ABSTRACT
Pyoderma gangrenosum is a rare but serious primary ulcerating condition of the skin that falls into the category of neutrophilic dermatoses. The prevalence of pyoderma gangrenosum is estimated to be of 0.01% among adult patients in the U.S.[1], and it is often associated with systemic diseases including inflammatory bowel disease, arthritis, and myeloproliferative disorders.[2]
Assessment of pyoderma gangrenosum is summarized in the 'Algorithm for Assessment of Pyoderma Gangrenosum'
History and Physical Examination: Pyoderma gangrenosum has different clinical presentations. Its morphological variants include classic ulcerative (most common), bullous, pustular, superficial granulomatous or vegetative pyoderma gangrenosum. Pyoderma gangrenosum may develop in periostomal, genital and extracutaneous locations. Pyoderma gangrenosum may also be part of several auto-inflammatory syndromes. See disease progression and clinical variants.
Diagnosis: There are no specific laboratory findings or pathognomonic histological features. For all patients, a through history, physical exam and biopsy are recommended. Diagnostic criteria have been developed and validated for ulcerative pyoderma gangrenosum. Differential diagnoses should be ruled out with workup guided by history. See diagnostic criteria, workup and biopsy for pyoderma gangrenosum
Differential diagnoses: The frequency of misdiagnosis of pyoderma gangrenosum has been shown to be as high as 10%. As many as 50% of patients who have had biopsies, have histopathological evidence of an alternative diagnosis on the initial or repeated biopsy.[3] Differential diagnoses include vascular occlusive disease, or venous disease, vasculitis, cancer, infection, exogenous tissue injury and other inflammatory disorders.
Documentation: Ulcers need to be properly documented for adequate ICD-10 assignment. See documentation checklist, tools and suggested patient reported outcomes instruments.
Treatment: See topic "Pyoderma Gangrenosum - Treatment"
When to refer to specialists:
- Dermatologists: for systemic treatment
- Rheumatologist, hematologist: if associated rheumatological or hematological diseases are present
- Gastroenterologist: if gastrointestinal symptomatology is prominent or problematic
- Wound care specialist: for non-healing wounds
- Ostomy specialist: if in a periostomal location
- Surgeon: if post-surgical onset
- Other specialists as needed to help rule out other differential diagnoses
ICD-10 Coding: See 'Coding' below.
INTRODUCTION
Pyoderma gangrenosum is a rare but serious primary ulcerating condition of the skin that falls into the category of neutrophilic dermatoses. Often associated with systemic diseases, pyoderma gangrenosum has been considered a diagnosis of exclusion, however recent diagnostic criteria may help discern positive findings that facilitate diagnosis of the most common clinical variant.[4][5] Originally described in 1930 by Brunsting et al, pyoderma gangrenosum is a distinct clinical entity characterized by chronic, recurring, destructive ulcerations of the skin, most commonly occurring on the lower extremities and trunk.[6]
Epidemiology
Incidence
- Pyoderma gangrenosum is a rare disease and the incidence of this disease is uncertain. It is estimated to occur in 3-10 patients per million population per year but the incidence is variable in different populations with a peak incidence occurring between the ages of 20-50 years with a slight female preponderance.[7] It is estimated that less than 5% of cases occur in children and adolescents.[8]
Prevalence
- The prevalence of pyoderma gangrenosum is estimated to be of 0.01% among adult patients in the U.S. Incidence increases to 0.18% among patients with hidradenitis suppurativa and to 3.68% among patients with hidradenitis suppurativa and Crohn's disease.[1]
Risk Factors
- The three most common disorders associated with pyoderma gangrenosum include inflammatory bowel disease, arthritis, and myeloproliferative disorders.[2]
- Other diseases associated with pyoderma gangrenosum include ankylosing spondylitis, rheumatoid arthritis, sarcoidosis, chronic active hepatitis, monoclonal gammopathies, myeloma and hidradenitis suppurativa.[9][1]
- As many as 40-50% of cases of pyoderma gangrenosum may be idiopathic in nature.[9]
Etiology
The pathogenesis of pyoderma gangrenosum is poorly understood. Defects in neutrophil dysfunction involving chemotaxis or hyperactivity [10] as well as overexpression of interleukin-8 [11] and interleukein-16 have been reported as playing a role in the pathogenesis of this condition.[12][13] These observations suggest that pyoderma gangrenosum represents an overreactive inflammatory response to traumatic, inflammatory, or neoplastic processes in susceptible persons.[3]
Recent literature has indicated that drug-induced pyoderma gangrenosum is rare, however more cases are being reported. The pathogenesis of drug-induced pyoderma gangrenosum remains unclear. Most pyoderma gangrenosum lesions resolve after discontinuation of medication and in these case reports of induction after re-challenge with the same drug has been described.[14][15]
Pathophysiology
The pathophysiology of pyoderma gangrenosum is also not fully understood. Current pyoderma gangrenosum pathogenic hypothesis is that the disease is autoimmune in nature with defects in cell-mediated immunity, neutrophil and monocyte function and humoral immunity. A patient’s genetic background can lead to aberrant activation of innate-immune complexes (inflammasomes). The activated immune system leads to increased levels of dermal cytokines and resultant neutrophilic tissue infiltration. Other inducers of pyoderma gangrenosum include drugs such as granulocyte colony stimulating factor, isotretinoin, propylthiouracil and sunitinib. Cocaine has also been implicated as a common agent to trigger pyoderma gangrenosum.[16]
ASSESSMENT
If pyoderma gangrenosum is suspected, the patient will need a comprehensive assessment including history, physical exam and a biopsy that includes ulcer margin and subcutaneous tissue.[14]
- Primary goals of assessment are:
- To assess for significant signs, symptoms and histological patterns. See sections 'History and Physical Examination' and 'Biopsy'
- To screen for associated underlying diseases, which are present in up to 75% of cases (most frequently, inflammatory bowel disease, inflammatory arthritis, and hematological disorders) [14]
- To differentiate from other types of ulcers, which may require different treatments. See section 'Differential Diagnosis'.
- To determine whether clinical and histological findings meet diagnostic criteria for ulcerative (classic) pyoderma gangrenosum. See section 'Diagnosis'
- To determine whether the disease is limited or aggressive
See Algorithm for Assessment of Pyoderma Gangrenosum below (Algorithm 1)
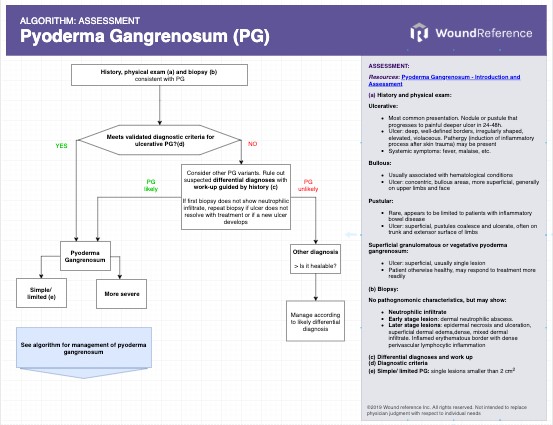
Algorithm 1. Assessment of Pyoderma Gangrenosum (click on this link to enlarge)
History and Physical Examination
Generally, the onset of pyoderma gangrenosum may follow either of two distinct clinical progression patterns [2]:
- In one of these clinical patterns, there may be sudden onset with rapid spread of lesions, clinically characterized by pain, systemic toxicity, fever, hemorrhagic pustules, suppuration and borders with an inflammatory violaceous halo. A band of erythema may extend from the wound edge, which defines the direction in which the ulcer will extend.
- A second clinical pattern begins with a slow and indolent onset with the presence of massive granulation within the ulcer margin, formation of crust and hyperkeratosis in the edges, large areas of skin involvement and evidence of spontaneous retrogression in certain areas and progression in others.
Clinical variants by morphologic subtype:
Pyoderma gangrenosum exhibits several clinical variants that include ulcerative, vesicular-bullous, pustular and superficial granulomatous (vegetative) forms.[17]
Classic ulcerative pyoderma gangrenosum
The most common presentation is classic ulcerative pyoderma gangrenosum that begins as a deep ulcer with a well-defined violaceous border that is irregularly shaped and elevated. The evolution of progression starts with a painful deep nodule or a sterile hemorrhagic superficial pustule that rapidly (within 24 to 48 hours) progresses to a necrotic ulcer.[18]
- Symptoms: Patients are often systemically unwell with symptoms of fever, malaise, arthralgia and myalgia.
- Ulcer characteristics (Figures 1 to 4): The surrounding skin is erythematous and indurated and the ulcers are deep, exudative, and extremely tender. The wound base is often filled with yellow slough with islands of necrosis and undermined wound edges. Lesions are usually severely painful and heal with cribriform (wrinkled paper) scar formation.
- Pathergy: can be defined as induction of inflammatory process after skin trauma. Pathergy occurs in 25-50% of cases of pyoderma gangrenosum, so elective surgery or debridement is usually contraindicated.[2] Insults to the skin that would be nearly meaningless for most patients can be devastating for patients with pyoderma gangrenosum.
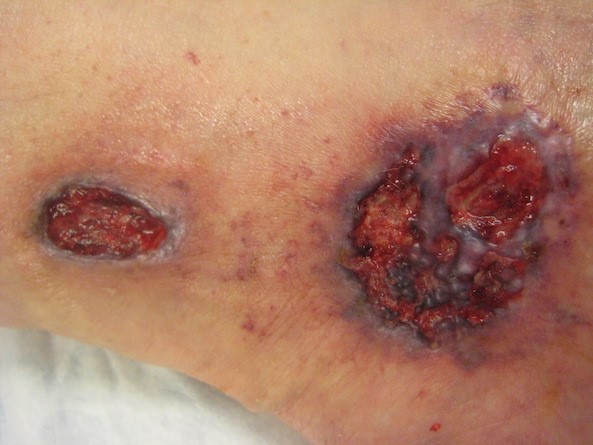 Fig. 1. Ulcerative pyoderma gangrenosum. Peripheral erythema, indurated, undermining margins Fig. 1. Ulcerative pyoderma gangrenosum. Peripheral erythema, indurated, undermining margins
| 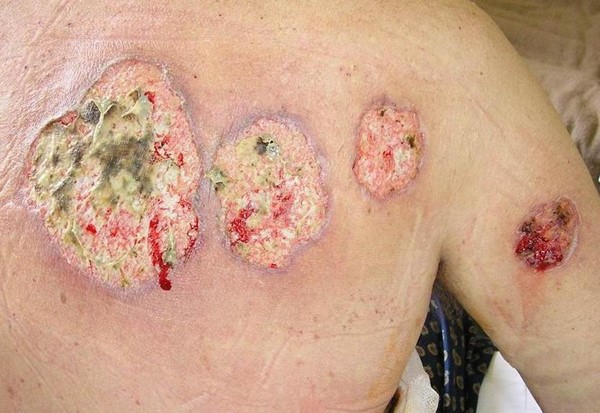 Fig. 2. Ulcerative pyoderma gangrenosum. Multiple lesions, wound base with yellow slough Fig. 2. Ulcerative pyoderma gangrenosum. Multiple lesions, wound base with yellow slough
|
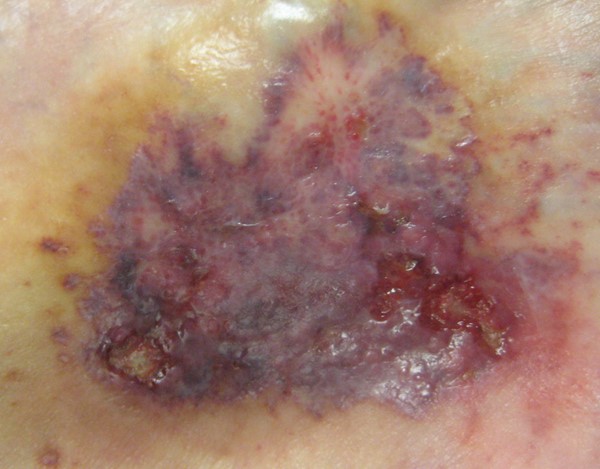
Fig. 3. Ulcerative pyoderma gangrenosum, early lesion | 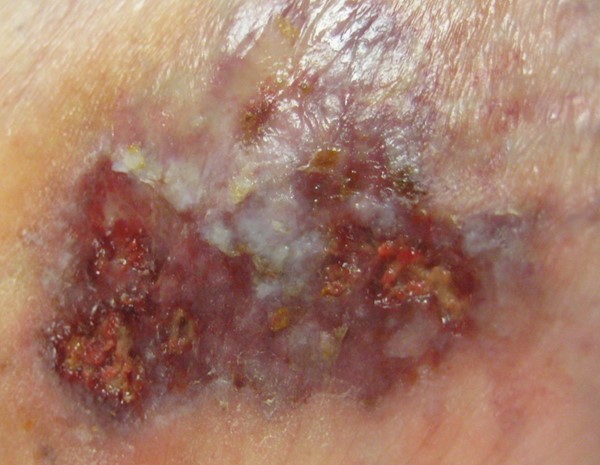
Fig. 4. Ulcerative pyoderma gangrenosum in figure 3 progressing to ulcer |
Bullous pyoderma gangrenosum
Bullous pyoderma gangrenosum is also a superficial variant that affects the upper limbs and face more than the lower extremities.
- Symptoms: It is mostly associated with hematological conditions.
- Ulcer characteristics: This form of the disease presents as concentric bullous areas that spread rapidly in a concentric pattern. The bullae rupture to form more superficial ulcers than those seen in classic ulcerative pyoderma gangrenosum but are characterized with a distinct violaceous border. Prognosis is poor because of the underlying hematological malignancy.[19][20]
pustular pyoderma gangrenosum
Pustular pyoderma gangrenosum is a rare superficial variant of the disease. This variant appears to be limited to patients with inflammatory bowel disease and tends to occur on the trunk and extensor surfaces of the limbs.[19][21][22]
- Ulcer characteristics: The process begins as a pustule or cluster of pustules that later coalesce and ulcerate. The process stops at the pustular stage and the patient may have painful pustular lesions that may persist for months. [19][21][22]
Superficial granulomatous or vegetative pyoderma gangrenosum
Superficial granulomatous or vegetative pyoderma gangrenosum also is a superficial form of the disease that seems to be less aggressive than other varieties. It is usually a single lesion in patients who are otherwise well and may respond to local treatment more readily than other forms of the disease. [19][21][22][23]
Clinical variants by location
extracutaneous pyoderma gangrenosum
The most common manifestation of extracutaneous pyoderma granulosum is pulmonary involvement, varying from asymptomatic to respiratory distress.[24] Findings of extracutaneous pyoderma gangrenosum include [24]:
- Fever, malaise, myalgia, arthralgia
- Spleen, liver, sclera, pulmonary, and kidney may have sterile granulomas, nodules, or abscesses
- Pulmonary interstitial fibrosis, cavitations, nodules, and pleural effusions
- Ulcerations of eyelid leaving permanent defects
Peristomal pyoderma gangrenosum
Peristomal pyoderma gangrenosum, which occurs close to abdominal stomas, comprises about 15% of all cases of pyoderma gangrenosum. Most of these patients have inflammatory bowel disease, but peristomal pyoderma gangrenosum can occur in patients who have had an ileostomy or colostomy for malignancy or diverticular disease.[25] In patients who have abdominal stomas, a 0.6% incidence of peristomal pyoderma gangrenosum has been reported.[25]
- Ulcer characteristics: The ulcers in these patients have a similar appearance to classic pyoderma gangrenosum but bridges of normal epithelium may traverse the ulcer base. Ulcers can be multiple, or coalesced.[26] The lesions are painful and often interfere with the stoma pouch adhering to the abdominal wall, which may irritate the skin more than usual.[25]
Post-surgical pyoderma gangrenosum
This clinical variant is rare, and arises in the surgical wound resembling a postoperative infection, thus diagnosis is often delayed.[27] Most common associated conditions among patients with post-surgical pyoderma gangrenosum following breast surgery have been reported to be malignancies (37%) and autoimmune deficiencies (17%). Microbiological examinations were found to have a negative result in 90%.[28]
- Ulcer characteristics: Resembles the classic pyoderma gangrenosum. Important clinical clues that help differentiate from surgical infection include irregular violaceous undermined border, mucopurulent discharge limited to the skin, bilateral involvement (in case of bilateral breast reconstruction), involvement of the abdominal wound, sparing of the mastectomy site, and relative sparing of the nipples and umbilicus.[29]
Clinical variants by associated comorbidities
Pyoderma gangrenosum also has a major clinical and diagnostic presentation in several auto-inflammatory syndromes including [30][31]:
- PAPA (pyogenic arthritis, pyoderma gangrenosum, and acne)
- PASH (pyoderma gangrenosum, acne, and suppurative hidradenitis)
- PAPASH (pyogenic arthritis, pyoderma gangrenosum, acne, and suppurative hidradenitis)
- PASS (pyoderma gangrenosum, seropositive spondylo-arthopathies, acne and suppurative hidradenitis)
- PsAPASH (psoriatic arthritis, pyoderma gangrenosum, acne, suppurative hidradenitis)
Diagnosis
Unfortunately, there are no specific laboratory findings or pathognomonic histological characteristics found in patients with pyoderma gangrenosum. This overlap in nonspecific histology and the variation in presentation has led to the belief that the diagnosis of pyoderma gangrenosum is a diagnosis of exclusion.[18] However, experts in the field have recently proposed that the diagnosis of the most frequent type, ulcerative pyoderma gangrenosum, be made more based on its clinical and pathologic features than based on exhaustive exclusion of other causes of ulceration.[4] Thus for all patients, it is recommended that a comprehensive history, physical exam, and biopsy be conducted.[32][33]
Diagnostic criteria:
- A set of diagnostic criteria for pyoderma gangrenosum was proposed in 2004, but to date have not yet been verified.[34] These diagnostic criteria require exclusion of other causes of cutaneous ulceration, which may be costly and impractical in the clinical setting.
- More recently, two new diagnostic criteria were created. Both attempt to de-emphasize the attempt to de-emphasize the need to exhaustively exclude other causes of ulceration and instead focus more on the clinical and pathologic features of the disease.[4] One of these sets of criteria is based on a scoring model in which criteria weight was determined by observed prevalence amongst pyoderma gangrenosum patients.[5] The other one was created with the Delphi method by an international expert committee and was validated against peer-reviewed established cases of pyoderma gangrenosum and mimickers.[32] It does not feature exclusion of other causes of ulceration as one of the required criteria for diagnosis of PG, which may be an advantage over the older diagnostic criteria described above. This new, validated criteria set includes 1 major criterion and 8 minor criteria.[32] For diagnosis, at least the major criterion and four minor criteria are required (see Table 1).
Table 1.Validated Diagnostic Criteria of Ulcerative Pyoderma Gangrenosum: A Delphi Consensus of International Experts [32]
For diagnosis of ulcerative pyoderma gangrenosum, at least the major criterion and four minor criteria are required
|
Major criterion: - Biopsy of ulcer edge demonstrating a neutrophilic infiltrate
|
Minor criteria: - History
- Pathergy (ulcer occurring at sites of trauma)
- Personal history of inflammatory bowel disease or inflammatory arthritis
- History of papule, pustule, or vesicle that rapidly ulcerated
- Clinical examination (or photographic evidence)
- Peripheral erythema, undermining border, and tenderness at site of ulceration
- Multiple ulcerations (at least 1 occurring on an anterior lower leg)
- Cribriform or “wrinkled paper” scar(s) at sites of healed ulcers
- Treatment
- Decrease in ulcer size within 1 mo of initiating immunosuppressive medication(s)
|
Biopsy:
Indications
- Biopsy of the ulcer is indicated in all potential pyoderma cases.[35] As the misdiagnosis rate of PG has been described to be as high as 10% [3], benefits of a biopsy outweigh the potential risk of pathergy due to biopsy.
- Biopsy of ulcer edge demonstrating a neutrophilic infiltrate is a required major diagnostic criterion for ulcerative pyoderma gangrenosum according to a recent international consensus.[32] However, it is important to note that biopsies taken after initiation of immunosuppressive therapy or during spontaneous resolution may fail to demonstrate a neutrophilic infiltrate. In those cases, it is important to obtain a new biopsy during subsequent flares or if the ulcer does not resolve with treatment.[32]
- As stated above, not all patients with PG develop pathergy and thus biopsy sites may heal uneventfully. However, if it is a concern, use of systemic corticosteroids (starting before the biopsy) to avoid pathergy has been reported.[35]
Procedure
- It is suggested that biopsies be taken at the advancing inflammatory margins of the pyoderma gangrenosum lesions, including uninvolved skin and be full thickness in depth, extending into the subcutaneous fat. [3][14][34]
- Elliptical incisional biopsy is preferable to punch biopsy. [3][34]
- Specimens including inflammatory margins should be sent for routine histopathologic examination (hematoxylin and eosin staining) and microbial stains (e.g. Gram, methenamine silver, or Fite).[34] Preferably, specimens should be evaluated by professionals with experience in dermatopathology. In addition, specimens from the ulcer edge should be sent for culture in appropriate medium to exclude bacterial, atypical mycobacterial and deep fungal infections.[3][34]
Pathology findings
- Although there are no pathognomonic histological characteristics in patients with pyoderma gangrenosum, the expert panel that devised the new validated diagnostic criteria strongly agreed that dermal edema with neutrophilic inflammation is a frequent histological finding in ulcerative pyoderma gangrenosum. [32] Absence of infection is frequently observed, but not required to diagnose pyoderma gangrenosum.[32]
- Histological features may also vary according to the stage of the disease [30]:
- An early lesion of pyoderma gangrenosum often demonstrates a dermal neutrophilic abscess. Vasculitis may occur with leukocytoclasia.
- Later stage lesions may show epidermal necrosis and ulceration, superficial dermal edema, and a dense, mixed dermal infiltrate that may extend to the subcutaneous tissue.[3]
- Direct immunofluorescence findings are neither sensitive nor specific for pyoderma gangrenosum. Thus, it is suggested that direct immunofluorescence be conducted when additional findings require exclusion of immuno-bullous disease, lupus, or vasculitis as potential causes of cutaneous ulceration.[34] Vascular deposition of IgM, C3, and fibrin can usually be found in the papillary and reticular dermal vessels of half of the biopsy specimens. IgG and IgA are occasionally present.[34]
Laboratory investigations:
In general, workup protocols are recommended in order to identify associated diseases and to exclude possible differential diagnoses.[34]
Clinicians might opt to start investigations with the studies in bold in Table 2 below and then proceed with investigations as needed in order to rule out suspected differential diagnosis, as guided by history. [14][34] Of note, no laboratory finding is diagnostic for pyoderma gangrenosum, but neutrophil leukocytosis and an elevated erythrocyte sedimentation rate are often seen in patients with pyoderma gangrenosum. [34]
Table 2. Pyoderma Gangrenosum Differential Diagnoses and Work-up (suggested studies for initial investigation in bold)
| Etiology | Conditions | Work-up |
|
Infection
|
- Deep fungal: blastomycosis, sporotrichosis
- Protozoa: leishmaniasis
- Bacterial: ecthyma
- Viral: herpes simplex
|
- Cultures
- Bacterial
- Fungal
- Atypical mycobacterial
- Chest X-ray (evaluates systemic involvement and possible infection prior to immunosuppressive therapy)
- Special stains
|
|
Vasculitis and autoimmune
|
- Behcet’s Vasculitis
- Cryoglobulinemia
- Antiphospholipid syndrome
- Lupus (lupus-associated neutrophilic dermatoses)
- Inflammatory bowel disease
|
- Antineutrophilic cytoplasmic antibodies (ANCA; to evaluate for granulomatous vasculitis as a cause of ulceration)
- Complete blood count (evaluates underlying hematological disorders)
- Erythrocyte sedimentation rate
- Comprehensive metabolic panel (liver and kidney function tests, glucose abnormalities prior to use of corticosteroids or immunosuppressant therapy)
- Coagulopathy panel (include antiphospholipid antibody screen, to evaluate for antiphospholipid syndrome as a cause of ulceration. Based on additional findings, assess other hypercoagulation states with cryoglobulins, Factor V Leiden, etc)
- Antinuclear antibody titer (ANA; assesses associated systemic lupus erythematosus or collagen vascular disorders)
- Colonoscopy (to assess inflammatory bowel disease as a cause of PG)
- Cryoglobulins (for cryoglobulinemia)
- Rheumatoid factor (to rule out cryoglobulinemia and rheumatoid arthritis
- Protein electrophoresis
- Direct immunofluorescence
- Urinalysis
- anti-DNA
|
| Neutrophilic | | |
|
Vascular
|
- Martorell ulcer
- Arterial
- Venous
|
- Deep elliptical biopsy
- Duplex scan
- Ankle-brachial pressure index (ABPI), doppler ultrasound and waveform analysis, angiography
|
|
Exogenous
|
- Factitious [36]
- Insect bite
- Bromoderma [37]
|
|
Differential Diagnosis
The misdiagnosis of pyoderma gangrenosum can have serious consequences. Treatment with high dose steroids or other immunosuppressive medications may be contraindicated in patients with many of the other diseases that may produce ulcerations resembling pyoderma gangrenosum. Patients suspected of having pyoderma gangrenosum often, upon further workup, prove to have a different cause for cutaneous ulcerations. The frequency of misdiagnosis of pyoderma gangrenosum has been shown to be as high as 10%. As many as 50% of patients who have had biopsies, have histopathological evidence of an alternative diagnosis on the initial or repeated biopsy.[3]
- Vascular occlusive disease, or venous disease, vasculitis, cancer, infection, exogenous tissue injury and other inflammatory disorders should be specifically ruled out (see Table 2 above and Table 3 below).
- When patients are treated for pyoderma gangrenosum and are refractory to treatment, an alternative diagnosis should be considered at which time repeat biopsies may be considered.
- Treatment directed at pyoderma gangrenosum can itself produce substantial complications or alternatively may result in temporary improvement and thus delay final diagnosis. Misdiagnosis of pyoderma gangrenosum in not uncommon and exposes the patient to substantial risks associated with its treatment.
Table 3. Some differential diagnoses of pyoderma gangrenosum [24]
| Findings | Pyoderma gangrenosum
| Granulomatosis with polyangiitis
| Polyarteritis nodosa
| Antiphospholipid syndrome
|
| Cutaneous | Classic ulcerative (most common), bullous, pustular, superficial granulomatous or vegetative pyoderma gangrenosum. See 'History and Physical Examination' | - Painful, palpable purpura with macules and papules, cystic nodules, indurated erythema.
- Commonly on legs
| - Painful nodules/purpura with indurated erythema.
- Livedo, petechia, ulcers can be present
- Commonly on legs
| - Livedo reticularis is common
- Superficial thrombophlebitis, skin ulcers ± necrosis, atrophie blanchie
|
| Laboratory | - Elevated ESR, CRP Leukocytosis
- Negative blood and tissue cultures
| - ANCA positive usually pr3 +/c-ANCA
- Elevated ESR and CRP
- Normocytic normochromic anemia indicating chronic disease
- Hematuria/proteinuria if renal involvement
| - Negative for ANCA, RF, cryoglobulins ANA (+), lupus anticoagulant (+)
- Elevated ESR and CRP, y-globulin
- Normocytic normochromic anemia indicating chronic disease
- Elevated IgM anti-phosphatidylserine–prothrombin complex
- Anti-streptolysin O antibody (+)
- HBV, HCV, HIV associations
| - Anti-cardiolipin antibody, lupus antico-agulant, anti-Beta2-glycoprotein I antibody (IgG or IgM)
- Hemolytic anemia, thrombocytopenia, and prolonged PT/PTT
- Phosphatidylethanolamine antibodies, annexin A5 resistance assay
|
Documentation
- The American Medical Association (AMA) recommends that ulcers be properly documented for adequate code (ICD-10) assignment. That means that clinicians should clearly describe etiology, anatomical location with laterality and severity of tissue damage in a way that coders can easily match descriptions of existing ICD-10 codes to the medical record.
Other items that need to be adequately documented include:
- For ulcer healing assessment purposes, documentation should include etiology, number and position of ulcers, size, depth, severity, description of wound edge, peri-wound area, wound base quality, amount and type of drainage, and infection, history of debridement.
- Signs of ulcer improvement: It is critical to assess healing progress, as it determines whether a treatment plan should be continued or not. Thus, ulcer healing progress should be recorded weekly or sooner if significant change is observed.
- Documenting signs of ulcer improvement to support medical necessity (Medicare):
- Reimbursement for wound care services on a continuing basis for a particular wound in a patient requires documentation in the patient's record that the wound is improving in response to the wound care being provided.
- Medicare expects that with appropriate care wound should demonstrate signs of improvement. Such evidence must be documented with each date of service provided.
- Evidence of improvement includes measurable changes (decreases) of some of the following:
- Drainage
- Inflammation
- Swelling
- Pain
- Wound dimensions (diameter, depth)
- Necrotic tissue/slough
- It is not medically reasonable or necessary to continue a given type of wound care if evidence of wound improvement cannot be shown. Medicare expects the wound-care treatment plan to be modified in the event that appropriate healing is not achieved.
- Tools that facilitate standardized assessment and documentation should be used whenever possible:
- Wound Reference Wound Prep&Dress Tool creates notes to help support medical necessity that can be copied and pasted to electronic medical records
- Validated wound assessment tools such as Bates-Jensen Tool [38]
- Wound imaging: digital photographs of the ulcer at the first consultation and periodically thereafter to document progress is helpful and ensures consistency of care among healthcare practitioners, facilitates telemedicine in remote areas, and illustrates improvement to the patient.
- Wound measurement: methods include manually measuring length and width (the longest length with the greatest width at right angles), manual tracing, digital photography, and software programs that calculate wound dimensions from a photograph of the lesion. Wound tracings that calculate the area via digital software are slightly better than linear measurement [39].
- Patient reported outcome (PRO) Tools: Validated instruments allow clinicians to obtain feedback on whether a given treatment plan is resulting in the best outcomes possible and is improving patient's quality of life (QOL). PRO assessment will continue to gain importance as health care systems transition from a volume-based reimbursement model to a value-based model.[40]
- QOL tools validated for pyoderma gangrenosum patients: Dermatology Life Quality Index (DLQI) [41]
- ? Medicare Quality Payment Program, Improvement Activity "Promote Use of Patient-Reported Outcome Tools" suggests use of Wound-Quality of Life (QoL) and patient-reported Wound Outcome.[42]
- Pain measurement:
- ? Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"
CODING
- Identify and document first any documented underlying condition (ICD-10-CM documentation)
- ICD-10: L88 - Pyoderma gangrenosum
- Specify laterality of ulcer
- Right, left or unspecified
- Specify ulcer severity of ulcer
- Limited to breakdown of skin
- With fat layer exposed
- With necrosis of muscle
- With necrosis of bone
- Unspecified severity
Diagnostic Related Group (DRG)
- ICD-10-CM L88 is grouped within Diagnostic Related Group(s) (MS-DRG v35.0):
- 602 Cellulitis with mcc
- 603 Cellulitis without mcc