ABSTRACT
Background:
- By default, chronic wounds are contaminated by several types of bacteria.[1][2] When the host (patient) does not adequately respond to bacterial contamination, this contamination can turn into colonization, which can further turn into infection. A wound culture is indicated when wound infection is suspected.[3]
-
Wound culture: wound culture is a standard microbiological analysis of a sample of tissue or fluid taken from the wound bed. It is used to determine the type and quantity of microorganisms in a wound.[4] After collection, the specimen is placed in a sterile container for transportation to the laboratory. In the laboratory, the sample is placed in a substance that promotes growth of organisms and the type and quantity of organisms that grow are assessed by microscopy.
Indications:
- Among other specific indications, a wound culture is warranted for:
-
- Chronic wounds with signs of spreading or systemic infection
- Chronic wounds with signs of local wound infection and/or delayed healing, especially in patients with compromised immune competency, such as those taking immunosuppressants or corticosteroids, or with diabetes mellitus or peripheral arterial disease, who often do not show classic signs of infection
Wound Culture Techniques:
- Sampling techniques for standard microbiological analysis (i.e. wound culture) include swabbing the wound bed, needle aspiration and deep-tissue biopsy.
Evidence and Recommendations:
-
2CFor patients with wounds, expert consensus suggests that wounds be cultured only if a proper indication is present, as described in the section 'Indications'.(Grade 2C)
- 2C
When a wound infection is first suspected, the IWII consensus suggests clinicians collect a wound swab using the Levine technique. For wounds with antibiotic-resistant species, and to determine the effect of antimicrobial intervention, a wound biopsy is preferred.(Grade 2C)
This topic provides evidence-based guidance on when and how to perform wound cultures, how to choose sampling techniques, documentation, coding, coverage and reimbursement.
Wound infection has been regarded as the one of the most important causes of delayed healing of chronic wounds.[5] On the other hand, overuse and inappropriate use of antibiotics and/or topical antimicrobial agents on non-infected wounds may lead to antimicrobial resistance and unnecessary adverse effects. Both sides of the spectrum can lead to increased utilization of resources.[6]
Identification of infection in a chronic wound is often challenging. As a result, variations in practice related to which culture method to use and when/how to initiate treatment are common among clinicians. [1]
This topic provides evidence-based guidance on when and how to perform wound cultures, how to choose sampling techniques, documentation, coding, coverage and reimbursement.[1][3] Other diagnostic modalities for wound infection (e.g. laboratory markers, imaging, and emerging diagnostic techniques) will be covered in a separate topic (coming soon). For sample policies and procedures on how to collect a wound swab see topic "How to Collect a Wound Swab (Levine Technique) for Culture". For guidance on when and how to perform wound biopsies see topic "How to Perform a Wound Biopsy".
Table 1. Common species recovered from wounds [2]
Aerobic and Facultative Anaerobic
|
Anaerobic
|
- Staphylococcus aureus: is most frequently isolated
- Pseudomonas aeruginosa
- Escherichia coli
- Enterobacter cloacae
- Klebsiella species
- Streptococcus species
- Enterococcus species
- Proteus species
|
- Peptostreptococcus
- Prevotella
- Porphyromonas
- Bacteroides,
- Finegoldia magna
- Peptoniphilus asaccharolyticus
|
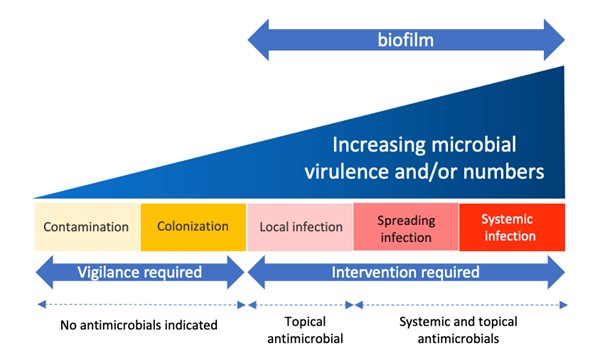
Figure 1. The wound infection continuum - click on picture to enlarge (based on the International Wound Infection Institute) [4]
Table 2: Signs and symptoms associated with stages of the wound infection continuum (based on the International Wound Infection Institute) [4]
| Contamination |
Colonization |
Local infection, covert (subtle) signs |
Local infection, overt (classic) signs |
Spreading infection |
Systemic infection |
- All wounds may acquire microorganisms.
- If suitable nutritive and physical conditions are not available for each microbial species, or they are not able to successfully evade host defenses, they will not multiply or persist; their presence is therefore only transient and wound healing is not delayed
|
- Microbial species successfully grow and divide, but do not cause damage to the host or initiate wound infection
|
- Hypergranulation (excessive ‘vascular’ tissue)
- Bleeding, friable granulation
- Epithelial bridging and pocketing in granulation tissue
- Wound breakdown and enlargement
- Delayed wound healing beyond expectations
- New or increasing pain
- Increasing malodor
|
- Erythema
- Local warmth
- Swelling
- Purulent discharge
- Delayed wound healing beyond expectations
- New or increasing pain
- Increasing malodor
|
- Extending in duration +/- erythema
- Lymphangitis
- Crepitus
- Wound breakdown/ dehiscence with or without satellite lesions
- Malaise/lethargy or nonspecific general deterioration
- Loss of appetite
- Inflammation, swelling of lymph glands
|
- Severe sepsis
- Septic shock
- Organ failure
- Death
|
General Indications and Contraindications
Indications
A wound culture is indicated when wound infection is suspected.[3] According to the International Wound Infection Institute (IWII) consensus, collection of a wound specimen for standard microbiological analysis is indicated for [4]:
- Acute wounds with classic signs and symptoms of infection (See Table 2 above)
- Chronic wounds with signs of spreading or systemic infection (See Table 2 above)
-
- In individuals showing signs of sepsis, blood cultures are also indicated, and other likely sites of infection should be considered for sampling
- Chronic wounds with signs of local wound infection and/or delayed healing, especially in patients with compromised immune competency, such as those taking immunosuppressants or corticosteroids, or with diabetes mellitus or peripheral arterial disease, who often do not show classic signs of infection (See Table 2 above)
- Infected wounds that have failed to respond to antimicrobial intervention, or are deteriorating despite appropriate antimicrobial treatment
- To be in compliance with local protocols for the surveillance of drug-resistant microbial species
- Wounds where the presence of certain species would negate a surgical procedure (e.g. beta hemolytic streptococci in wounds prior to skin grafting)
Practical aspects to consider:
- Subtle changes in the wound may indicate infection. Signs and symptoms of infection in chronic wounds can be subtle, therefore changes in the wound reported by the patient at weekly visits can be indicative of infection (e.g. increased odor, exudate, rubor, pain, etc).
- Collection of specimen prior to institution of antibiotic therapy is preferred.
- For patients or caregivers with a higher level of anxiety, a heightened level of awareness of subtle signs and symptoms of infection will help determine when a wound culture should be collected.
Contraindications
Wound cultures should not be undertaken routinely or without substantial cause (e.g. no signs of infection or delayed healing). Exceptions include the need to meet local protocols that require screening of drug-resistant organisms, or if the presence of certain species would negate a surgical procedure.[4]
Contraindications include:
- Inability to transport a specimen within 4-24 hours from collection.[4][9][10]
- Specimens collected from fresh bites, as infectious agents may not be recovered.[10]
- Wounds covered with slough, debris and necrotic tissue. Cleansing and debridement should be completed before specimen collection, so as to recover species at and below the wound surface.[4]
- Previous wound culture performed within the last 24-72 hours. The usual laboratory turnaround time for wound cultures can take up to a week (3-5 days for aerobic, 4-7 days for anaerobic culture)[9][11]
Wound Culture Techniques
Sampling techniques for standard microbiological analysis through wound culture include wound bed swabs, needle aspiration and deep-tissue biopsy (Figure 2).[3][4] These sampling techniques for wound culture are described below and in Table 3:
Wound swab
-
Indication: for initial monitoring of chronic wounds with suspected infection, the IWII consensus suggests obtaining a wound specimen for culture using a wound swab and the Levine technique.[4]
-
Description of the technique: wound swabs are minimally invasive, easier to perform and widely employed in clinical practice, but techniques vary.[12][13] Swab results are usually reported as the number of organisms per swab. Comparative studies show that the Levine technique is superior to the Z-swab technique for both acute (P≤ 0.001) and chronic wounds (P≤ 0.001).[1][4][12][13]
-
- The Levine technique involves rotating the wound swab over a 1cm2 area of the wound.[13] See topic "How to Collect a Wound Swab (Levine Technique) for Culture".
- The Z technique involves rotating the swab between the fingers in a zigzag fashion across the wound without touching the wound edge [13]
- Wound cleansing without antimicrobials and debridement should be completed before collecting a wound swab.[4]
- The swab should be placed in a bacterial swab transport (e.g. ESwab, Amies Agar gel transport media).[14]
-
- Some swab transports may have ESwab. ESwab is a liquid-based, multipurpose open platform, collection and preservation system that maintains viability of aerobic, anaerobic and fastidious bacteria for up to 48 hours at refrigerator and room temperature.[14]
- Advantages: practical, noninvasive, and inexpensive.
-
Disadvantages: wound swabs collect only the surface-colonizing bacteria and not the pathogenic strains invading deeper tissues.[3] Wound swabs often do not detect biofilm infection.[3] Laboratories usually prefer tissue or aspirate as specimens for microbiological wound culture as they yield more microbes [15], however those specimens are not as available or practical to obtain as wound swabs.
-
Accuracy of wound swab in detection of wound infection: when compared with specimens collected by biopsy, sensitivity and specificity of the swab culture collected with Levine technique range from 80-90% and 54-57%, respectively.[16][17] That is, a wound culture of a swab collected with the Levine technique correctly identifies 80-90% of infected chronic wounds, but it misses 10-20%. On the other hand, the test can correctly confirm that ~50% of non-infected wounds are not infected indeed, but the test is incorrect about 50% of the time.[18]
Tissue biopsy
-
Indication: a tissue biopsy for C&S is indicated for wounds with antibiotic resistant species and to determine the effect of antimicrobial intervention.[4][12]
-
Description of the technique: a tissue biopsy is an aseptic technique for obtaining a wound tissue sample for qualitative and quantitative culture.[19]
-
-
Skin biopsy: According to the AMA CPT Codebook guidelines, there are three distinct techniques for skin biopsies: tangential, punch and incisional biopsies.[20][21] For details, see topic "How to Perform a Wound Biopsy".
-
Tissue processing: at the laboratory the sample is sterilely processed (e.g. vortexed, homogenized, etc) to extract bacteria, and the resulting liquid is cultured and incubated aerobically or anaerobically.[22] Quantitative biopsy results are usually reported as the number of organisms per gram of tissue.
-
Advantages: tissue biopsy for C&S is considered the gold standard for identifying wound infection, as results are more conclusive and accurate compared to other tests. [3][23]
-
Disadvantages: despite advantages, biopsies are relatively invasive, costly, require skilled operators, potentially exacerbate infection, and involves extensive processing techniques that can be difficult for most clinical microbiology laboratories.[3][12][15] As a result, tissue biopsies are usually not the first choice of specimen collection technique, unless surgical debridement is required and an intraoperative sample can be retrieved.[24] A tissue biopsy is indicated in the presence of antibiotic resistant species and to determine the effect of antimicrobial intervention.[4][12]
-
Accuracy of a tissue biopsy in detection of wound infection: moderate certainty evidence show that tissue biopsies (e.g. punch biopsies) provide qualitative and quantitative information about the bacterial load and tissue reaction with nearly 100% sensitivity, 90% specificity and 95% accuracy for predicting wound closure.[12]
Needle aspiration
-
Indication: needle aspiration is indicated when collections of tissue fluid or pus are present. [25][15][4]
-
Description of the technique: for wounds with fluid or purulent exudate, the specimen can be aspirated with a 22 gauge needle attached to a 10 cc syringe. The sample is collected by pulling back on the plunger to create pressure, and positioning the tip of the needle in different areas of the wound [25]
-
- Aspirate fluid should be submitted in a sterile tube (and not inoculated into a wound swab).[15] Purulent material is transferred to a sterile tube or anaerobe transport media.
- When incision and drainage are performed, a sample of the advancing margin or base of the infected lesion may also be collected and submitted for analysis.
-
Advantages: needle aspirates are one of the preferred types of specimens for wound culture by microbiological laboratories (along with biopsy samples).[15]
-
Disadvantages: this technique allows for sampling only of a limited portion of the wound. Needle aspirates may underestimate the number of bacterial isolates compared to tissue biopsy specimens. Also, there is risk of spreading bacteria to non-infected areas by having the needle enter those areas.[12]
-
Accuracy of needle aspiration in detection of wound infection: some studies have compared the needle aspiration technique with quantitative tissue biopsy and swab cultures. One study reports a 81.7% sensitivity of fine needle aspiration in detection of surgical wound infection.[24] However, due to methodological limitations, sensitivity, specificity, and accuracy of quantitative needle-aspiration remain unclear.[25]
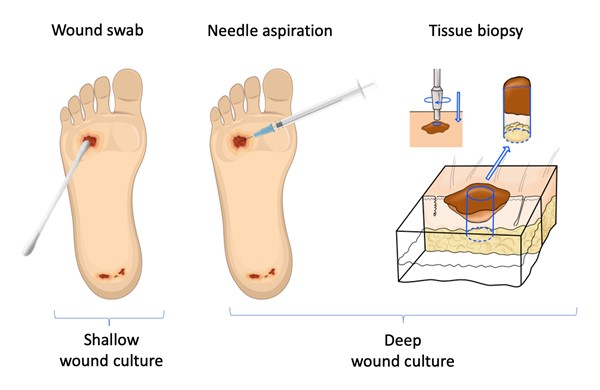
Figure 2. Wound sampling techniques for wound culture
Table 3. Comparison of sampling techniques for wound culture
|
Wound Swab |
Tissue Biopsy |
Needle Aspiration |
| Indication |
For initial monitoring of chronic wounds with suspected infection [4]
|
For wounds with antibiotic resistant species and to determine the effect of antimicrobial intervention [4][12]
|
When collections of tissue fluid or pus are present.[4][15][25]
|
| Advantages |
Practical, noninvasive, and inexpensive
|
Gold standard for identifying wound infection [3][23]
|
One of the preferred types of specimens for wound culture by microbiological laboratories (along with biopsy samples)[15]
|
| Disadvantages |
Can collect only the surface-colonizing bacteria and not the pathogenic strains invading deeper tissues.[3] Wound swabs often do not detect biofilm infection.[3]
|
Relatively invasive, costly, require skilled operators, potentially exacerbate infection.[3][12][12][15]
|
Allows for sampling only of a limited portion of the wound. Risk of spreading infection to non-infected areas.
|
| Accuracy |
Compared with specimens collected by biopsy, sensitivity and specificity of the swab culture collected with Levine technique range from 80-90% and 54-57%, respectively.[16][17]
|
Nearly 100% sensitivity, 90% specificity and 95% accuracy for predicting wound closure.[12]
|
Unclear [25]
|
Limitations
Many pathogenic microorganisms cannot be detected by standard microbiological analysis (i.e aerobic C&S).[4] Those include anaerobic bacteria, fastidious bacteria, samples that were collected after prolonged antibiotic treatment, unculturable bacteria and viable but non-culturable bacteria (VBNC).[26] VBNC bacteria present measurable metabolic activity, but do not grow in conventional culture medium like normal bacteria.[27]
To address limitations of standard microbiological analysis, scientists have been developing molecular and imaging techniques that can more precisely identify species of microbes in a wound specimen.
-
Molecular techniques include nucleic acid amplification techniques such as polymerase chain reaction (PCR), DNA sequencing and typing, direct molecular probe techniques (in situ hybridization), and nucleic acid quantitation.[28]
-
- Broad-range 16S rRNA gene-specific PCR assay has been shown to be useful in identifying pathogenic microorganisms in polybacterial infections or when fastidious, unculturable or VBNC bacteria are present in the sample.[29] A study that analyzed 97 specimens (pus or swabs) collected from patients with clinical signs of surgical site infection but "no bacterial growth" in conventional wound culture showed that the PCR assay identified the presence of bacterial pathogen in 53 (54.63%) cases, including VBNC bacteria, obligatory anaerobes, unculturable bacteria and poly bacterial infections.[29]
- An example of imaging technique is the use of fluorescence imaging to detect higher bacterial loads (Figure 3). These emerging technologies will be covered in another topic (coming soon).
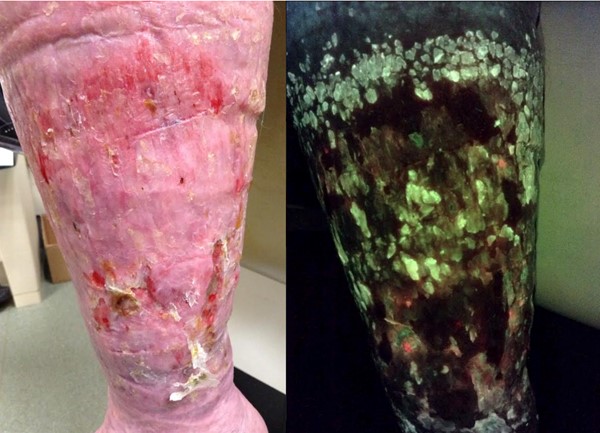
Figure 3. Venous leg ulcers on the left leg (left). Fluorescence imaging (right) of the same ulcers allows clinicians for detect elevated bacteria loads* (>104 CFU/g). By Cathy Milne, APRN, MSN, CWOCN-AP
Wound Culture Risks and Complications
-
Wound swab: if debris, slough and necrotic material are present on the wound debridement is recommended, which may result in bleeding and other complications. See topic "How to Perform Conservative Sharp Wound Debridement".
-
Deep tissue biopsy: there is risk of postsurgical trauma, wound disruption, and bacteremia.[3]
-
Needle aspiration: may extend infection by having the needle enter uninfected areas.[12]
Wound Culture: Evidence and Recommendations
- 2CFor patients with wounds, the IWII consensus suggests that wounds be cultured only if a proper indication is present, as described in the section 'Indications' above.(Grade 2C) [1][4]
-
- Rationale: all chronic wounds are contaminated with bacteria [1], therefore positive culture results might not be clinically significant in the absence of an appropriate indication. For chronic wounds, evidence suggests that a potential infection be considered early in the presence of subtle (overt) signs of infection. See signs and symptoms associated with stages of the wound infection continuum in Table 2 above.
-
2CWhen a wound infection is first suspected, the IWII consensus suggests clinicians collect a wound swab using the Levine technique. For wounds with antibiotic-resistant species, and to determine the effect of antimicrobial intervention, a wound biopsy is preferred.(Grade 2C) [1][4]
-
-
Rationale: wound swab is the most common technique of specimen collection for wound culture, as it is practical, non-invasive and less costly. [1][3][12] Among the methods for wound swab collection, the Levine method has been shown to be superior to others, as it detects more organisms per swab.[1][12] Because of its superior effectiveness and accuracy, wound biopsy is considered the gold standard for wound culture.[12] However, wound biopsies are relatively invasive, costly, require skilled operators and potentially exacerbate infection. As a result, wound biopsies are not routinely performed in practice.[4][12]
How to Select a Wound Culture Technique
- For initial monitoring of chronic wounds with suspected infection, the IWII consensus suggests obtaining a wound specimen for culture using a wound swab and the Levine technique.[4][12]
- For wounds with antibiotic resistant species and to determine the effect of antimicrobial intervention, literature suggests a tissue biopsy of the wound for quantitative analysis.[4][12]
-
- In clinical practice, whenever a bone debridement is performed and bone is removed, the sample should be cultured if possible. In other scenarios, wound biopsies might not be as common/practical due to cost, availability of services and discomfort to the individual.[4]
- Where pus is present it should be collected directly by syringe or swab.[4]
- For patients with clinical signs of infection but "no bacterial growth" in conventional wound culture, clinicians might opt for PCR assays (DNA sequencing) to identify the presence of bacterial pathogen.[29]
- Because standard techniques have limitations that prevent many microorganisms from being cultured, new molecular and imaging technologies have been developed but those are not yet widely available (e.g. PCR assays, DNA sequencing techniques).[4] DNA sequencing techniques can more precisely identify microbial species in a wound specimen, including microbes not identified by culture-based techniques.[4]
How to Collect Specimens for Wound Culture
-
See topics "How to Collect a Wound Swab (Levine Technique) for Culture" and "How to Perform a Wound Biopsy".
Prescribing/ Ordering Wound Cultures
- In the United States, an order should be obtained from a provider prior to performing a wound C&S. [30]
Types of tests for culture
- Tests that can be ordered for wounds include include aerobic or anaerobic C&S, Gram stain, fungal, mycobacteria.
- For open wounds, the default test is aerobic C&S. When not specified by the provider, laboratories will usually perform an aerobic C&S.
- If anaerobic, fungal, and mycobacterial pathogens are suspected, appropriate cultures should be requested if clinically indicated.
-
-
Anaerobic C&S is indicated if the following soft tissue infections are suspected: anaerobic cellulitis, necrotizing fasciitis, clostridial myonecrosis (gas gangrene), anaerobic streptococcal myositis or myonecrosis, synergistic nonclostridial anaerobic myonecrosis, infected vascular gangrene, infection resulting from human or other bite. These infections, particularly clostridial myonecrosis, necrotizing fasciitis, and nonclostridial anaerobic myonecrosis, may be fulminant and are frequently characterized by the presence of gas and foul-smelling necrotic tissue.[11]
- Culture of biopsied bone can be useful in establishing the diagnosis of anaerobic osteomyelitis when suspected.[11][31]
- Susceptibility studies: these tests are performed if pathogens are isolated in culture. Results of the testing are reported as “susceptible” (likely, but not guaranteed to inhibit the pathogenic microorganism), “intermediate” (may be effective at a higher than normal concentration), and “resistant” (not effective at inhibiting the growth of the organism). If there is more than one pathogen, the laboratory will report results for each one. The test results should be used to guide antibiotic choice. The results of antimicrobial susceptibility testing should be combined with clinical information and experience when selecting the most appropriate antibiotic for the patient.[7]
Table 4 provides a summary on tests commonly performed for wound culture.
Table 4. Common tests for wound culture [9][10][11][32]
| Test |
Description, Specimen and Limitations |
|
Aerobic culture (CPT: 87070)
|
- Description: Isolation and identification of aerobic pathogens. If pathogens are isolated, drug susceptibility tests may be performed.
- Specimen: swab stained with wound exudate in a bacterial swab transport or ESwab; 0.5 ml of aseptically obtained pus or tissue in a sterile screw-cap container; tissue must be kept moist (e.g. in sterile saline).
- Limitations: only rapid-growing, nonfastidious aerobic organisms (i.e. bacteria that do not have complex nutritional requirements) can be recovered and identified by routine methods. Only organisms that predominate will be identified. Unless specifically requested by the physician, fastidious organisms may not be isolated. The procedure will not detect Chlamydia, viruses, fungi, or mycobacteria.
|
|
Anaerobic culture (CPT: 87075)
|
- Description: Isolation and identification of anaerobic pathogens. If pathogens are isolated, drug susceptibility tests may be performed.
- Specimen: aspirates or tissue are preferable to swabs. Place 0.5 mL pus, or other fluid or tissue from aspirated site in anaerobic transporter. If swabs are used, place swab in anaerobic transporter or ESwab transport. Some anaerobes will be killed by contact with molecular oxygen for only a few seconds.
|
Gram stain (CPT: 87205)
|
- Description: laboratories recommend collecting a separate specimen from the same site as the culture for Gram stain, to facilitate interpretation. The quality of the specimen and determination of extent of workup is often based on Gram stain results.
- Specimen: a thin, air-dried smear, or if the specimen is collected with swabs, obtain one swab for culture and one for Gram stain, or use ESwab transport.
|
|
Fungal culture (CPT: 87102)
|
- Specimen:
-
- Biopsy: Surgical specimen in sterile container. A small amount of sterile nonbacteriostatic water should be used to prevent drying.
- Wound: Purulent material, fluid, scraping of lesion border, or swab (least preferred) in sterile transport container. Swabs cannot be split for other tests.
|
|
Acid-Fast Bacteria Culture (for mycobacteria, CPT: 87116)
|
- Specimen: skin biopsy is recommended. Contact your laboratory for collection instructions.
-
- Skin biopsies of cutaneous lesions to identify acid-fast staining bacilli and cultures represent the cornerstone of diagnosis of cutaneous mycobacterial infection.[33] Additionally, histopathological evaluation of skin biopsy specimens may be useful in identifying leprosy, Buruli ulcer, and cutaneous tuberculosis.[33]
|
Considerations when ordering wound culture
- If there are any questions on the type of specimen to be collected, and on the type of test to be ordered, it is important that clinicians seek advice from their local microbiologist and pathologist.
- Characterization of microbial flora takes at least 24 hours. Testing for fungi and anaerobic bacteria requires additional investigations and processing.[34]
- When rapid investigation is required (e.g. in cases of sepsis) a blood culture may yield results within 4 hours, or microscopic examination of specimens by more specialized laboratory staff may guide antimicrobial therapy faster.[34]
Documentation Requirements
Documentation should include:
- Wound assessment and characteristics (e.g. appearance, exudate, signs of infection)
- Medical record documentation supporting medical necessity (i.e., for Medicare beneficiaries, CMS requires use of at least one of the ICD-10-CM code(s) that support medical necessity. A sample list is provided by the Local Coverage Article: Billing and Coding: Susceptibility Studies (A57176)).[30]
- A wound C&S order from the treating physician/nonphysician practitioner (as required by CFR, Title 42, Volume 2, Chapter IV, Part 410.32(a) Ordering diagnostic tests)[30]
- Description of the specimen collection procedure, patient's tolerance to the procedure and response to analgesia (when used)
- Labeling of the specimen container and completion of the laboratory requisition. The following documentation should be included [34]:
-
- Patient identification
- Date and time of collection
- Information on the patient: demographics and relevant comorbidities
- Information on the wound: anatomical location, duration and etiology, any suspected unusual organisms (e.g. Nocardia)
- Clinical indication for the wound sample: signs and symptoms, and suspected microbes
- Current or recent antibiotic use
- Identification of the person who collected the specimen
Examples of documentation that may be requested for medical review of claims for laboratory tests are [35]:
- Clinical evaluations, physician evaluations, consultations, progress notes, physician’s office records, hospital records, nursing home records, home health agency records, records from other healthcare professionals and test reports. This documentation is maintained by the physician and/or provider.
CODING COVERAGE AND REIMBURSEMENT
Medicare Administrative Contractors and Local Coverage Determinations
Medicare coverage of provider and facility fees related to wound cultures is managed by Medicare Administrative Contractors (MAC), under Medicare Part A or Part B. Each jurisdiction may have its own specific local coverage determination and policies. See topic "Medicare Coverage Determinations for Wound Care".
CPT codes
Wound swab
- There is no CPT code for the swab of a wound, it is included in the evaluation and management (E/M) charge. Adequate documentation of the procedure will help justify a higher level of E/M. See topic "2021 Office/Outpatient E/M Services Updates for Wound Care and HBOT".
- If conservative sharp wound debridement is performed before collecting a wound swab, this procedure may be billed instead of the E/M. See topic "Coding and Billing Essentials in Wound Care".
Biopsy of Skin
- A specimen can be divided and sent for C&S and histopathological analysis. For a skin biopsy CPT to be used, the specimen needs to be sent for histopathological analysis.
- Biopsy of the skin is reported under three distinct techniques: tangential, punch, or incisional.[20]
- See CPT codes for skin and bone biopsies in the Appendix - CPT Codes.
APPENDIX
Summary of Evidence (SOE)
Wound Culture
(back to text)
Systematic reviews
- A 2016 systematic review found moderate quality evidence that punch biopsies provide qualitative and quantitative information about the bacterial load and tissue reaction with nearly 100% sensitivity, 90% specificity and 95% accuracy for predicting wound closure. However, biopsies are relatively invasive, costly, require skilled operators, and potentially exacerbate infection. Wound swabs are minimally invasive, easier to perform and widely employed in clinical practice, but techniques vary. In comparative studies, the Levine technique was superior to the Z-swab techniques, and biopsies were more sensitive for antibiotic-resistant wounds than Levine or Levine-like swabs, suggesting that swabs may be useful for initial wound monitoring, but biopsies are preferred when antibiotic resistance is suspected.[12]
- A 2018 literature review evaluated 4 articles that compared the Levine and the Z swab techniques. Overall, results revealed that the Levine technique detected more organisms than the Z-technique.[1][13][16] Comparative studies show that the Levine technique is superior to the Z-swab technique for both acute (P≤ 0.001) and chronic wounds (P≤ 0.001).[1][4][12][13]
Glossary of Terms
Selected terms, as per the International Wound Infection Institute [4]
| Terms and Definitions |
- Aerobe: An organism that requires the presence of oxygen in its environment in order to survive and multiply
- Anaerobe: An organism that can survive and multiply in the absence of oxygen in its environment. Some bacteria are classified as facultative anaerobes as they can sense concentration of oxygen in their environment and adjust their metabolism accordingly.
- Antimicrobial: A substance that acts directly on a microbe in a way that will either kill the organism or significantly hinder development of new colonies. The term incorporates disinfectants, antiseptics and antibiotics. Antimicrobial therapy may be required when other methods of eradication of wound infection are insufficient to manage localized wound infection, or when the infection is systemic/spreading.
- Antibiotics: A small natural or synthetic molecules that have the capacity to destroy or inhibit bacterial growth. Antibiotics target specific sites within bacterial cells while having no influence on human cells, thus they have a low toxicity. They may be administered systemically or in topical preparations. Antibiotic resistance is a major global health concern.
|
CPT Codes
Skin biopsy
Skin biopsy CPT codes [20]
| CPT Codes |
Description |
11102
|
Tangential biopsy of skin (e.g., shave, scoop, saucerize, curette), single lesion
|
+11103
|
Each additional lesion
|
11104
|
Punch biopsy of skin (including simple closure, when performed), single lesion
|
+11105
|
Each additional lesion
|
11106
|
Incisional biopsy of skin (e.g., wedge; including simple closure, when performed), single lesion
|
+11107
|
Each additional lesion |
Bone biopsy
Bone biopsy CPT codes [20]
| CPT Codes |
Description |
20220
|
Biopsy, bone, trocar, or needle; superficial (eg, ilium, sternum, spinous process, ribs
|
20225
|
Biopsy, bone, trocar, or needle; deep (eg, vertebral body, femur)
|
20240
|
Biopsy, bone, open; superficial (eg, sternum, spinous process, rib, patella, olecranon process, calcaneus, tarsal, metatarsal, carpal, metacarpal, phalanx)
|
20245
|
Biopsy, bone, open; deep (eg, humeral shaft, ischium, femoral shaft) |