ABSTRACT
A surgical wound complication is defined as a disruption to normal incisional wound healing following surgery.[1] In 2014, total Medicare spending estimates for all wound types ranged from $28.1 to $96.8 billion.[2] Including infection costs, the most expensive estimates were for surgical wounds (up to $38.3 billion).[2] Despite advances in wound care, surgical wound complications continue to be the most commonly managed wound type in some clinical care settings, more so than pressure injuries and other wound types.[1] Surgical wound complications include but are not limited to surgical site infections (SSI), surgical wound dehiscence, hematoma, seroma, scarring, medical adhesive-related skin injury (MARSI). etc.
This topic provides an updated, summarized review on assessment and management of surgical wound complications commonly seen by wound care clinicians.
Development of surgical wound complications is multifactorial, stemming from physiological, surgical and environmental factors. See 'Risk Factors'
Assessment: Patients with surgical wound complications often present with multiple other comorbidities that may impede healing. Thus, it is recommended that a qualified professional multidisciplinary team evaluate and perform a complete assessment of the patient. In regards to assessment of the surgical wound, timing is key. Surgical wound complications may arise within the first hours after surgery (e.g. hematoma). SSI is most commonly reported between days 7 and 9.[1] Suggestive findings upon physical examination include local signs such as erythema, pain, swelling, pus, wound breakdown and separation, and systemic findings such as fever, malaise, elevated C-reactive protein (CRP). Diagnosis is mainly clinical, but depending on the suspected surgical wound complication, exams are used to confirm diagnosis. See sections History and Physical Exam, Diagnosis, Differential Diagnosis.
Treatment: For management of surgical wound complications, a holistic assessment and appropriate clinical examination of the patient is needed prior to implementing interventions. The exact nature of the treatment relies on the specific type of surgical wound complication (see
Table 6). Local wound care should follow established principles of wound bed preparation.
INTRODUCTION
Overview
Surgical wounds are present in all surgical procedures. In addition to being obvious, the fact that they result from a surgical procedure differentiates them from wounds of exogenous etiology (trauma, insect bites, burns, for example) or endogenous etiology (e.g. arterial or venous leg ulcers). Surgical wound complications are a highly relevant problem. In 2014, total Medicare spending estimates for all wound types ranged from $28.1 to $96.8 billion.[2] Including infection costs, the most expensive estimates were for surgical wounds (up to $38.3 billion).[2]
This topic provides an updated, summarized review on assessment and management of surgical wound complications commonly seen by wound care clinicians.
Background
Definitions
- Surgical wound: is a wound resulting from an intervention that interrupts the intact integumentary system (skin, hair, nails and sweat glands).[3]
- According to the "Outcome and Assessment Information Set-E" (OASIS-E, a modification to the OASIS that home health agencies in the United States must collect in order to participate in the Medicare program)[4]:
- Surgery on a pre-existing wound or due to a traumatic injury involving the skin is not classified as a surgical wound; however, surgery to repair or remove an injured internal organ due to trauma, where the skin was intact is considered a surgical wound.
- A surgical site closed primarily (with sutures, staples, or a chemical bonding agent) is generally described in documentation as a surgical wound until re-epithelialization has been present for approximately 30 days, unless it dehisces or presents signs of infection. After 30 days, it is generally described as a scar and should not be included in this item.
- Surgical wound complication: A surgical wound complication is defined as a disruption to normal incisional wound healing following surgery.[1] Examples of surgical wounds complications include:
- Surgical site infection (SSI): SSI is an infection related to a surgical procedure that occurs around the surgical site within 30 days of surgery (or up to 90 days if an implant was involved). SSIs are described as incisional SSIs (superficial, deep) and organ/space SSIs.[5]
- Surgical wound dehiscence (SWD): SWD is the separation of the margins of a closed surgical incision that has been made in skin, with or without exposure or protrusion of underlying tissue, organs or implants. Separation may occur at single or multiple regions, or involve the full length of the incision, and may affect some or all tissue layers. A dehisced incision may, or may not, display clinical signs and symptoms of infection.[6]
- This severe complication may lead to immediate surgical intervention, increased risk for repeat dehiscence, SSI and/or development of incisional hernia formation [3]
- Hematoma (i.e accumulation of clotted blood in the operated area) or seroma (i.e. accumulation of serous fluid in the operated area): these fluid collections may increase pressure, compress blood vessels, causing wound ischemia and, if untreated, may cause tissue necrosis.
- Fistula (see topic "Case: Managing High-output Enteroatmospheric Fistulas - A Case Study and Review)
- Delayed wound healing
Relevance
- Surgical wound complications are a highly relevant problem. In the U.S., in 2014, nearly 15% of Medicare beneficiaries (8.2 million) had at least one type of wound or infection.[2]
- SSI were the largest prevalence category (4.0%), followed by diabetic infections (3.4%).[2]
- Total Medicare spending estimates for all wound types ranged from $28.1 to $96.8 billion.[2] Including infection costs, the most expensive estimates were for surgical wounds (up to $38.3 billion).[2]
- SSI extends hospital length of stay by 9.7 days, with cost of hospitalization increased by more than $20,000 per admission.[5]
- SSI is the leading cause for hospital readmission.[1]
- Seventy-seven percent of surgical patient deaths are reported to be related to infection.[7] Infection has a direct correlation to open surgical wounds.
The wound healing process
- To better understand how surgical wound complications occur, it is important to review the normal wound healing process. Wound healing of is one of the most important and complex physiological functions of the skin. Didactically it is divided into phases: hemostasis, inflammation, proliferation and maturation (Figure 1). However, in practice, they are dynamic, overlapping each other, presenting periods in which a relevant event of the phase stands out and names the phase itself.
- In addition, skin repair requires a complex synchronization of different cell types, resident or autochthonous of the skin, as well as migratory cells. When the skin suffers an injury sufficiently capable of triggering the wound healing, several types of these cells initiate the coordination of the repair in the previously mentioned stages to restore the skin integrity.
- For details, see topic "The Skin" and section 'The Wound Healing Process' in topic "Principles of Wound Healing".
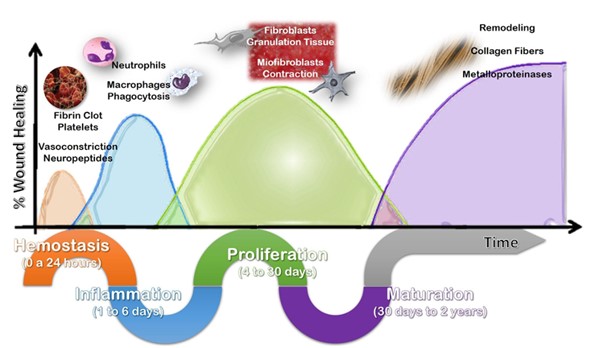
Figure. 1 : Schematic illustrating the wound healing process, highlighting the main phases (hemostasis, inflammation, proliferation and maturation), events and cellular components. However, it should be noted that the phases overlap and are dynamic
Types of wound closure
It is also important to review how surgical wounds are typically closed. Surgical wound closure can be classified as primary, secondary and delayed primary closure [8] :
Primary wound closure (or primary intention)
- Wound closure may be achieved by approximating the wound margins with sutures using surgical stitches. This method, called wound closure by first (primary) intention, reduces the size of the wound and facilitates the process of wound healing.
Delayed primary wound closure (or tertiary intention)
- Some more extensive wounds that cannot be closed primarily may be initially managed with local dressings and debridements, until the wound can be closed with interrupted sutures. This delayed method is referred to as healing by delayed primary wound closure or tertiary intention.
Secondary wound closure (or secondary intention)
- When the decision is taken to achieve wound closure by physiological mechanisms of wound healing, through local debridement and application of dressings, wound closure is known as closure by second (or secondary) intention. The majority of chronic wounds that heal do so by secondary or tertiary intention.
Classification of surgical wounds
The Centers for Disease Control and Prevention (CDC) classifies surgical wounds (not procedures) as clean, clean-contaminated, contaminated or dirty or infected wounds. Surgery on anatomical areas that are considered ‘clean’ (e.g., in prosthetic surgery) have relatively low SSI rates, whereas operations on dirty anatomical areas (e.g., the bowel) can exceed 10%. [9]
Table 1 includes the CDC classification, as well as descriptions and examples of surgical wounds commonly seen in wound clinics and community settings [3][5]:
Table 1. Surgical wound classification by the CDC and examples
| Classification | Description | Risk of infection (%) | Examples |
| Clean | An uninfected operative wound in which no inflammation is encountered and the respiratory, alimentary, genital or uninfected urinary tracts are not entered. In addition, clean wounds are primarily closed and, if necessary, drained with closed drainage. Operative incisional wounds that follow non-penetrating (blunt) trauma should be included in this category, if they meet the criteria.
| < 2 | - Lower leg vascular bypass/saphenous vein harvesting
- Skin graft and donor sites
- Recipient sites
- Hip and knee arthroplasty
|
| Clean-contaminated | Operative wounds in which the respiratory, alimentary, genital or urinary tracts are entered under controlled conditions and without unusual contamination. Specifically, operations involving the biliary tract, appendix, vagina and oropharynx are included in this category, provided no evidence of infection or major break in technique is encountered.
| < 10 | - Caesarian (C) section incision (fetal fluids are contaminated with the endogenous vaginal flora)
|
Contaminated
| Operations on new, open traumatic wounds; operations where there is a major break in aseptic technique; and operations in which there is gross spillage from the gastrointestinal tract or acute inflammation without pus being encountered.
| ~ 20 | - Open abdomen with or without new ileostomy or colostomy due to bowel perforation/ enterocutaneous fistula
|
Dirty or infected wounds
| Operations in which acute inflammation with pus is encountered, or in which perforated viscera are found; this includes operations on traumatic wounds that have retained dead and severely damaged tissue, foreign bodies or fecal contamination. Operations include those where the micro-organisms causing post-operative infection are likely to have been present before surgery.
| ~ 40 | - Hernia repair with infected mesh
- Incision and drainage (I&D) of abscesses
- Pilonidal sinus surgery
|
Risk factors
In general, risk factors for surgical wound complications can be grouped into physiological factors, surgical factors and environmental factors (Figure 2).
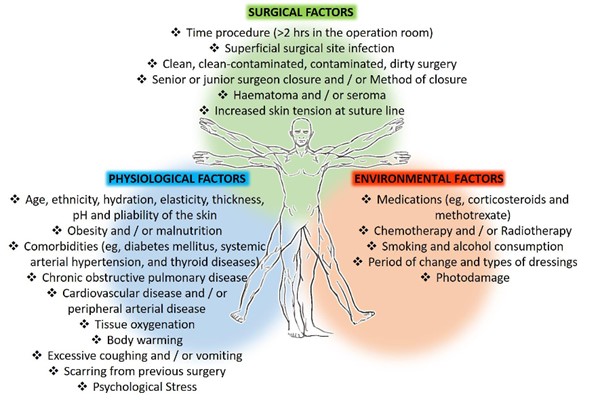
Figure 2. Risk factors for surgical complications
Physiological risk factors
Physiological risk factors for surgical wound complications include [3]:
- Advanced age: with aging, functions of cutaneous appendages and the overall of global inflammatory activity decreases. This contrasts with the characteristics of young skin, with greater cellular activity and a high prevalence of hypertrophic scars in the incisions.
- Ethnicity: ethnic characteristics of the skin (e.g. amount of pigmentation, differences in hydration, elasticity, thickness, pH and pliability of the skin) are associated with different wound healing responses.
- Obesity: higher body mass indexes (BMI, males with a BMI > 25 kg/m2 and women with a BMI > 30 kg/m2) are associated with a five times greater risk of an SSI.[10]
- Malnutrition
- Hypertension and coronary artery disease
- Pre-existing body site infection
- Diabetes mellitus (poor glycemic control)
- Size and virulence of the microbial load in the surgical area
- General health and co-morbid disease processes, including medications that affect integrity of the individual’s host defenses
- Previous complications with anesthetic and surgeries[5]
Environmental risk factors
Environmental risk factors for surgical wound complications include [3]:
- Medications
- Chemotherapy/radiotherapy
- Smoking
- Alcohol or substance use
- Physical activity and mobility limitations
Surgical risk factors
Surgical risk factors are summarized in Table 2.
Table 2. Specific intra-operative and post-operative factors that increase risk for surgical site complications, especially surgical site infection [3]
| Intra-operative risks | Post-operative risks |
- Longer procedures [11]
- Higher levels of surgical contamination (see Table 1)
- Open surgeries (vs. laparoscopic)
- Presence of conditions that restrict peripheral perfusion and decrease tissue oxygenation (e.g. hypovolemia, excessive pain, vasoconstricting drugs and hypothermia)
- Emergent surgery (vs. elective)
- Implants (vs. no implants)
- Use of internal mammary artery grafts (for coronary artery bypass graft)
- Prolonged ventilation
- Use of blood products
| - Saturated and/or leaking wound dressings, which allow migration of bacteria to the wound in a rapid manner.
- Disruption of the sutures or staples by vigorous cleansing before it has re-epithelialized can introduce bacteria below the dermis.
|
Risks for wound dehiscence
- Surgical wound dehiscence is often noticed between 4 to 14 days post-operatively.[6]
- Risk factors include continuing wound exudate past 48 hours post-op, an SSI, poor glycemic control, malnutrition, obesity, mechanical stress on the wound bed from heavy lifting, coughing, vomiting, sneezing and straining.[6]
Pathophysiology
- Usually, no single primary factor results in a surgical wound complication. Rather, multiple issues combine to disrupt the wound healing process (Figure 3).

Figure 3. Wound dehiscence status post abdominoplasty. Patient is a tobacco smoker and wound was sutured under tension. Poor tissue perfusion combined with mechanical tension led to local tissue necrosis.
ASSESSMENT
History
The non-exhaustive checklist below highlights relevant factors that should be assessed when taking history of a patient with a surgical wound complication. Risk factors for development of surgical wound complications are indicated with the icon
Chief Complaint and History of Present Illness
- Age: with aging, functions of cutaneous appendages and the overall of global inflammatory activity decreases. See 'Risk Factors'
- Anatomical location and type of surgical wound complication: see types of 'surgical wound complications'.
- Which anatomical structures are involved? (e.g. wound dehiscence of a vascular bypass surgery)
- Duration/onset
- History of physiologic, surgical and environmental risk factors that increase risk for surgical complications See 'Risk Factors'.
- Reason for surgery and date
- Type of surgery? Emergency or elective?
- Are pre-op and operative notes available?
- Intra-operative and post-operative complications (e.g. hemorrhage, hypothermia, duration of surgery, SSI, closure method)
- Was an artificial hardware implanted?
- Size and virulence of the microbial load in the surgical area?
- Previous complications with anesthetic and surgeries?
- Pain: may indicate SSI, hematoma, seroma, dehiscence. Increased localized pain is a significant predictor of deep compartment infection.
- History of other conditions that may lead to chronic non-healing ulcers: for instance, chronic venous insufficiency, peripheral neuropathy, pressure, inflammation, infection
- Past treatment history, including past surgical history: for instance, previous problems with wound healing (e.g. surgical wound dehiscence, SSI), radiotherapy, chemotherapy, attempts to treat the complication with antibiotic therapy, debridement, etc
Medications and Allergies
Assess medication history (prescribed and self-prescribed):
- Medications that delay wound healing: anticoagulants, antimicrobials (various antibiotic classes), anti-angiogenesis agents (eg, bevacizumab, aflibercept), antineoplastic drugs, anti-rheumatoid drugs (eg, methotrexate, aspirin/nonsteroidal anti-inflammatory drugs [NSAIDs]), colchicine (anti-gout drug), topical hydrogen peroxide, topical iodine, full-strength 0.5% Dakin’s solution (sodium hypochlorite), nicotine, steroids, and vasoconstrictors.[12][13]
Assess history of allergies and sensitivities.
Family History
- Assess history of comorbidities that may be inherited (e.g. diabetes, obesity, hypertension) among parents or siblings. See 'Risk Factors'.
Social History
- Occupational: work settings that increase susceptibility for trauma may increase risk for wound dehiscence, hematoma, seroma, SSI .
- Tobacco: smoking (including second-hand passive smoke) increases the risk for delayed wound healing .
- Alcohol: increases the risk for delayed wound healing .
- Substance abuse: many illicit drugs can either cause wounds (not related to surgical wound complications per se) or instigate scratching that can produce wounds.[14]
Review of Systems
- General:
- Age
- Obesity or malnutrition .
- Pre-existing body site infection .
- Cancer . [15]
- Cardiovascular:
- Hypertension and coronary artery disease
- History of heart disease, stroke or transient ischemic attack (TIA)
- History of deep vein thrombosis (DVT) and/or lower leg injury
- Respiratory:
- History of episodes of chest pain, hemoptysis or pulmonary embolus
- Endocrine:
- Home glycemic control and monitoring if patient is diabetic
- Gastro-intestinal/ Genito-urinary
- Kidney function: check for hydration . [15]
- Neurological
- Ability to carry out self-care activities
- Dementia . [15]
- Musculoskeletal
- Ability to carry out self-care activities
- Skin
- Skin conditions that affect wound healing
Nutritional Assessment
- Poor diet with high caloric intake and low fibers leads to dyslipidemia, which in turn increases the risk for peripheral artery disease (PAD), metabolic syndrome, cardiovascular disease, and stroke.[16][17] See topic "How to Screen, Assess and Manage Nutrition in Patients with Wounds".
- Best practices and guideline recommend referring patients with surgical wound complications for nutritional counseling to identify nutritional and vitamin deficiencies.[3][18]
- The following tools may be utilized:
- Standardized tools such as the "Nestlé MNA" and "Self-MNA®" by Nestlé can be used to screen for malnutrition.
- Medicare Quality Payment Program, Quality Measure:
- "Process Measure: Nutritional Screening and Intervention Plan in Patients with Chronic Wounds and Ulcers"
- "Patient Reported Nutritional Assessment and Intervention Plan in Patients with Wounds and Ulcers"
- "Preventative Care and Screening: Body Mass Index (BMI) Screening and Follow-Up"
Patients’ and caregivers' concerns
Patient's and caregiver's concerns and psychosocial status should be assessed and taken into consideration when creating a treatment plan:
- Evaluate patient's concerns: pain and ability to carry out daily activities
- Medicare Quality Payment Program, Quality Measure: "Pain Assessment and Follow-Up"
- Evaluate psychosocial aspects of the patient, caregiver and family: cognitive, functional, emotional status, presence of depression, understanding of the wound and risk factors, preference for treatment, motivation for adherence to the care plan, financial concerns
- We recommend use of "Patient-Reported Outcome Tools" to assess aspects above and measure impact of interventions.[19]
Physical Examination
It is important to complete a comprehensive physical examination and head-to-toe skin assessment to determine risk of or presence of additional skin issues not directly related to the surgical interventions. [3]
- Assess for systemic signs and symptoms of infection. See section 'Background" in topic "Wound Culture - Swabs, Biopsies, Needle Aspiration"
- Complete a head-to-toe skin assessment: assess for skin tears, pressure injuries, medical-device related pressure injuries, mucosal pressure injuries and/or moisture-associated skin damage (MASD) including: incontinence-associated dermatitis (IAD), intertriginous dermatitis (intertrigo) and moisture-related damage due to tube or drain use or to peristomal issues.[3]
- If the surgical wound is on lower leg, complete a lower leg assessment. See topic "How to Assess a Patient with Chronic Wounds".
Wound Characteristics
- Wound and peri-wound assessment is best performed using a validated and reliable wound assessment tool. See section 'Ulcer exam' in topic "How to Assess a Patient with Chronic Wounds". Wound parameters to be evaluated include [20]:
- Size
- Depth
- Edges
- Undermining
- Type and amount of necrotic tissue
- Type and amount of exudate
- Skin color surrounding wound
- Peripheral tissue edema and induration
- Granulation tissue and epithelialization
- In regards to assessment of the surgical wound, timing is key. It is also important to note when to look for changes in the surgical wound. Table 3 lists outcomes and expected timeframes for positive and negative results of surgical wound healing by primary intention.[21]
Table 3. Outcomes and expected timeframes for positive and negative results of surgical wounds closed by primary intention
Parameters
| Positive outcomes
| Negative outcomes
|
Incision
| - Day 1-4: red with approximated edges
- Days 5-14: bright pink
- Day 15-1 year: pale pink
| - Day 1-4: colors of the incision may be red days 1 to 4, but there may be tension on the incision line.
- By days 5-9: the incision may no longer be well-approximated, and the tension remains.
- By days 10-14: the color may remain red or progress to bright pink, and over the next year, there may be prolonged epithelial resurfacing and/or keloid or hypertrophic scarring.
|
Scar tissue
| - Light-skinned persons will have white or silver scarring, while persons with darkly pigmented skin will progress from pale pink to darker than usual skin color.
| - There may be prolonged epithelial resurfacing and/or keloid or hypertrophic scarring.
|
Peri-incision
| - There is edema, erythema or skin discoloration, and patient may have warmth or pain
- By day 5: the above should resolve
| - There may be absence of inflammation: no edema, erythema, skin discoloration or warmth, and minimal pain at incision site.
- By days 5 - 9, the signs of inflammation may be present, extending to days 10 to 14.
- By days 10-14: may have hematoma or seroma forming
- Beyond this, healing can stall or plateau, with no healing and ongoing inflammation lasting 1 to 2 years or longer.
|
Exudate
| - Days 1-4: there is minimal/moderate sanguineous to serous exudate
- By day 5: the above should resolve
| - Days 5-9: minimal to moderate exudate, may be serosanguinous, serous or purulent;
- Any type or amount of exudate beyond that is abnormal.
|
Closure
| - By day 4: epithelial closure should be seen along the entire incision.
- Days 5-9: a healing ridge of newly formed collagen can be felt along the whole incision line
- Days 9-14: wound closure materials are removed, with skin closure strips or tape strips used after their removal.
| - For wounds closed primarily: when healing is not progressing as expected, removal of skin closures will be delayed.
- For wounds healing by secondary intention: the edges do not approximate, and the wound fails to contract.
- By day 4: there will be a lack of epithelial resurfacing of the entire incision, or it may be only partially present
- By day 14: with lack of the collagen healing ridge and dehiscence evident
- Long-term results will be keloid or hypertrophic scarring.
|
- For wounds healing by secondary intention, the expected reduction in wound size is 20-30% in the first 3-4 weeks.[22]
Diagnosis
The diagnosis of a surgical wound complication is accomplished through a comprehensive history and physical exam. For surgical wound complications on the lower limb, objective diagnostic evidence of impaired lower limb perfusion can be obtained through noninvasive arterial tests. See section 'Diagnosis' in topic "How to Assess a Patient with Chronic Wounds".
Other exams include:
- For suspected SSI: Gram stain and culture. See topics " Wound Culture - Swabs, Biopsies, Needle Aspiration" and "How to Collect a Wound Swab (Levine Technique) for Culture".
- For suspected abscess: if patient has systemic signs and symptoms of infection, presence of an undrained abscess should be ruled out. Imaging modalities include:
- For subcutaneous fluid collection: ultrasound
- For deep or organ space SSIs: computed tomography (CT scan) or magnetic resonance imaging (MRI)
- For patients with prior gastrointestinal resection and possible anastomotic dehiscence or intra-abdominal infection: imaging with oral contrast
- For hematoma or seroma: ultrasound, other imaging modalities such as CT scan and MRI.
- Fistulas: for stable patients, diagnostic imaging exams such as CT scan, gastrointestinal contrast study, or fistulogram can be performed, to help evaluate the fistula anatomy and assist with planning for surgical abdominal reconstruction.
Differential Diagnosis
Table 4 includes common differential diagnosis for surgical wound complications.
Table 4. Surgical wound complications: differential diagnosis [5]
| Conditions | Tissue/structures involved | Signs and Symptoms | Comments |
SSI: Superficial
| Only skin and subcutaneous tissue of the incision
| - Purulent drainage, localized pain or tenderness, localized swelling, erythema, or heat
|
|
SSI: Deep incisional
| Deep soft tissues of the incision (for example, fascial and muscle layers)
| Purulent drainage from the deep incision.
Deep incision that spontaneously dehisces due to SSIFever (>38°C); localized pain or tenderness.
Abscess | - Clinical diagnosis of deep SSI may be challenging, particularly in obese patients
- Imaging modalities may be helpful
|
SSI: Organ/space
| Any part of the body deeper than the fascial/muscle layers that is opened or manipulated during the operative procedure
| - Overlying skin changes may be absent
- Purulent drainage from a drain that is placed into the organ/space
- An abscess or other evidence of infection involving the organ/space
| - Imaging study demonstrates an abscess in the cavity or organs involved in the prior operation
- Gram stain and culture analysis of the drainage (percutaneous, surgical) confirms the infection.
|
Necrotizing fasciitis
| Infection may spread through any tissue
| - Soft tissue edema, erythema, severe pain (pain out of proportion to findings is an early indicator of deep fascial infection)
- Systemic illness
| - Medical emergency requiring surgical debridement
- Definitive diagnosis is surgical [23]
- See topic "Necrotizing Soft Tissue Infections And Fournier's Gangrene"
|
| Hematoma | Surgical site (soft tissue) | May occur within the first hours after surgery, due to leaking blood vessels, Can also occur later due to trauma to the surgical areaMay happen quickly causing a drop in hemoglobin | - Ultrasound or computed tomography may facilitate diagnosis
|
| Seroma | Surgical site (soft tissue) | - Upon palpation of the wound, a soft fluid wave can be felt with absence of significant pain.
| - Ultrasound or computed tomography may facilitate diagnosis
|
| Superficial incisional dehiscence (non-infected) | Skin +/- subcutaneous tissue | - Wound breakdown and separation without purulence and/or surrounding erythema
| - May occur due to excessive mechanical tension, or if sutures are removed too soon
|
Skin sensitivity to suture or wound care products
| Skin +/- subcutaneous tissue
| - Erythema, pain, pruritus, pustules, papules
| - May have history of prior skin reactions
- Braided polyester and silk sutures used intraoperatively are more likely to cause skin reactions
|
Classification
For surgical site infections
The CDC identifies the following three levels of SSI [5]:
- Superficial incisional: date of event occurs within 30 days after any operative procedure. Involves only skin and subcutaneous tissue of the incision. Signs and symptoms of infection are purulent drainage from the superficial incision AND localized pain or tenderness; localized swelling; erythema; or heat.
- Deep incisional: date of event occurs within 30 or 90 days after the operative procedure. This affects the fascial and muscle layers. Signs and symptoms are the presence of pus or an abscess AND patient has at least one of the following signs or symptoms: fever (>38°C); localized pain or tenderness.
- Organ/space infection: date of event occurs within 30 or 90 days after the operative procedure. This affects any part of the anatomy other than the actual wound incision. Signs and symptoms are drainage of pus or formation of an abscess.
For a detailed description of the criteria for each of the three categories of SSI, visit the CDC website.
Ulcer healability
Categorization of wound healability (i.e., healable, maintenance, or non-healable) is of particular importance.[24] This designation defines for the clinician, patient, and family an expected course of action, plan of care, and healing rate. A framework is summarized below. For details, see topic “How to Determine Healability of a Chronic Wound".
As a prerequisite to setting realistic treatment objectives, wounds may be classified as [24]:
- Healable: have sufficient vascular supply, underlying cause can be corrected, and health can be optimized
- Maintenance: have healing potential, but various patient factors are compromising wound healing at this time
- Non-healable/palliative wound: has no ability to heal due to untreatable causes such as terminal disease or end-of-life. It may also be that the wound is deemed “non-healable,” because major surgery is required to close the wound but the person’s health precludes that from happening.[3]
Documentation
Documenting signs of improvement to support medical necessity (Medicare):
- Documenting signs of improvement to support medical necessity (Medicare):
- Reimbursement for wound care services on a continuing basis for a particular wound in a patient requires documentation in the patient's record that the wound is improving in response to the wound care being provided.
- It is not medically reasonable or necessary to continue a given type of wound care if evidence of wound improvement cannot be shown.
- Medicare expects the wound-care treatment plan to be modified in the event that appropriate healing is not achieved. Such evidence must be documented with each date of service provided.
- Evidence of improvement includes measurable changes (decreases) of some of the following:
- Drainage
- Inflammation
- Swelling
- Pain
- Wound dimensions (diameter, depth)
- Tissue type: necrotic tissue/slough
Documentation tools
- Tools that facilitate standardized assessment and documentation should be used whenever possible:
- Validated wound assessment tools:
- Bates-Jensen Wound Assessment Tool [20]
- Wound Reference Wound Prep & Dress Tool creates notes to help support medical necessity that can be copied and pasted to electronic medical records
- Wound imaging: digital photographs at the first consultation and periodically thereafter to document progress is helpful and ensures consistency of care among healthcare practitioners, facilitates telemedicine in remote areas, and illustrates improvement to the patient.
- Wound measurement: methods include manually measuring length and width (the longest length with the greatest width at right angles), manual tracing, digital photography, and software programs that calculate wound dimensions from a photograph of the lesion. Wound tracings that calculate the area via digital software are slightly better than linear measurement.[25]
OASIS-E (home health agencies in the United States)
According to OASIS-E M1342, “Status of Most Problematic (Observable) Surgical Wound,” there are four possible choices when describing the status of a surgical wound, each with further description (see Table 5).[4]
Table 5. OASIS-E Surgical Wound Descriptors [4]
| Status | Description |
Newly epithelialized
| - the wound bed is completely covered with new epithelium; no exudate; no avascular tissue (eschar and/or slough); no signs or symptoms of infection.
- the incision site of an implanted venous access device or infusion device is healed and without signs and symptoms of infection.
|
Fully granulating
| - the wound bed is filled with granulation tissue to the level of the surrounding skin and
- has no dead space,
- has no avascular tissue;
- has no signs or symptoms of infection;
- has open wound edges.
|
Early/partial granulation
| - ≥25% of the wound bed is covered with granulation tissue;
- < 25% of the wound bed is covered with avascular tissue,
- may have dead space; and
- wound bed has no signs or symptoms of infection;
- wound has open edges.
|
Not healing
| - The wound bed has ≥25% avascular tissue – AND/OR
- signs/symptoms of infection – AND/OR
- the wound bed is clean but non-granulating – AND/OR
- wound edges are closed/hyperkeratotic – AND/OR o persistent failure to improve despite appropriate comprehensive wound management.
|
TREATMENT
Surgeons are legally responsible for the care of surgical wounds until they heal.[3] The care team should work in conjunction with the patient and caregiver to establish an appropriate treatment plan.
An adequate treatment plan for surgical wound complications aims to [3]:
- Treat the cause and other factors impeding healing
- Assess patient's and caregiver's concerns
- Provide effective local wound care
Treatment goals
Treatment goals are determined in conjunction with the patient and caregivers and are guided by healability and patient concerns. For best results, patient-driven goals should be developed based on SMART (Specific, Measurable, Attainable, Relevant, Timely) principles.
Healable surgical wounds
- For patients with a closed surgical wound healing by primary intention, the goal is for the wound to heal without complications and with minimal scar formation.
- Re-epithelialization of the uppermost approximated skin edges normally occurs within 24 to 48 hours and wound closure at 2 to 3 days.[3]
- In practice, however, sutures or staples are kept intact for 7 to 10 days and sometimes longer at the surgeon’s discretion.
- For patients with contaminated surgical wounds that are left to heal by tertiary intention (i.e. with delayed primary closure), goals include infection prevention/resolution and wound healing.[3]
- For patients with dirty or infected surgical wounds that are left to heal by secondary intention (i.e. the wound is not closed and heals when granulation tissue fills the wound from the base up), goals include infection resolution and wound healing.
Non-healable or maintenance surgical wounds
- For non-healable or maintenance surgical wounds, palliative wound care goals include prevention of ulcer progression and complications, pain relief, bioburden control. Patient's concerns and comfort should be prioritized when creating a treatment/care plan.
Management of surgical wound complications
Surgical wound complications may result from different etiologies and management interventions should address the underlying cause(s) of the complications. Interventions should include medical and surgical strategies, as well as appropriate local wound care. Table 6 summarizes management of surgical wound complications that may be seen by wound care clinicians.
Table 6. Management of surgical wound complications
| Complication | Sample conditions | Treat the cause | Local wound care |
| Surgical site infection | - Hernia repair with infected mesh
| - Medical therapy
- Antibiotic therapy
- Removal of the infected mesh may be considered if local wound care cannot manage the wounds and drainage.
| - Chronic draining areas may periodically require debridement of mesh as it protrudes out of the wound.
- Antimicrobial dressings
- Principles of chronic wound self-care should be applied along with management of bacteria, odor, exudate and periwound irritation.
|
| Hematoma | - Hematoma s/p low-transverse C-section (hematoma is the strongest independent risk factor for SSI) [3]
| - Medical therapy
- Surgical strategy:
- Hemostasis/control bleeding
- Clean/evacuate hematoma to prevent SSI
- May opt for conservative management (long-term resorption of hematoma/seroma)
| - Application of local pressure to prevent bleeding
|
| Seroma | - Seroma after application of full-thickness and partial-thickness skin grafts
| - Medical therapy
- Avoid mechanical trauma to the surgical site
- Negative pressure wound therapy or tie-over bolster dressings to eliminate dead space between graft and wound bed
| - May aspirate seroma under sterile technique
|
| Fistula | - Enterocutaneous fistula. See topic "Case: Managing High-output Enteroatmospheric Fistulas - A Case Study and Review"
| - Medical therapy
- Treat SSI and intestinal anastomotic dehiscence
| - Fistula isolation device to control output of the fistula
|
Superficial incisional dehiscence (non-infected)
| - Surgical wound dehiscence s/p abdominoplasty
| - Medical therapy
- Address technical issues with the closure of the incision
- Address mechanical stress (e.g. coughing)
- Optimize healing (e.g. manage comorbidities and SSI)
| - Understand structures (e.g. implants, vital organs, etc) located directly beneath the dehisced wound to avoid causing a more serious complication
- Secondary or delayed primary healing
- Cleansing, conservative debridement, infection and bioburden control, periwound care, moisture balance
|
Treat the cause and co-factors impeding healing
Strategies to promote resolution of surgical wounds complications include treating underlying causes, improving circulation, preventing infection and encouraging self-management.
Medical Therapy
Tobacco and nicotine cessation
- Advise patient to stop smoking and discuss referral to a smoking cessation program.
- Educational, pharmacological and behavioral techniques should be utilized.
MANAGEMENT OF PRE-EXISTING COMORBIDITIES
- Encourage patient to monitor pre-existing illnesses such as stroke, peripheral vascular disease, diabetes mellitus, renal disease, or cardiac disease and consult a provider if changes occur.
MANAGEMENT OF DIABETES
- 1AFor patients with and without diabetes, implement perioperative glycemic control and use blood glucose target levels less than 200 mg/dL in patients with and without diabetes (Grade 1A) [26]
Optimize nutrition and hydration
- Maximize the patient's nutritional status through adequate calorie intake and high protein supplements. See topic "How to Screen, Assess and Manage Nutrition in Patients with Wounds".
Control pain
- Pain interferes with deep breathing and coughing, and limits movements
- Depending on the surgery type, post-operative pain can be either nociceptive or neuropathic, or a combination. Pain control can be achieved with:
- Use of analgesics at regular intervals rather than only as needed
- Use of nonsteroidal anti-inflammatory agents (NSAIDS) to help to reduce the amount of opiates required in the acute phase, thus reducing opioid side effects.
Local wound care
Local wound care should follow the wound bed preparation principles.[27][28]
Cleansing
- For surgical wounds healing by primary intention within 48 hours after surgery, clinicians may opt to use a non-touch aseptic technique with sterile cleanser (e.g. saline). After 48 hours, showering is often permitted.[3]
- For surgical wounds that dehisce, are to heal by secondary intention or have an increased bacterial load, after 48 hours clinicians may consider cleansing with potable tap water, boiled and cooled water, sterile saline or wound cleansers.[3]
Debridement
- For healable wounds with necrotic or infected tissue, debridement should be performed.
- Choice of type of debridement varies according to several factors. See topic "Debridement".
Infection and bioburden control
- If an infection is suspected, a semi-quantitative wound swab for culture and sensitivity should be done to determine appropriate antibiotic intervention.
- When an SSI occurs, treatment will be based on assessment of patient, infection and organism factors.
- See topic "Antimicrobial Stewardship in Wound Care".
Periwound skin care
- To protect the periwound skin from moisture-associated skin damage (MASD) due to prolonged exposure to exudate, clinicians might opt to choose a dressing capable of absorbing wound exudate and wicking it from the periwound skin, and to change the dressing at a frequency that prevents skin irritation.[3]
- The volume and composition of the wound exudate are key factors that contribute to development of MASD. Wound exudate containing heparin-binding proteins, proteolytic enzymes, high concentrations of inflammatory cytokines or high bacterial burden and their associated toxins all increase the likelihood of MASD.[3]
Moisture balance
Selection of primary and secondary dressings should follow established wound bed preparation principles.[29][30] For customized, wound-specific recommendations, use Wound Prep and Dress Tool. For practical information on dressing indications, contraindications, application, Medicare coverage and brands see "Dressing Essentials" and "Dressing Brands Quick Reference". General recommendations and evidence regarding dressings for surgical wounds are listed below.
- For surgical wounds, dressing selection is determined by the type of closure (primary, delayed primary or secondary intention), the amount of wound exudate and the presence of any tunnels, undermining, underlying vascular grafts or hardware. Consideration should also be given to patient concerns, caregiver knowledge and time, setting and available resources.60,90,91
Primary intention
- For incisions closed by primary intention, clinicians might opt for application of a dry, sterile semipermeable cover dressing for 24 to 48 hours.[3]
- The wound is expected to re-epithelialize within 2-3 days. [31]
Secondary intention
- For acute surgical wounds healing by secondary intention, clinicians might consider interactive advanced wound products over wet gauze to maintain a moist wound environment and prevent bacterial penetration of the wound.
- For heavily exudative wounds, use of a wound pouch may be considered.[3]
Negative Pressure Wound Therapy
- For incisions closed by primary intention, negative pressure wound therapy (NPWT) can be considered to promote wound healing and reduce SSI rates. [32]
- NPWT can be applied in the operating room under sterile conditions and stay in place for five to seven days so as to remove exudate, reduce the lateral incisional tension and reduce hematoma or seroma formation at the surgical site. [33]
- For open wounds, NPWT can be used as a method of bolstering flaps and skin grafts.
- Patient selection, wound bed preparation, debridement, dressing selection, application and clear interprofessional team communication is crucial for successful use of NPWT.
- See topic "How to Select and Apply Negative Pressure Wound Therapy Devices".
Plan Reassessment
- Assessment and measurements of the wound need to be recorded at least once a week.
- Re-epithelialization of the uppermost approximated skin edges normally occurs within 24 to 48 hours and wound closure at 2 to 3 days.[3]
- Wounds healing by secondary intention that fail to reduce in size by 20-30% in the first 3-4 weeks should be reassessed. [22]
PREVENTION
Several clinical practice guidelines provide guidance on the prevention of surgical wound complications:
- NICE guideline: Surgical site infections: prevention and treatment [32]
- Centers for Disease Control and Prevention Guideline for the Prevention of Surgical Site Infection [26]
- WHO Global guidelines for the prevention of surgical site infection [18]
CODING
OASIS-E
According to OASIS-E Guidance Manual, Section M: Skin Conditions [4]
- A surgical site closed primarily (with sutures, staples, or a chemical bonding agent) is generally described in documentation as a surgical wound until re-epithelialization has been present for approximately 30 days, unless it dehisces or presents signs of infection. After 30 days, it is generally described as a scar and should not be included as a surgical wound on OASIS-E.[4]
- Old surgical wounds that have resulted in scar or keloid formation are not considered current surgical wounds and should not be included in this item.
- An incision line is considered a surgical wound. The staple or suture sites are not considered surgical wounds.
Pressure ulcer/injury:
- Any type of flap procedure performed to surgically replace a pressure ulcer is reported as a surgical wound, until healed. It should not be reported as a pressure ulcer/injury on M1311.
- A pressure ulcer treated with any type of graft is no longer reported as a pressure ulcer/injury, and until healed, should be reported as a surgical wound on M1340.
- A pressure ulcer that has been surgically debrided remains a pressure ulcer and should not be reported as a surgical wound on M1340.
Ostomy:
- A bowel ostomy is excluded as a surgical wound, unless a "take-down" procedure of a previous bowel ostomy is performed, in which case the surgical take-down produces a surgical wound. A bowel ostomy being allowed to close on its own is excluded as a surgical wound.
- All other ostomies are excluded from consideration under this item and should not be counted as surgical wounds.
- There are many types of "ostomies," all of which involve a surgically formed opening from outside the body to an internal organ or cavity. Examples include cystostomy, urostomy, thoracostomy, tracheostomy, gastrostomy, etc.
Other types of surgical wounds:
- Other examples of surgical wounds include, but are not limited to:
- Orthopedic pin sites,
- Central line sites (centrally inserted venous catheters),
- A PICC line (peripherally inserted venous catheter), either tunneled or non-tunneled, is NOT a surgical wound, when it is peripherally inserted
- Wounds with drains,
- Medi-port sites and other implanted infusion devices, or
- Venous access devices
HCPCS
Incision and Drainage (I&D)
How to choose between two codes for I&D of a superficial skin abscess [34]:
- 10060 Incision and drainage of abscess; simple of single
- 10061 Incision and drainage of abscess; complicated or multiple
The difference between a simple and complicated I&D is that a complicated I&D contains:
- Multiple incisions
- Drain placements
- Probing to break up loculations
- Extensive packing or
- Subsequent wound closure
Documentation must also state if abscess is superficial or deep and location. If the abscess is deep, code choice is based on the location of the abscess and is not dependent simply on single versus multiple, and simple versus complicated. Appearance and signs and symptoms can assist with determining simple versus complex.[34]