Last updated on 1/4/24 | First published on 8/1/22 | Literature review current through May. 2026
[cite]
Authors:
Elaine Horibe Song MD, PhD, MBA,
Topic editors:
Cathy Milne APRN, MSN, CWOCN-AP,
Scott A. Robinson MD,
more...
Coauthor(s)
Elaine Horibe Song, MD, PhD, MBACo-Founder and Editor, Wound Reference, Inc;
Professor (Affiliate), Division of Plastic Surgery, Federal University of Sao Paulo;
Volunteer, Association for the Advancement of Wound Care;
Google Scholar Profile
Disclosures: Nothing to disclose
Editors
Cathy Milne, APRN, MSN, CWOCN-AP
Disclosures: Nothing to disclose
Scott A. Robinson, MD
Disclosures: Nothing to disclose
For full access to this topic and more
premium content, upgrade today. Or get started with a Free Basic account (limited content and tools).
ABSTRACT
Atypical wounds comprise approximately 20% of all chronic wounds.[1][2] Delay in diagnosis can lead to higher mortality rates [3], not only due to comorbidities but also due to pain, impaired quality of life and lower socio-economic circumstances leading to higher rates of suicide.[1][4]
This topic provides an updated, summarized review on assessment and management of this often misdiagnosed condition. For a list on types of atypical ulcers, see Table 2 below and also refer to topic "Inflammatory Ulcers".
Atypical ulcers may be caused by different types of etiologies and can be neoplastic, inflammatory, vasculopathic, infectious, drug-induced, genetic or due to other chronic conditions.[1][5] See section 'Etiological groups and risk factors'.
Assessment: Patients with atypical ulcers often present with multiple other comorbidities that may impede healing. Thus, it is recommended that a qualified professional multidisciplinary team evaluate and perform a complete assessment of the patient. It is important to obtain a comprehensive history of the patient’s current condition, recurrence, past medical and surgical history, medications, lifestyle, diet, smoking cessation, exercise and other risk factors related to atypical ulcers. In addition to comorbidities causing the atypical wound, patients may also present with concurrent mixed etiologies, for instance arterial and/or venous insufficiency, neuropathy or local pressure. Suggestive findings upon physical examination include hypergranulation, pathergy, violaceous and undermined borders, necrosis, atypical location, severe pain (out of proportion which is normally associated with wounds), livedo racemosa and reticularis, rapidly progressive ulceration. Skin biopsy is an important element in confirming diagnosis. See sections History and Physical Exam, Diagnosis (biopsy and laboratory tests), Differential Diagnosis
Treatment: When treating atypical wounds, a holistic assessment and appropriate clinical examination of the patient is needed in order to ensure that an accurate diagnosis can be made before any treatment regime is initiated. Once a diagnosis has been established, an interdisciplinary approach should be adopted in order to reach a successful outcome. The exact nature of the treatment relies on the specific type of atypical wound (see
Table 3). Local wound care should follow established principles of wound bed preparation.
INTRODUCTION
Background
Definition
- Atypical wounds are considered as wounds that do not fall into a typical non-healing wound category, i.e. venous, arterial, mixed, pressure ulcer (PU) or diabetic foot ulcers (DFU).[1][6]
- Atypical ulcers may be caused by different types of etiologies and can be neoplastic, inflammatory, vasculopathic, infectious, genetic or other chronic conditions.[1][5]
- An atypical wound can be suspected if the wound has an abnormal presentation or location, pain out of proportion of the size of the wound and does not heal within four to 12 weeks with a good treatment plan.[1][7]
Relevance
- Atypical ulcers are usually one of the manifestations of underlying systemic comorbidities. With an aging population and increase in comorbidities these numbers are expected to rise.[1] Unfamiliarity with comorbidities and potential etiologies contribute towards delay in diagnosis, which can lead to higher mortality rates [3], not only due to comorbidities but also due to pain, impaired quality of life and lower socio-economic circumstances leading to higher rates of suicide.[1][4]
Etiological groups and risk factors
Table 1 below lists the main etiological groups, along with examples of conditions representing each group and respective risk factors.
Table 1. Atypical ulcers: etiological groups, examples of conditions representing each group and respective risk factors
| Etiology | Definitions and examples | Risk Factors |
Neoplastic ulcers
| - Malignant wounds can be classified into primary or secondary:
- Primary malignant wound: ulcers due to tumors of cutaneous origin such as basal cell carcinomas (BCC), squamous cell carcinomas (SCC), and malignant melanoma. BCC and SCC are more common than melanoma.
- Secondary ulcerating skin tumors: malignant tumors that develop from chronic ulcers, burn scars or osteomyelitis sinuses or due to distant metastasis.
| - For primary malignant wounds: risk factors include fair skin and red hair (Fitzpatrick skin type 1), family history, older age, lifetime cumulative UV-light exposure, longstanding immunosuppression [8]
|
| Inflammatory ulcers | - Ulcerations in which inflammation is the primary pathologic process resulting in a lesion; that is, cases in which inflammation is the cause rather than the consequence of the ulcer.[9] Examples include:
- Pyoderma gangrenosum: see topic "Pyoderma Gangrenosum - Introduction and Assessment".
- Small/ medium vessel vasculitis: inflammation during which the primary event is the destruction of the walls of blood vessels.[1] Includes vasculitis associated with systemic auto-immune diseases such as scleroderma, rheumatoid arthritis, systemic lupus erythematosus.[2] See topic "Cutaneous Vasculitis"
- Hidradenitis suppurativa
- Ulcerative necrobiosis lipoidica
- Ulcerative lichen planus
- Ulcerative sarcoidosis
| - For pyoderma gangrenosum: the three most common disorders associated with pyoderma gangrenosum include inflammatory bowel disease, arthritis, and myeloproliferative disorders.[10]
- For small/ medium vessel vasculitis: risk factors include rheumatoid arthritis, connective tissue disease and others
- For hidradenitis suppurativa: risk factors include hypertension, obesity, type 2 diabetes.[1]
|
| Vasculopathic ulcers | - Ulcers caused by an occlusive vasculopathy, which is initially a non-inflammatory blood vessels disorder mainly characterized by a decrease or complete occlusion of the vessel lumina.[1] They may be caused by a systemic or local vascular coagulopathy, by embolisation, by platelet plugging or by cold-related gelling of molecules, such as cryoglobulins.[1] A clinical hallmark of occlusive non-vasculitic vasculopathic disorders is retiform purpura.[1]
- Examples of conditions that cause vasculopathic ulcers and respective risk factors include:
- Calciphylaxis.[1]
- Cryofibrinogenemia
- Antiphospholipid antibody syndrome
| - For calciphylaxis with end-stage renal disease (ESRD): diabetes, obesity, female sex, dependence on dialysis, calcium and phosphate disturbances, overuse of vitamin D and calcium substitutes and/or calcium-based phosphate binders and vitamin K depletion (from malnutrition or malabsorption) or medication with vitamin K antagonists.[1][11][12]
- For calciphylaxis without ESRD: morbid obesity associated with type 2 diabetes and essential hypertension
- For cryofibrinogenemia: malignancy (particularly prostatic cancer, other solid organ tumors, and lymphomas), infection, collagen vascular disease, thromboembolic disease
- For antiphospholipid antibody syndrome: risk factors include systemic lupus erythematosus (SLE) and the presence of positive antiphospholipid antibodies (aPL).[13]
|
| Infectious ulcers | - Infectious ulcers occur most commonly after primary inoculation.
- Bacteria (group A beta-haemolytic streptococci, which causes ecthyma), mycobacteria (Mycobacterium tuberculosis), yeast (Candida albicans), mycetes (Sporotricosis), protozoa (Leishmania), skin parasites (Conus, Tunga) and arthropod bites (Entomodermatosis) are some of the different etiological pathogens of infectious ulcers.
- Ecthyma is an ulcerative infectious disease and may be caused by different bacteria, mainly group A beta-haemolytic streptococci, such as Streptococcus pyogenes.
| - Infectious ulcers often have an endemic distribution or may be associated with outdoor activities.[5]
|
| Drug-induced ulcers | - The cutaneous side effects of hydroxyurea treatment include hyperpigmentation, alopecia, melanonychia and painful ulcers with a well-defined and adherent fibrinous wound bed and often appear at the same sites of a previous trauma.[14]
- Heroin induced ulcers show an irregular wound bed with necrotic fibrinous areas and frequent colonization. The perilesional skin presents numerous post-injection scars.[5][15]
| - Hydroxyurea ulcers affect patients with hematological disorders.[14]
- Heroin extravasation and secondary ulcers are typical in patients with heroin addiction.[5][15]
|
| Factitious ulcers | - Factitious ulcers are a form of dermatitis artefacta, which can be defined as the deliberate and conscious production of self-inflicted lesions, such as ulcers, to satisfy an unconscious psychological or emotional need.[16]
| - Female gender, underlying psychiatric diagnoses, periods of increased stress [16]
|
Pathophysiology
- Pathophysiology varies according to the type of atypical ulcer.
- Regardless of its etiology however, like any non-healing wound, atypical ulcers present varying degrees of inflammation.[1]
Epidemiology
- Atypical wounds comprise approximately 20% of all chronic wounds.[1][2]
ASSESSMENT
Patients with atypical ulcers often present with multiple other comorbidities that may impede healing. Thus, it is recommended that a qualified professional multidisciplinary team evaluate and perform a complete assessment of the patient. It is important to obtain a comprehensive history of the patient’s current condition, recurrence, past medical and surgical history, medications, lifestyle, diet, smoking cessation, exercise and other risk factors related to atypical ulcers. Table 1 above illustrates risk factors that can be potentially associated with specific etiologies. See topic "How to Assess a Patient with Chronic Wounds".
History
- A comprehensive and detailed history is essential for the correct diagnosis of atypical ulcers. In order to diagnose an atypical ulcer, exclusion of typical etiologies is the first step.[5] See topics "Arterial Ulcer - Introduction and Assessment", "Venous ulcers - Introduction and Assessment", "Diabetic Foot Ulcer - Introduction and Assessment", "Pressure Ulcers/Injuries - Introduction and Assessment".
Patient's and caregivers' concerns
- From the patient perspective, atypical wounds can be very painful and have prolonged healing times which lead to impaired quality of life. [17]
- Many patients suffer physically from odor, exudate, pain, reduced mobility and psychologically with negative emotions, loneliness and depression. [18]
Physical Examination
- In addition to comorbidities causing the atypical wound, patients may also present with concurrent mixed etiologies, for instance arterial and/or venous insufficiency, neuropathy or local pressure. Therefore, careful arterial evaluation (e.g. pulse palpation, measurement of the ankle-brachial pressure index (ABI)) is needed for every patient. The patient should also be assessed for signs of venous insufficiency (e.g. edema, varicose veins, etc) . Exclusion of neuropathy using microfilament sensory testing is also crucial. See topics "Arterial Ulcer - Introduction and Assessment", "Venous ulcers - Introduction and Assessment", "Diabetic Foot Ulcer - Introduction and Assessment", "Pressure Ulcers/Injuries - Introduction and Assessment".
Wound Characteristics
- Wound size, depth, tissue, exudate, odor, and periwound skin should be assessed. If infection is suspected a wound specimen is needed for culture and sensitivity to ensure appropriate antibiotics are used. See topics "How to Assess a Patient with Chronic Wounds" and "How to Collect a Wound Swab (Levine Technique) for Culture".
- Characteristics of atypical wounds vary according to the etiology, but signs that may indicate an atypical cause include [1]:
- Hypergranulation
- Pathergy
- Violaceous and undermined borders
- Necrosis
- Atypical location
- Severe pain (out of proportion which is normally associated with wounds)
- Livedo racemosa and reticularis
- Rapidly progressive ulceration
Diagnosis
- Diagnosis of an atypical ulcer is based on a detailed and comprehensive history and physical examination, along with additional tests including skin biopsy, laboratory blood and imaging tests.
- In order to diagnose an atypical ulcer, exclusion of typical etiologies (i.e. venous insufficiency, arterial insufficiency, neuropathy and pressure) is the first step.[5] If the wound does not fit within these typical presentations, an atypical cause of wound should be suspected.[1] Prompt and exact diagnosis is essential also as many of the atypical wounds need specific treatment differing from the traditional ones and an incorrect diagnosis can lead to devastating outcomes and increased mortality.[1][19]
- A skin biopsy is often used to confirm unclear diagnosis. [1][5]
- In addition, for all wounds that have been initially diagnosed as a "typical ulcer" but have not shown signs of healing within 4 to 12 weeks of adequate treatment, at least one biopsy should be obtained. [1]
- In order to obtain a reliable specimen, the biopsy should be taken from the wound edge with a scalpel.
- In case of suspected infectious ulcers, a wide biopsy for tissue cultures for atypical bacterial, mycobacterial and fungal infections should be taken.
- For specific guidance on biopsy for suspected cancerous, autoimmune, vasculitic, inflammatory or infectious lesions see section 'Selection of the Biopsy Site, Technique and Transport Medium' and algorithm 'How to select skin biopsy technique, site and transport media' in topic "How to Perform a Wound Biopsy".
- For all patients with a lower extremity ulcer, blood flow to the ulcer should be evaluated with a non-invasive arterial test (see section 'Non-invasive arterial tests' in topic "Arterial Ulcers - Introduction and Assessment").
- For patients with signs of venous insufficiency, a duplex ultrasound of lower extremities should be performed (see section 'Diagnosis' in topic "Venous Ulcer - Introduction and Assessment")
- Further laboratory testing is often needed to clarify the diagnosis, including complete blood cell count, metabolic profile, urinalysis and possible stool guaiac test.
- For suspected vasculitis or vasculopathy: testing should include also anti-nuclear antibodies (ANA), anti-neutrophilic cytoplasmic antibodies (ANCA), rheumatoid factor, anticyclic citrullinated peptide, serum protein electrophoresis, hepatitis profile, cryoglobulins, lupus anticoagulant, antiphospholipid and anticardiolipin antibodies, protein C, protein S, factor V Leiden and antithrombin III levels, cryofibrinogens and coagulation.[1][6][20]
Differential Diagnosis
- Table 2 below lists some differential diagnosis among atypical ulcers.
Table 2. Differential diagnosis among atypical ulcers
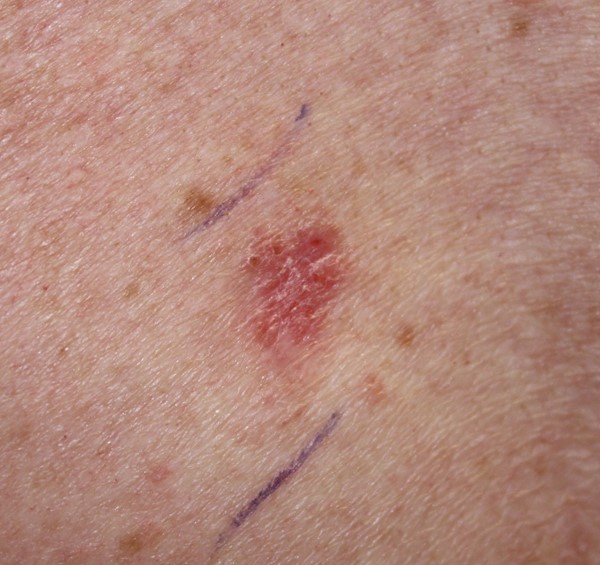
| Neoplastic | Picture |
Primary skin cancer: basal cell carcinoma
| 
|
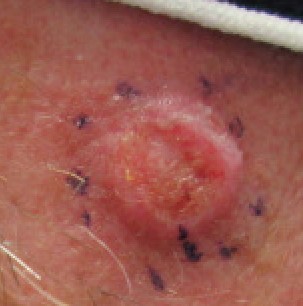
Primary skin cancer: squamous cell carcinomas
| 
|
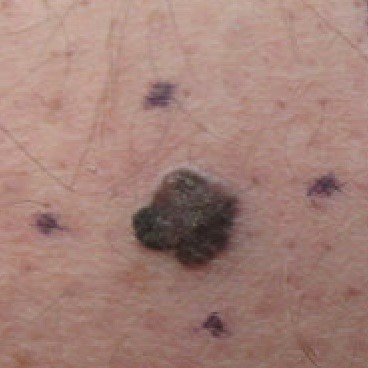
Primary skin cancer: malignant melanomas
| 
|
Malignant fungating wounds
|
|
Soft tissue sarcomas in skin
|
|
Kaposi's sarcoma
|
|
| Inflammatory |
|
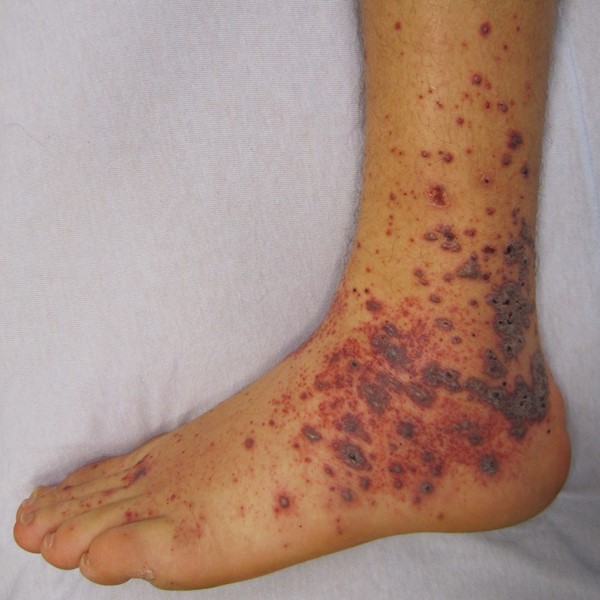
Drug-induced vasculitis
| 
|
Hidradenitis suppurativa
|
|
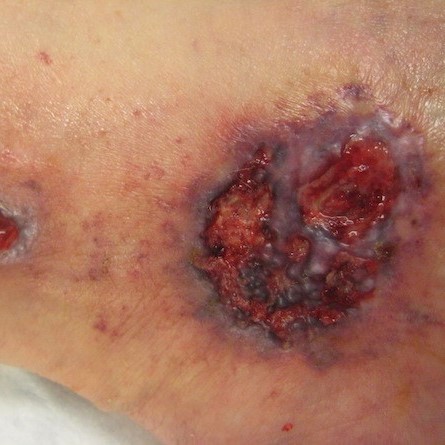
Pyoderma gangrenosum
| 
|
| Cutaneous vasculitis |
|
Ulcerative lichen planus
|
|
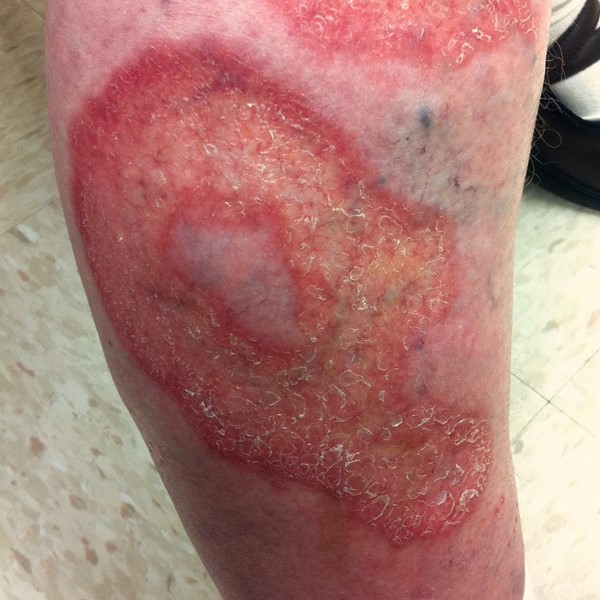
Ulcerative necrobiosis lipoidica
| 
|
Ulcerative sarcoidosis
|
|
Cutaneous Manifestations Associated With COVID-19
|
|
Vasculopathic
|
|
Antiphospholipid antibody syndrome
|
|
Calciphylaxis
|
|
Cryofibrinogenemia
|
|
Sickle cell disease ulcers
|
|
Drug-induced
|
|
Coumadin-induced skin necrosis
|
|
Hydroxyurea-induced ulcer
|
|
Infectious Ulcer
|
|
Buruli ulcer
|
|
Cutaneous leishmaniasis
|
|
| Factitious Ulcer |
|
Dermatitis artefacta
|
|
Documentation
- Documentation should include all observations and assessments, findings, test results, management plan, rationale, and schedule for reassessment in order to aid monitoring and facilitate communication among clinicians.[5]
TREATMENT
Treatment goals
- When treating atypical wounds, a holistic assessment and appropriate clinical examination of the patient is needed in order to ensure that an accurate diagnosis can be made before any treatment regime is initiated. Once a diagnosis has been established, an interdisciplinary approach should be adopted in order to reach a successful outcome. This might include one or more of the following health professionals: dermatologist, vascular and plastic surgeon, nephrologist, rheumatologist, tissue viability nurse, podiatrist. [1]
- The exact nature of the treatment relies on the specific type of atypical wound.
- Weekly assessment and measurements of the wound need to be recorded and if no improvement is seen after four weeks, plan reassessment and/or a specialist referral is required.[1][21]
Treat the cause and co-factors impeding healing
Atypical ulcers require a complex therapeutic approach. Local and systemic therapies specific to each etiology is summarized in Table 3.[5]
Table 3. Systemic therapies and local care for atypical wounds [5]
| Etiologies | Systemic therapies | Local care may include: |
| Neoplastic ulcers | Adjuvant and neoadjuvant therapies | Surgery, radiotherapy, electrochemotherapy, non-adherent dressings |
| Inflammatory ulcers | Corticosteroids, immunosuppressants, biological therapies
| Non-adherent dressings, autolytic and enzymatic debridement, absorbent dressings, anesthetic cream, compression bandaging
|
| Vasculopathic ulcers | Anticoagulants, antiplatelets, pentoxifylline | Non-adherent dressings, autolytic and enzymatic debridement, absorbent dressings, compression bandaging |
| Infectious ulcers | Antibiotics, antimycotic, antiparasitic drugs
| Antimicrobial dressings, secondary dressings
|
| Drug-induced ulcers | Heroin-induced ulcers Hydroxyurea-induced ulcers - Drug suspension or replacement, prostaglandin E1, pentoxifylline
| Heroin-induced ulcers - Silver dressings, PHMB dressings, iodine, binding bacteria dressings, compression bandaging
Hydroxyurea-induced ulcers
- Dressings, compression bandaging
|
Use of local corticosteroids
- The use of local corticosteroids in pyoderma gangrenosum is widely accepted. However, topical corticosteroids may also help to reduce excessive inflammation that impairs wound healing in other atypical wounds. [1][22]
- Topical steroids can be used as a single steroid topical agent or in combination with antimicrobial and antifungal agents in wounds displaying excessive and abnormal inflammatory features despite good clinical wound care practice.[1][22]
Local wound care
Local wound care should follow the wound bed preparation principles.[1][23]
Pain control
- Pain is a common feature of atypical wounds. Therefore, a validated assessment tool should be used to establish an individual’s level of pain and ensure analgesia is given before dressing change as many patients with atypical wounds suffer unnecessarily before, during and after dressing changes if pain is not managed well.
- Techniques to minimize pain or trauma to periwound skin include:
- Avoid adhesive dressings such as gauze. Non-adherent dressings have been found to be practical in clinical use, as pyoderma gangrenosum and vasculitis wounds may present with pathergy (i.e. worsen with minor trauma)
- Consider the wear time, moisture balance levels to ensure comfort for patients.
Cleansing
- Wound cleansing helps optimize the healing environment and decreases the potential for infection.[5]
Debridement
- Good tissue perfusion is essential for wound healing. However, the presence of necrosis or devitalized tissue hinders adequate perfusion and therefore needs to be removed by an appropriate method.
- Some atypical wounds may need regular debridement.
- Vasculitic ulcers and pyoderma gangrenosum should not be debrided during their active inflammatory phase, as debridement could be deleterious to the ulcer. However, gentle debridement may be carried out after the immunosuppressive treatment has reduced the inflammation.[1]
Infection and bioburden control
- In atypical wounds, the presence of bacteria delays healing therefore reducing the wound bioburden should reduce adverse effects.
- In cases of infection, antibiotics may be indicated, but holistic assessment of the patient is needed first to ensure correct diagnosis is made. Many patients with pyoderma gangrenosum have completed multiple courses of antibiotics before being diagnosed.
Periwound skin care
- An appropriate emollient after cleansing will help prevent cracks in skin and dry scales building up.[1]
Moisture balance
- Exudate facilitates the inflammatory response and promotes autolytic debridement and epithelization. Wound exudate should be assessed at dressing change noting color, amount, high viscosity, odor and consistency. However, too much moisture can lead to maceration and damage surrounding skin.[1]
- Dressing selection should be based on the condition of the wound bed and the nature/tissue types present. For customized, wound-specific recommendations, use Wound Prep and Dress Tool.
Plan Reassessment
- Weekly assessment and measurements of the wound need to be recorded and if no improvement is seen after four weeks a specialist referral is required.[1][21]
Official reprint from WoundReference® woundreference.com ©2026 Wound Reference, Inc. All Rights Reserved
Use of WoundReference is subject to the
Subscription and License Agreement.
NOTE: This is a controlled document. This document is not a substitute for proper training, experience, and exercising of professional judgment. While every effort has been made to ensure the accuracy of the contents, neither the authors nor the Wound Reference, Inc. give any guarantee as to the accuracy of the information contained in them nor accept any liability, with respect to loss, damage, injury or expense arising from any such errors or omissions in the contents of the work.