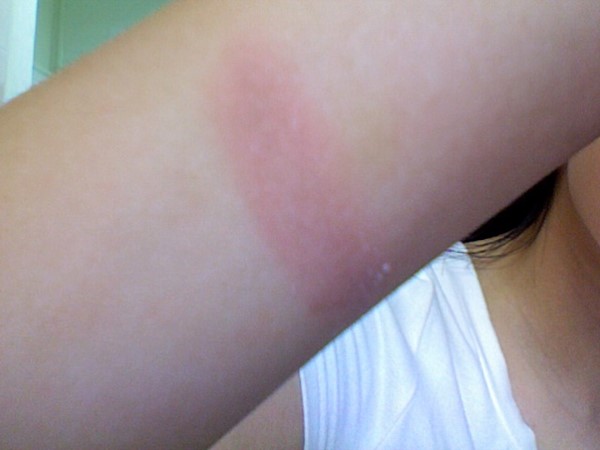
|
Involves only the epidermis, without forming blisters
Common causes include UV light, minor thermal burns
|
There may be desquamation of the epidermis
|
Local pain and erythema that resolves within 4 to 6 days, with complete resolution in 10 daysNo scar formation
|
|
Superficial partial thickness (superficial second degree)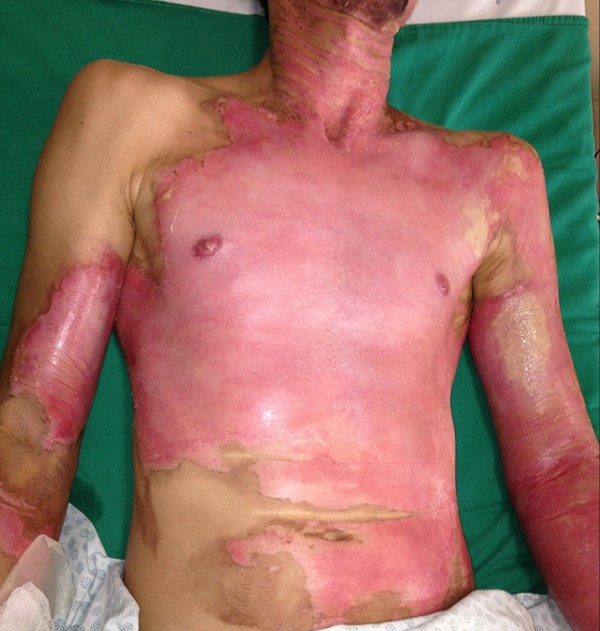
|
Affects the epidermis and upper layers of papillary dermis, may present with bullae
|
Red, moist and painful base, often with erythematous, weeping bullae
|
Repair occurs within 7 to 14 daysGenerally no scarring, but pigment changes possible
|
|
Deep partial thickness (deep second degree)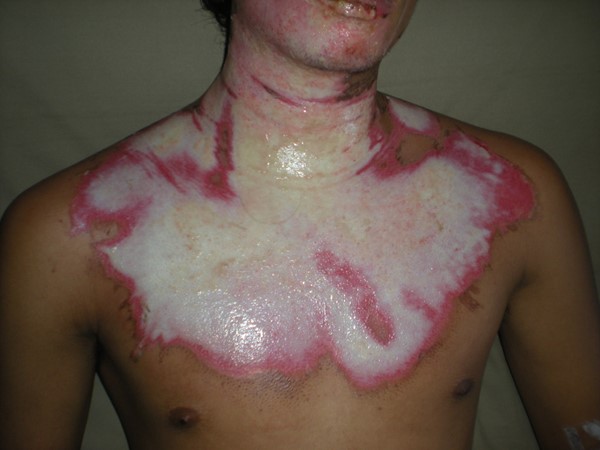
|
Affects the epidermis and most of the dermis, including the reticular dermis
|
White, dry base, less painful than superficial partial thickness
|
Repair can take 21 days or more depending on concomitant injuries and patient condition at time of injury
Typically causes some scarring and potentially contractures
|
|
Full thickness (third degree)
|
Affects the entire skin structure
|
Tight, leathery appearance, painless
Presence of plaques that vary in color from white to black
|
No repair, no re-epithelialization
Requires excision and skin graftingMay require escharotomy depending on site
|
By extent (total body surface area)
See calculation of TBSA in section ‘Burn Total Body Surface Area Calculation (TBSA)’ above. Table 2 illustrates a classification system based on TBSA. [21]
Table 2. Classification of burn injuries by total body surface area
| Classification | Total body surface area (TBSA)
| Grafted Area |
|
Small
|
Less than or equal to 10%
|
Less than 4%
|
|
Medium
|
Greater than 10% and less than or equal to 25%
|
Between 4 and 10%
|
|
Large
|
Greater than 25%
|
Greater than 10%
|
By severity
Burns can be classified by severity according to the Table 3 below, based on data from The American Burns Association and The American College of Surgeons. [22][23]
Table 3. Burns classification by severity.[22][23] Regardless of severity classification, patients that meet at least one of the referral criterion established by The American Burns Association (in general, moderate or major burns) should be referred to a burn center (see ‘Burn center referral criteria'). Many moderate burns may ultimately be treated at hospitals with experience in burn care, as determined by the burn center that would potentially manage the patient.
| Minor | Moderate | Major/ Severe |
- Partial-thickness burns <10% TBSA in patients 10 to 50 years old
- Partial-thickness burns <5% TBSA in patients under 10 or over 50 years old
- Full-thickness burns <2 % TBSA in any patient without other injury
Also:
- Does not meet any of the burn center referral criteria as defined by the American Burn Association
- Isolated injury
- Does not affect feet, face, hands, perineum or major joints
- Is not circumferential
|
- Partial thickness burns with 10–20% TBSA in adults
- Partial thickness burns between 5-10% TBSA in children or those over age 50
- 2–5% full thickness burn
- Low voltage burn, suspected inhalation injury, circumferential burn
- Concomitant medical problem predisposing to infection (e.g., diabetes, sickle cell disease)
|
- Partial thickness burns greater than 20% TBSA in adults
- Partial thickness burns greater than 10% TBSA in children or those over age 50
- >5% full thickness burn
- High voltage burn,
- Chemical burn,
- Any clinically significant burn to the eyes, ears, genitalia, or major joints,
- Clinically significant associated injuries (e.g., fracture, other major trauma)
|
Burn center referral criteria
After assessment is complete, and if at least one criterion proposed by the American Burns Association (ABA) is met, patients are triaged and transferred to a burn unit. Criteria for transfer are listed below [24]:
- Partial thickness burns greater than 10% TBSA.
- Full thickness burns in any age group
- Burn injury including the face, eyes, perineum, hands, feet and joints
- Electrical burns
- Chemical burns
- Inhalation injury or circumferential burn injury of the chest or limbs
- Associated illnesses that could complicate management, prolong recovery, or affect mortality.
- Associated multi-system trauma,
- Burned children in hospitals without qualified personnel or equipment for the care of children.
- Burn injury in patients who will require special social, emotional, or rehabilitative intervention, or patients with attempted self-harm
Infection Assessment
Acute burn injuries are initially sterile, however within 24-48 hours normal bacterial flora from the surrounding non-injured skin spreads into the injured area, increasing risk for infection. In addition, patients with severe burns admitted to acute care facilities are at risk for hospital-acquired infection.
- For patients with severe burn injuries (TBSA > 20%), the American Burn Association recommends constant monitoring for local and systemic signs of infection.[25]
- The standard diagnostic criteria for infection and sepsis do not apply to burn patients.[25] Patients with severe burns are in a state of chronic systemic inflammatory stimulation, with a baseline temperature elevated to about 38.5°C. Tachycardia and tachypnea persist for months in patients with extensive burns. White blood cell count is abnormally elevated, making leukocytosis a poor indicator of sepsis. Locally, erythema, pain and exudate are constantly observed in non-infected burn injuries. As a result, patient and wounds need constant surveillance, so that subtle systemic or local changes that may signal infection can be detected.[25]
- Definitions according to the American Burn Association Consensus on infection in burns [25]:
- Wound infection: Presence of bacteria alone is not considered infection. A burn wound is considered infected if bacteria is present in the wound and wound eschar at high concentrations (>10^5 bacteria/g tissue), and there is no invasive infection.
- Invasive infection: Infection is invasive if there is “presence of pathogens in a burn wound at concentrations sufficient in conjunction with depth, surface area involved and age of patient to cause suppurative separation of eschar or graft loss, invasion of adjacent unburned tissue or cause the systemic response of sepsis syndrome", culture shows >10^5 bacteria/g tissue, there is invasion or destruction of unburned skin/tissue. Invasive infection may occur with or without sepsis, but many burn wound invasive infections, however, are life threatening and need urgent treatment (usually wound excision).
- Diagnosis of wound infection: practically, diagnosis of wound infection is made when clinical signs of infection are observed and an organism is isolated from the wound organism.[25][26] Criteria by the American Burn Association are listed below [25]:
- Objective criteria:
- Quantitative biopsy (can be used to confirm but is not reliable. It may help with identifying the organism)
- Quantitative swab (poor test but may help with identifying organism)
- Tissue histology
- Subjective criteria:
- Changes in pain, erythema, exudate, color of the wound bed
- Yellow/green exudate may not be indicative of infection, as colonizing Pseudomonas aeruginosa produces exudate with this characteristics. Purple-black and “punched-out” areas of the wound suggests invasive Pseudomonas, which is a surgical emergency.
- Small papules of purulence may indicate Candida infection.
- Gray-brown plaques that can be scooped out of the wound suggests Aspergillus.
- Punched out lesions in the wound may indicate herpes simplex.
- Unexpected change in the appearance or depth of the wound
- Systemic changes
- Premature separation of burn eschar (if eschar has not been surgically excised)
- For management, see section 'Infection Management' in topic "Acute Burns - Treatment".
Documentation
Clinicians should ensure appropriate documentation of all encounters is in place, to support medical necessity. Important information to be included are listed below:
History and Physical Exam
- Age of the patient
- Burn mechanism and etiology of injury
- Burn depth and extent
- Time elapsed since injury, any first aid administered on site and during transport, mode of transport
- Primary and secondary assessment, presence of inhalation injury
- Presence of infection, co-morbidities
- Late effects/sequelae of burn injury
- Any criterion that would require patient to be referred to a specialized burn center (see section 'Burn center referral criteria')
- Work up, imaging, laboratory, and other diagnostic testing ordered
Impression
State mechanism of injury, agent, TBSA%, burn depth, if inhalation injury, presence of infection, co-morbid conditions
Plan
Depends on the severity of the acute burn, and should include:
- Disposition: should the patient be referred or treated at a designated burn center? If not, where is the place of service?
- Systemic treatment provided/ to be provided:
- initial stabilization
- pain control
- Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"
- coagulopathy management
- thromboprophylaxis
- antibiotics
- nutrition
- Medicare Quality Payment Program, Quality Measure:
- "Process Measure: Nutritional Screening and Intervention Plan in Patients with Chronic Wounds and Ulcers"
- "Patient Reported Nutritional Assessment and Intervention Plan in Patients with Wounds and Ulcers"
- "Preventative Care and Screening: Body Mass Index (BMI) Screening and Follow-Up"
- physical therapy
- disposition plans
- Local interventions:
- escharotomy
- excision with or without xenograft or autograft
- debridement
- cleansing
- topical therapy
- dressings
See topic "Acute Burns - Treatment"
CODING
ICD-10
See all Burn Injuries ICD-10 codes in the Appendix - ICD-10 Coding
Identify [27]:
- Burn type:
- Thermal burn: due to a heat source (e.g., fire, hot appliance, electricity, and radiation - excludes sunburns)
- Corrosion burn: due to chemicals. This code requires identification of the chemical substance, which is the first listed diagnosis. The chemical substance is found in the Table of Drugs and Chemicals. These codes identify the substance and the external cause or intent, so an external cause of injury code is not required. Assign codes for the place of occurrence, activity and external cause status.
- Burn depth:
- First degree (superficial): erythema
- Second degree (superficial or deep partial thickness): blistering
- Third degree (full thickness)
- Burn extent:
- Total body surface area (TBSA%) burned
- Total body surface area (TBSA%) burned with third degree burns
- Agent:
- Corrosive (T code - see Table of Drugs and Chemicals)
- Acids
- Alkalines
- Caustics
- Chemicals
- Thermal (except sunburn) (X-code - see Alphabetic Index to External Causes )
- Electricity
- Flame
- Heat (gas, liquid, or object)
- Radiation
- Steam
Note: Burns of the eye and internal organs are classified by site but not by degree.
Coding Guidelines for burns, related conditions, and complications of burns are as follows:
- Code first:
- For multiple external burns, first list the code that reflects the highest degree of burn
- For cases in which the site of the burn is not specified, or if there is a need for additional information on the extent of the burns, first list codes from category T31 Burns classified according to the extent of body involved, or T32 Corrosions classified according to the extent of body surface involved
- For each burn site, assign separate codes
- For burns of the same anatomic site:
- If different degrees of burn are present in the same anatomic site, assign only the highest degree recorded in the diagnosis (e.g., for second and third degree burns of right thigh, assign only code T24.311-)
- It is not recommended to use codes for 'unspecified sites' as these are extremely vague (codes from category T30)
- Classifying burns and corrosions by extent of body surface area involved (codes from category T31 and T32)
- Check TBSA% recorded with Lund-Browder tables, Rule of nines, or Palm Rule (see section 'Classification of Acute Burn Injuries')
- Codes from category T31 should be used whenever possible to:
- Provide data for evaluating burn mortality, such as that needed by burn units
- When there is mention of a third-degree burn involving 20% or more of body surface
- Non-healing burns: code as acute burns with 7th character extension 'A'. Necrosis of burned skin is coded as a non-healing burn
- Infected burns: use additional code for the infection
- Late effects/ sequelae of burns: use a burn or corrosion code with the 7th character 'S'. Both a code for a current burn or corrosion code and a code for sequela may be assigned on the same record
APPENDIX
ICD-10 Coding
- T20-T25 Burns and corrosions of external body surface, specified by site
- T20 Burn and corrosion of head, face, and neck
- T21 Burn and corrosion of trunk
- T22 Burn and corrosion of shoulder and upper limb, except wrist and hand
- T23 Burn and corrosion of wrist and hand
- T24 Burn and corrosion of lower limb, except ankle and foot
- T25 Burn and corrosion of ankle and foot
- T26-T28 Burns and corrosions confined to eye and internal organs
- T26 Burn and corrosion confined to eye and adnexa
- T27 Burn and corrosion of respiratory tract
- T28 Burn and corrosion of other internal organs
- T30-T32 Burns and corrosions of multiple and unspecified body regions
- T30 Burn and corrosion, body region unspecified
- T31 Burns classified according to extent of body surface involved
- T32 Corrosions classified according to extent of body surface involved
DRG
- DRG 927 EXTENSIVE BURNS OR FULL THICKNESS BURNS WITH MV >96 HOURS WITH SKIN GRAFT
- DRG 933 EXTENSIVE BURNS OR FULL THICKNESS BURNS WITH MV >96 HOURS WITHOUT SKIN GRAFT
- DRG 928 FULL THICKNESS BURN WITH SKIN GRAFT OR INHALATION INJURY WITH CC/MCC
- DRG 929 FULL THICKNESS BURN WITH SKIN GRAFT OR INHALATION INJURY WITHOUT CC/MCC
- DRG 934 FULL THICKNESS BURN WITHOUT SKIN GRFT OR INHALATION INJURY
- DRG 935 NON-EXTENSIVE BURNS
See principal or secondary diagnosis for each DRG in the ICD-10-CM/PCS MS-DRG v38.1 Definitions Manual
REVISION UPDATES
| Date | Description |
| 6/3/19 | Added section on Infection Assessment |
CURATED ARTICLES