For full access to this topic and more
premium content, upgrade today. Or get started with a Free Basic account (limited content and tools).
INTRODUCTION
Overview
Around 80% of fires in hyperbaric chambers occur due to the presence of prohibited items in the chamber.[1] Why should each hyperbaric unit have a prohibited items assessment process/checklist? The unit safety program is the foundation of every hyperbaric medicine service. The safety program provides guidance in order to ensure that the risks and hazards associated with hyperbaric oxygen therapy are identified and eliminated. In certain circumstances, the risk cannot be eliminated (e.g. a wound care product cannot be removed) and mitigating measures must be taken to reduce the risk potential. The NFPA 99 Chapter 14 provides an algorithmic process that should be followed to determine risk of patient care items that may be introduced into the hyperbaric environment.[2] The Hyperbaric Safety Program, as a standard process, should require that all patient care items introduced into the hyperbaric environment have a risk assessment completed prior to use in the chamber.
However, in practice it is often challenging to complete and document such a risk assessment. Challenges include lack of product information to conduct a meaningful risk assessment, and lack of enough detail upon completion of a risk assessment to determine whether an item may or may not be safely allowed into the hyperbaric chamber. This topic evaluates those challenges and provides a practical scoring approach to help safety officers and medical directors determine whether patient care items may be allowed into the chamber. For an updated decision tree, scoring system, and standardized evaluation process to identify and mitigate potential safety go/no go risks in the hyperbaric chamber environment, see topic "How to Assess HBOT Prohibited Items: The Go-No/Go Risk Assessment Tool and The Burman Scoring System". For an overview on safety in HBOT, refer to topic "Safety and Hyperbaric Oxygen Therapy".
Background
Definitions and relevance
-
“Prohibited item” is an item that is needed and may be allowed in the chamber with caution, after a careful risk assessment is completed and risk mitigation strategies are in place (see blog article "Prohibited Items Vs Restricted?")
- According to the NFPA 99:
-
- “The physician in charge, with the concurrence of the safety director, shall be permitted to use one of the following prohibited items in the chamber: (1) Suture material; (2) Alloplastic devices; (3) Bacterial Barriers; (4) Surgical dressings; (6) Synthetic Textiles”
- “Physician and safety director approval to use prohibited items shall be stated in writing for all prohibited materials employed”.
-
- The ”prohibited item approval form” indicates the type of prohibited item allowed in the chamber. The safety director and medical director should sign the form upon approval of the item. See sample approval form.
THE NFPA RISK ASSESSMENT ALGORITHM
The NFPA 99 Chapter 14 Hyperbaric Facilities risk assessment algorithm is a detailed process for effectively managing patient care product(s) in a hyperbaric environment (Figure 1).[2] The NFPA risk assessment algorithm is intended for use when evaluating wound care dressings, products, and patient care devices for use in the hyperbaric environment. This process can also be applied to textiles that may be used in the hyperbaric chamber.
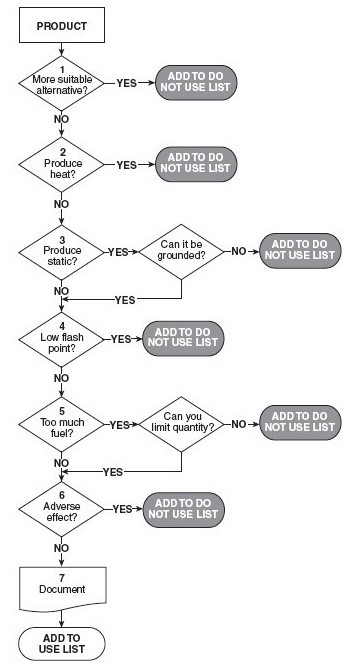
Fig. 1. NFPA Prohibited Item Risk Assessment Process[2]
The risk assessment algorithm is a methodical decision tree process that considers safety concerns such as spontaneous heat production, static electricity, flammable vapor, ignition temperature, as well as the total fuel load. However, the aforementioned risk assessment tool does not provide a means for documentation of the risk assessment, nor does it attempt to quantify the significance of the risk.
A.14.3.1.6.4.3 of the NFPA 99 2021 states:
"The hyperbaric facility should maintain a “use list” and a “do not use list” of items that have been evaluated for hyperbaric use."
In a regulatory sense, the word “should” does not mean “optional,” it means “required.” In addition to this list, it is important to keep documentation on file explaining the risk assessment for each item. Hence, the unit-specific foundation to require an “exceptions log.”
Each area within the assessment algorithm should be evaluated independently. The risk assessment evaluation begins by obtaining the Safety Data Sheet (SDS) for the product or prohibited item and answering several basic questions. Sometimes there is no specific SDS for wound care/dressing items or the flash point information is missing. However, one can find information on the individual components of the dressing or product (e.g., the SDS of petroleum gauze dressing - such as "Xeroform" - does not have information on flash point. The SDS of petroleum jelly - one of its main components - provides information on its flash point. For a repository of SDS, see topic "Safety Data Sheets".
Specifically, what is the prohibited item? Can the item or product be removed prior to treatment? If not, is there a suitable option? What mitigating measures can be taken to reduce risk? For example, does the item produce heat? If so, what mitigating measures can be taken?
Obstacles preventing compliance
Risk assessment in our current practice is challenging as the information required to complete the assessment may not be readily available. Guidance on the concerns below may be unclear to many:
- Understanding the purpose of the risk assessment algorithm
- Finding the answers to the questions that are posted in the algorithm (e.g. often answers are mere assumptions)
- How to proceed when information is not readily available
- What to do when wound care dressing and product safety data sheets (SDS) routinely do not report all the information needed.
- What to do if completion of the risk assessment process does not provide the necessary detail to identify the level of risk, the exposure potential, and the consequences if the item is allowed into the chamber
- What to do if completion of the risk assessment process does not guarantee adequate documentation
THE BURMAN RISK SCORING SYSTEM
Quantifying risk magnitude through a practical scoring approach
When considering a material, dressing, disinfectant, piece of equipment, or other product for use in a hyperbaric chamber, a consistent process should be followed to ensure that any identified, real risks can be effectively mitigated.
As a general rule, very few products are manufactured specifically for hyperbaric use, or evaluated and declared effective and/or safe by manufacturers. However, and in equal measure, many typical healthcare products do not present any significant risk of endangering patients, staff, or facilities.
Medical products require FDA approval prior to marketing and sale, but even with this approval, this does not apply in many cases to cover a hyperbaric medicine application. In order to meet the FDA requirements for off-label use, the medical director is required to make a decision both in the best interests of the patient and with due regard to safety.[3]
The exposure of patients, staff, and equipment to the hyperbaric oxygen environment will always present some degree of risk, which should be evaluated and mitigated as much as is practically and realistically achievable.
A risk assessment process based on identified hyperbaric facility hazards should allow each item to be properly evaluated and an appropriate risk level determined, enabling the risk management team and the medical director to make an appropriate decision.
Types of Risks
Three categories of risk need to be considered: fire, mechanical and physiological. Items under consideration should be evaluated in order to determine whether any or all of these categories apply, and if so, what the magnitude of the risk would be.
Definitions
- Risk: a risk can be defined as the probability of an exposure to a hazard leading to negative consequences. This in turn allows each of the above three identified areas of concern to be scored, using a Likert Scale (i.e. a type of rating scale) for probability, frequency of exposure and severity of potential consequences.
-
Fire hazards: fire has been proven to be the greatest of the risks encountered in hyperbaric chambers through the review of more than 95 years of experience in this field.[1] Items introduced into the hyperbaric environment should be considered in terms of a potential source of fuel, an ignition source, and the exposure to oxygen to promote and accelerate combustion.
-
Mechanical hazards include anything that leads to [4]:
-
- Impaired access: any restriction to movement inside the chamber due to placement of medical devices, cables or hoses may result in a trip injury or the equipment falling and injuring an occupant.
- Reduced visual access inside the chamber: reduction or restriction of visual access of chamber operators reduces their effectiveness as safety monitors (e.g. an external fixator that scratches the chamber acrylic window).
- Collapse or rupture during changes in pressures: for instance, sealed or semi-sealed containers that are not adequately vented left inside the chamber will either collapse under pressure (possibly resulting in adiabatic heating of the contents and thus imposing a fire or explosion risk); or explode on resurfacing if the gas trapped within the container cannot escape during the ascent.
- Malfunction, disruption or inoperativeness of many standard items when placed in service under hyperbaric conditions: includes the implosion of lamps and vacuum tubes (e.g., cathode ray tubes in medical monitors), failure of pressure-touch switches, overloading of fans due to a higher gas density, inaccurate operation of flowmeters, pressure gauges and regulators.
-
Physiological hazards [4]: Physiological hazards for the purpose of the Risk Assessment Tool include those arising as a result of mechanical, electrical or other safety malfunctions. For instance:
-
- General hazards: these include electric shock or overheated materials leading to acute burns, CO poisoning, intoxication by products generated during the combustion of materials (e.g., cyanide and chlorine).
- Uncomfortably high noise for the patient: may occur during operation of medical equipment, such as operation of a fan or a mechanical ventilator.
- Probability of an accident: the likelihood that an incident or accident such as combustion due to fire/explosion, mechanical failure of the chamber, or undesired physiological event will happen as a result of exposure to the hazard.
- Frequency of exposure: how often will the facility, occupant or staff member be exposed to the hazard, if no mitigation actions are taken.
- Severity of potential consequences: severity of the outcome, should an accident occur.
The Burman Risk Assessment Scoring Process
- 1. Use Tables 1-3 to assign scores to the items below.[4] When assigning scores, make a realistic assessment of the actual risk of each of three types of events (i.e. fire and explosion, mechanical hazards, physiological and medical hazards):
-
- a) The probability of an accident (i.e., the probability/ likelihood of occurrence) (Table 1),
- b) The frequency of exposure to the hazard (Table 2), and
- c) The severity of the outcome (i.e., potential negative consequences) should an accident occur (Table 3)
-
- When determining severity of the outcome, consider a realistic worst-case scenario
- 2. Multiply the score assigned to the probability of an accident, the frequency of exposure to the hazard, and the severity (Risk Score = probability x exposure x consequence):
-
- For instance: if the probability of fire and explosion is definite (score=5), the frequency of exposure to the hazard is continuous during an entire shift (score=5), and the severity of the outcome should fire and explosion happen is catastrophic (score=5), then the risk score for fire and explosion is 125 (5x 5 x 5 =125)
- 3. Compare the calculated risk score (e.g. 125) to the ranges in Table 4 and identify the risk level
- 4. Where exposure to the hazard leads to more than one type of risk, each risk should be assessed separately (that is, the risk score should be calculated as probability x exposure x severity) and the highest score used to determine if the item can or cannot go into the chamber.
-
- For example, if there is risk of fire and explosion and also risk of mechanical hazard, calculate the risk score for fire and explosion and also calculate the risk score for mechanical hazard using the formula Risk Score = probability x exposure x consequence.
Table 1. Burman Scoring System Risk Assessment Process: Scoring the Probability/Likelihood Of An Event
Probability/likelihood of an event (incident or accident) occurring
|
|
Fire and explosion
|
Mechanical hazards
|
Physiological & Medical Hazards
|
|
Combustion definite
|
5
|
Failure definite
|
5
|
Event definite
|
5
|
|
Combustion expected
|
4
|
Failure expected
|
4
|
Event expected
|
4
|
|
Combustion possible
|
3
|
Failure possible
|
3
|
Event possible
|
3
|
|
Combustion unusual
|
2
|
Failure unusual
|
2
|
Event unusual
|
2
|
|
Combustion unlikely
|
1
|
Failure unlikely
|
1
|
Event unlikely
|
1
|
Table 2.
Burman Scoring System Risk Assessment Process: Scoring the Frequency of Exposure To The Hazard
Exposure to the hazard
|
|
Continuous: an entire shift
|
5
|
|
Daily: < say twice a day
|
4
|
|
Weekly: < say twice a week
|
3
|
|
Monthly: < say twice a month
|
2
|
|
Annually: < say twice a year
|
1
|
Table 3. Burman Scoring System Risk Assessment Process: Scoring the Severity of the Outcome
Exposure to the hazard
|
|
Catastrophic: e.g. death; life-threatening injury; destruction
|
5
|
|
Severe: e.g. significant injury; facility no longer available
|
4
|
|
Serious: e.g. reduced ability to treat/treatment quality
|
3
|
|
Significant: e.g. minor damage/injury; additional staff needed
|
2
|
|
Noticeable: e.g. inconvenience; additional work required
|
1
|
Table 4. Magnitude Of The Risk: Consolidated Risk Score
Risk Score
|
Risk Level
|
Risk Description
|
| >= 100 |
5 |
Extremely dangerous: do not operate facility |
| 50 - 99 |
4 |
Very high: stop use of specific equipment, process or procedure |
| 20 - 49 |
3 |
High: Requires urgent attention; proceed with great care |
| 5 - 19 |
2 |
Medium: Attention needed but operation may continue |
| 0-4 |
1 |
Low: Acceptable risk but noted |
Examples
The following examples illustrate the process when applied to items typically seen in a hyperbaric environment.
Example 1: Blankets for use in an oxygen-filled monoplace chamber:
The potential hazards are fire and the accumulation of lint blocking the exhaust system. There are no likely physiological risks.
The initial risk assessment:
-
Risk: Fire and explosion
- Identifying risks:
- Fuel: the NFPA 99 allows a suitable blend of cotton and polyester. The most likely fuel would thus be lint.
- Oxygen: continuous exposure
- Ignition source: blanket materials with the exception of those prohibited by NFPA 99 (e.g. silk or wool) require a significant amount of heat to initiate combustion.
- Assigning scores:
- Probability of fire: 1 (combustion unlikely)
- Exposure: 4 (less than twice a day)
- Consequence: 5 (potentially catastrophic based on history)
- Quantifying the risk level:
- Risk score: 1 x 4 x 5 = 20
- According to Table 4, a score of 20 represents a medium fire risk (risk level 2), requiring the usual attention and discipline in operation
-
Risk: Mechanical:
- Identifying risks:
- Lint production is based on the actual material to be used.
- Assigning scores:
- Probability of sufficient accumulation: 1 (failure unlikely)
- Exposure: 4 (less than twice a day)
- Consequence: 1 (slowed ascent rate due to outlet filter blockage, increase ascent rate where the exhaust regulator has lint on the seat).
- Quantifying the risk level:
- Risk score: 1 x 4 x 1 = 4
- According to Table 4, a score of 4 represents a very low mechanical risk (risk level 1), easily mitigated through regular inspection and cleaning.
Impression
With due attention to keeping out any known ignition sources – such as any substance that could have an exothermic reaction and resulting in temperatures higher than 140°F, selecting as blanket material that has restricted lint production and potential for static electricity generation, and through regular cleaning of the chamber, this risk can be rendered as ‘Acceptable’ - certainly no greater than typical scrubs used in a facility – and especially where mandatory grounding of patients is required.
Example 2: Wound-care product that cannot be removed prior to hyperbaric oxygen treatment as per physician orders
The potential hazards are fire and a potential reaction to the product through exposure to an elevated partial pressure of oxygen.
THE INITIAL RISK ASSESSMENT:
1. Risk: Fire and explosion
- Identifying risks:
-
- Fuel – The product Safety Data Sheet (SDS) should be consulted to determine whether any volatile vapors can be released at expected chamber temperatures and elevated partial pressure of oxygen (this information is not always easy to obtain – the manufacturer may need to be consulted).
- Ignition source – in the event of any potentially flammable materials, the required heat to initiate a fire is relatively low.
- Assigning scores:
-
- Probability of fire: 3 (combustion possible if any flammable vapors can be released)
- Exposure: 4 (less than twice a day)
- Consequence: 5 (potentially catastrophic)
- Quantifying the risk level
-
- Risk level 3 x 4 x 5 = 60
- According to Table 4, a score of 60 represents high risk - risk mitigation needed prior to use. Ensuring no flammable vapors can be released, grounding the patient and ensuring prohibited items are excluded from the chamber, would reduce a probability of fire to a 1 (new risk score 1 x 4 x 5 = 20). In addition, a damp towel could be placed over the dressing area to further protect against any possible ignition.
2. Risk: Physiological
- Identifying risks:
-
- Amended uptake of medication/dressing product.
- Quantifying the risk level:
-
- No possibility for any human reaction would yield a risk score of 1 x 4 x 0 = 0
- Potential reaction of medication with oxygen (not as yet been reported): 3 x 4 x 3 = 36
- A score of 36 represents a medium risk – treatment not advised if this specific product is used.
Impression
Risk assessments and mitigation steps taken should be undertaken in writing and presented to the medical director for endorsement.
CONCLUSION
The NFPA 99 Chapter 14 provides an algorithmic process that should be followed to determine risk of patient care items that may be introduced into the hyperbaric environment.[2] Despite the NFPA's requirement that all facilities have their own “use list” and a “do not use list” of items that have been evaluated for hyperbaric use" [2], several challenges prevent compliance. Frequent obstacles include lack of understanding of the purpose of the NFPA risk assessment algorithm, and the scarcity of information even upon completion of the risk assessment process to determine whether an item may or may not be allowed into the chamber. The Burman risk scoring system provides a practical scoring approach to help safety officers and medical directors determine whether patient care items may be allowed into the chamber. For an updated decision tree, scoring system, and standardized evaluation process to identify and mitigate potential safety go/no go risks in the hyperbaric chamber environment, see topic "How to Assess HBOT Prohibited Items: The Go-No/Go Risk Assessment Tool and The Burman Scoring System".
CATEGORY A CONTINUING EDUCATION CREDIT
This topic has been reviewed and approved by the National Board of Diving and Hyperbaric Medical Technology (NBDHMT) for one (1) Category A Credit. 'Meeting the Category A requirement related directly to any combination of hyperbaric operations, related technical aspects and chamber safety.'
To claim the credit:
- 1. Read the topic
- 2. Answer the examination and course critique questions.
- Take the quiz via CliniPaths by Wound Reference
- To receive a certificate, a passing score of 70% is required
- 3. After passing, click the provided link to access your certificate. If prompted, sign into your WoundReference account.
For more information on Category A continuing education credits see blog post " Hyperbaric Certification and Continuing Education for Technicians & Nurses".
Official reprint from WoundReference® woundreference.com ©2026 Wound Reference, Inc. All Rights Reserved
NOTE: This is a controlled document. This document is not a substitute for proper training, experience, and exercising of professional judgment. While every effort has been made to ensure the accuracy of the contents, neither the authors nor the Wound Reference, Inc. give any guarantee as to the accuracy of the information contained in them nor accept any liability, with respect to loss, damage, injury or expense arising from any such errors or omissions in the contents of the work.