Recognizing the urgency to adopt strategies to help prevent AMR, the Joint Commission implemented an antimicrobial stewardship standard (MM.09.01.01) for the Hospital, Critical Access Hospital, and Nursing Care Center accreditation programs on January 1, 2017.[5] Subsequently in 2020, new antimicrobial stewardship requirements also became applicable to Joint Commission accredited ambulatory health care organizations that routinely prescribe antimicrobial medications, which includes wound care clinics (Standard MM.09.01.03).[11]
This topic provides guidance on how institutions can minimize misuse and/or overuse use of antimicrobial medications and establish an antimicrobial stewardship program. For a list of Guidelines, Quality Measures and other resources on Wound Infection, see topic "Wound Infection - Overview". For evidence-based guidance on diagnosis of wound infection, when and how to perform wound cultures, how to choose sampling techniques, documentation, coding, coverage and reimbursement, see topic "Wound Culture - Swabs, Biopsies, Needle Aspiration". For sample policies and procedures on how to collect a wound swab see topic "How to Collect a Wound Swab (Levine Technique) for Culture". For guidance on when and how to perform wound biopsies see topic "How to Perform a Wound Biopsy".
Background
Definitions
- Antibiotic: antimicrobial agent with the ability to kill or inhibit bacterial growth.[12]
- Antimicrobial: an agent or substance derived from any source (microorganisms, plants, animals, synthetic or semi-synthetic) that acts against any type of microorganism, such as bacteria (antibacterial), mycobacteria (anti-mycobacterial), fungi (antifungal), parasite (anti-parasitic) and viruses (antiviral). All antibiotics are antimicrobials, but not all antimicrobials are antibiotics.
- Antimicrobial resistance (AMR): AMR occurs (as a result of misuse, such as over-prescribing) when certain pathogens fail to respond to treatment with antimicrobials. Resistance is the inherited ability of microorganisms to grow in the presence of antibiotics, regardless of the duration of treatment.[13]
- Antimicrobial tolerance: alongside resistance, tolerance is another method by which bacteria evade antibiotic treatment. Tolerance prolongs the duration of treatment that bacteria can sustain, for example by remaining dormant. Dormancy protects bacteria from the lethality of many types of antibiotics, whose action requires growth (e.g. beta-lactams and quinolones) as long as they remain dormant.[13]
- Antimicrobial stewardship: a coherent set of actions which promote the responsible use of antimicrobials. This definition can be applied to actions at the individual level as well as the national and global level, and across human health, animal health and the environment.[12] Antimicrobial stewardship is further defined as ‘an inter-professional effort across the continuum of a patient’s care that involves the timely and optimal selection of antimicrobial agents, their doses and the duration of their use'; the aim is to achieve the best clinical outcome with minimal toxicity to the patient and the environment.[3]
- According to the British Society for Antimicrobial Chemotherapy, one of the main AMS principles in the clinical environment is to prescribe "The right antibiotic, for the right patient, at the right time, with the right dose, and the right route, causing the least harm to the patient and future patients." [3]
- Antimicrobial stewardship program: An organizational or system-wide health-care strategy to promote appropriate use of antimicrobials through the implementation of evidence-based interventions.[12]
- Antiseptic: An antiseptic is a topical antimicrobial agent with broad-spectrum activity that inhibits multiplication of, and can kill microorganisms. Depending upon its concentration, an antiseptic may have a toxic effect on human cells; however, modern antiseptics show good tissue compatibility.[14]
- Even though development of resistance to topical antiseptics is uncommon, antibiotic stewardship principles need to be applied to topical antiseptics due to some isolated topical antiseptic resistance and cross-resistance to oral antimicrobials. [14][15]
Relevance
- AMR is now a leading cause of death in worldwide.[1] It has been estimated that in 2019, almost 5 million deaths were associated with bacterial AMR.[1] AMR is caused by genetic modifications of microorganisms (e.g. bacteria, fungi, viruses, and parasites) upon exposure to antimicrobial drugs (e.g. antibiotics, antifungals, antivirals, antimalarials, and anti-helminthics.[12]
- Misuse and/or overuse of antimicrobial drugs largely contribute to AMR. A study showed that in both outpatient and inpatient settings, up to 50% of antibiotic treatment courses are unnecessary or inappropriate.[3] The resulting new generation of ‘superbugs’ is not easily treated with existing drugs.[16]
- By default, chronic wounds are contaminated by several types of bacteria.[7][8] Because chronic wounds are often recurrently infected, patients with chronic wounds may undergo repeated courses of antibiotic therapy, increasing the risk of AMR.[2] For instance, a study in Norway showed that in primary care, most patients with wounds are given antibiotics inappropriately.[17]
- Antimicrobial misuse in patients with wounds is often a result of clinicians' uncertainty related to the presence of bacterial infection in the wound, insufficient knowledge on when to treat with antibiotics, fear of having a bad outcome, or patient demands.[3]
- To successfully prevent AMR, it is essential that all wound care professionals, especially those prescribing, dispensing and using antimicrobial agents, be aware of their appropriate and inappropriate use.[2] This goal can be accomplished most effectively through the practice of antimicrobial stewardship in wound care.
ANTIMICROBIAL STEWARDSHIP IN WOUND CARE
AMS in wound care generally includes the principles listed in Table 1 [3]:
Table 1. Antimicrobial stewardship principles in wound care
| Strategy/ Principle | Comments |
Only prescribe antibiotics for wounds that are clinically infected
| - Do not administer antibiotics for clinically uninfected/ colonized/ contaminated wounds, or for non-bacterial infections
- Wound infection is primarily diagnosed by clinical findings, usually supported by microbiological data
|
When antimicrobial therapy is indicated, prescribe an appropriate regimen [i.e. the narrowest spectrum for the likely or proven pathogen(s)]
| - Before starting antibiotic therapy, obtain optimal specimens for culture. If the patient is clinically stable, consider discontinuing any active antibiotic before taking culture
- Select empirical antibiotic therapy based on available clinical and laboratory data
- Cover likeliest pathogens, based on clinical presentation and local antibiotic resistance data
- Aim for narrow-spectrum regimen, unless host is severely ill or immunocompromised
|
Use antimicrobial with the least risk for adverse effects for the patient and the community
| - Revise (and constrain) therapy based on clinical response and culture/ susceptibility results
- If clinically responding, attempt to narrow antimicrobial spectrum; change regimen from parenteral to oral
- Consider switching to topical therapy or non-antibiotic antimicrobial
- In a polymicrobial infection, it is often unnecessary to treat low virulence bacterial species
|
Aim for optimal dose, appropriate route and correct therapy duration (i.e. just long enough to achieve resolution of the signs and symptoms used to diagnose infection)
| - Treat only until all clinical evidence of infection has resolved, irrespective of wound healing rate
- Duration of therapy is usually 1 to 2 weeks for soft tissue, and about 6 weeks for bone infection
|
Algorithm 1 summarizes a simplified approach to antimicrobial therapy for wounds. Main steps discussed below include: diagnosis of infection, specimen collection/ processing/ reporting, and infection management, including the use of topical antimicrobials, and use of empiric and definitive antibiotic therapy.
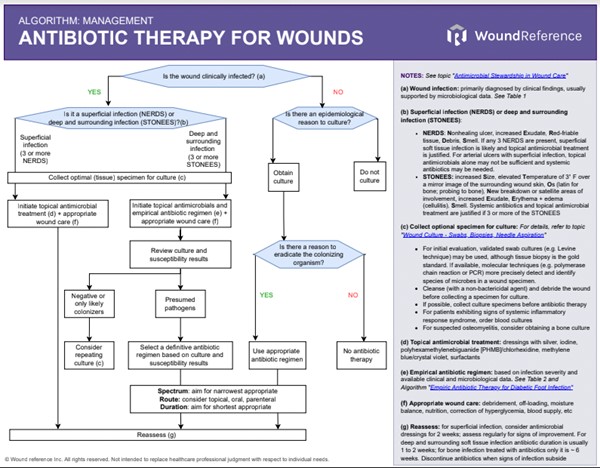
Algorithm 1. Antibiotic Therapy for Wounds (click to on this link to enlarge)
Diagnosing infection
- Frequency of assessment: patients should be assessed for wound infection at the first, and at every subsequent wound assessment.[3]
- Diagnosis: wound infection is primarily diagnosed by clinical findings, usually supported by microbiological data.[3]
- For signs and symptoms associated with stages of the wound infection continuum, see section 'Relevance' in topic "Wound Culture - Swabs, Biopsies, Needle Aspiration'.
- In general, wound infections can be characterized as superficial or deep and surrounding infections [18][19][20]:
- Superficial infection: The NERDS mnemonic can help identify soft tissue infection. If any 3 NERDS are present, superficial soft tissue infection is likely and topical antimicrobial treatment is justified. Systemic antibiotics may not be needed. For arterial ulcers with superficial infection however, topical antimicrobials alone may not be sufficient and systemic antibiotics may be necessary.[21][22] NERDS stand for:
- Nonhealing ulcer
- increased Exudate
- Red-friable tissue
- Debris
- Smell
- Deep and surrounding infections: The STONEES mnemonic can help identify deep and surrounding infections. Systemic antibiotics and topical antimicrobial treatment are justified if 3 or more of the STONEES signs are present. STONEES stand for:
- increased Size,
- elevated Temperature of 3° F over a mirror image of the surrounding wound skin
- Os (latin for bone): probing to bone
- New breakdown or satellite areas of involvement,
- increased Exudate
- Erythema + edema (cellulitis)
- Smell
Specimen collection
Culturing clinically infected wounds is helpful in defining pathogens and their antibiotic susceptibilities. Tissue biopsy or quantitative, validated swab cultures (e.g. Levine technique), biofluorescent scans, or molecular techniques (e.g. polymerase chain reaction or PCR) may be used to confirm diagnosis of infection. For details, see topics "Wound Culture - Swabs, Biopsies, Needle Aspiration", "How to Collect a Wound Swab (Levine Technique) for Culture" and "How to Perform a Wound Biopsy".
- For optimal specimen collection, consider the following [3]:
- To lessen the likelihood of identifying colonizers (rather than pathogens), cleanse (with a non-bactericidal agent) and debride the wound before collecting a specimen for culture
- If possible, collect culture specimens before starting antibiotic therapy
- For patients exhibiting signs of systemic inflammatory response syndrome, blood cultures should be ordered
- For suspected osteomyelitis, consider obtaining a bone culture
It is important to note that all open wounds become colonized. Therefore, prescribing antibiotic therapy as a result of a positive culture in the absence of clinical signs and symptoms of infection is not recommended.[3]
Infection management
- For chronic wounds without clinical signs of infection, clinical guidelines and expert opinion recommend against routine use of antimicrobial dressings or systemic antibiotics.[3][19][20][23][24][25][26]
- For healable chronic wounds, interventions to manage infection include topical antimicrobial agents (i.e. antiseptics or topical antibiotics), systemic antibiotics and debridement.[19][3] To determine healability of a wound, see topic "How to Determine Healability of a Chronic Wound".
- For superficial infections of chronic wounds, clinical guidelines and expert opinion suggest use of topical antimicrobial agents to decrease bacterial load and promote wound healing.[19][3] For arterial ulcers with superficial infection however, topical antimicrobials alone may not be sufficient and systemic antibiotics may be necessary.[21]
- Topical antimicrobial agents for clinical use are usually classified as antiseptics and topical antibiotics.[27] Due to their local effect, a small amount of topical antimicrobial can achieve high levels at the site of infection without systemic adverse effects.[3] The optimal topical treatment strikes a balance between antimicrobial efficacy and tissue tolerance. [28]
- Antiseptics include silver, iodine, polyhexamethylenebiguanide [PHMB]/ chlorhexidine, methylene blue/crystal violet, surfactants, acetic acid.
- For example, acetic acid has bacteriostatic and bactericidal activity against many gram-negative and gram-positive organisms, particularly Pseudomonas.[29] However, repeated or prolonged use of antiseptic agents beyond short-term application to target pathogens and control infection, can have harmful effects, potentially delaying healing.[28] For guidance on selection of antimicrobial dressings, see "Interactive Feature Matrix: Antimicrobials".
- Topical antibiotics include bacitracin, neomycin, metronidazole, mupirocin, etc. [20][27][30] Use of topic antibiotics for non-infected chronic wounds is not recommended, as there is limited evidence of their effectiveness and they often select for resistant colonizing bacteria.[3][19]
- Antibiotic stewardship emphasizes minimizing unnecessary antibiotic exposure. In severe or persistent cases of bacterial skin infections, antibiotics are often prescribed as first-line therapy. However, for localized infections, clinicians may consider initiating treatment with topical antibiotics. For example, in methicillin-resistant Staphylococcus aureus (MRSA) local/superficial skin infections - which commonly cause cellulitis, impetigo, and infections secondary to atopic dermatitis - topically administered agents such as mupirocin ointment has been shown to be as effective as oral antibiotics.[30][31][32]
- If the wound is healable and the cause of the ulcer is addressed, improvement is usually expected within 4 weeks.[20]
- For deep, spreading and/or systemic infections, systemic antibiotics are recommended, in addition to topical antimicrobial dressings.[19][3]
- Empirical antibiotic therapy: prior to obtaining culture and susceptibility results, initial antibiotic therapy is empirical, that is, based on best guess of the causative pathogens and their antibiotic susceptibility, informed by available clinical and epidemiological data.[3] Clinical guidelines offer helpful guidance on empirical therapy selection, however choices should ultimately be tailored to local circumstances such as local antibiotic resistance data and the institution's AMS strategy.[3] For guidance on empirical antibiotic therapy for diabetic foot infection, see 'Algorithm for Selection of Empiric Antibiotic Therapy for Diabetic Foot Ulcer Associated with Infection' in topic "Diabetic Foot Ulcer Associated with Infection - Management". For guidance on empirical antibiotic therapy for deep and surrounding infection, see 'Algorithm: Antibiotic Therapy for Wounds' above.
- Definitive antibiotic therapy: once culture and susceptibility results are available, antibiotic therapy should be adjusted according to the test results and the clinical response to empirical therapy.[3] When the infection has clinically responded and microbiological results are available, consider narrowing the spectrum of antibiotic therapy, changing from intravenous to oral therapy with an agent with good orally bioavailability, or discontinuing therapy if signs and symptoms of infection subside.[3] For deep and surrounding soft tissue infection antibiotic duration is usually 1 to 2 weeks.
- For bone infection (osteomyelitis), primarily surgical or primarily medical (antibiotic therapy) strategies can be used. Selection of approach should be based on several factors such as patient condition and preference, mobility, comorbidities and others. For diabetic foot osteomyelitis treated primarily with antibiotics and no bone surgical resection, patients should receive antibiotics for 6 weeks.[33] See details in section 'DFU with Osteomyelitis' in topic " Diabetic Foot Ulcer Associated with Infection - Management".
- For non-healable/maintenance ulcers that are infected, keeping the ulcer dry, topical antiseptic agents (e.g., povidone iodine or chlorhexidine), and systemic antibiotics are usually appropriate.[19]
- For guidance on infection and bioburden management for specific conditions see below:
- Arterial ulcer: refer to section 'Infection' in topic "Arterial Ulcer - Treatment"
- Venous leg ulcer: see section 'Local Wound Care - Infection Management' in topic "Venous Ulcers - Treatment and Prevention"
- Diabetic foot ulcer: see topic "Diabetic Foot Ulcer Associated with Infection - Management"
- Pressure ulcers/injuries: see section 'Local Wound Care - Infection Management' in topic "Pressure Ulcers/Injuries - Treatment"
ANTIMICROBIAL STEWARDSHIP PROGRAM IN WOUND CARE
The Joint Commission has developed a new standard in the Medication Management (MM) chapter (Standard MM.09.01.03) with 5 new elements of performance (EPs) addressing antimicrobial stewardship in the ambulatory setting.[11]
The new EPs align with current recommendations from scientific and professional organizations and address the following concepts:
- Identifying an antimicrobial stewardship leader.
- Establishing an annual antimicrobial stewardship goal.
- Implementing evidence-based practice guidelines related to the antimicrobial stewardship goal.
- Providing clinical staff with educational resources related to the antimicrobial stewardship goal.
- Collecting, analyzing, and reporting data related to the antimicrobial stewardship goal
The Joint Commission's comments are highlighted in Boxes 1 to 3.
Box 1. The Joint Commission's expectations for an antimicrobial stewardship program - Antimicrobial Stewardship Program Components (Manual: Critical Access Hospital) [5]
Antimicrobial Stewardship Program Components
- The organization needs to have a document indicating how each of the core elements listed MM.09.01.01, EP 5 is addressed in its antimicrobial stewardship program. This information can be located in a separate document or can be included in other antimicrobial stewardship documents. Documentation does not have to be provided in a lengthy format but needs to describe how the core elements are addressed in the antimicrobial stewardship program.
|
Identifying an antimicrobial stewardship leader
Antimicrobial resistance is growing, so improving the use of antimicrobial medications across the care continuum is a patient safety priority. Identifying an individual(s) to be accountable for an organization’s antimicrobial stewardship activities increases the likelihood of success by establishing clear lines of accountability. Identifying an individual(s) for this role also demonstrates an organizational commitment to improving the use of antimicrobial medications. Antimicrobial stewardship activities may be the individual(s) primary job responsibility or may be in addition to other duties.[11]
The clearest choice to satisfy EP-1, identifying an antimicrobial stewardship leader, is the medical director of the wound care center. Medical directors use their dedicated nonclinical time to direct the program, direct education, and assist in data collection.[34]
Establishing an annual antimicrobial stewardship goal
Establishing an annual antimicrobial stewardship goal allows the organization to focus its efforts on a specific opportunity to improve antimicrobial prescribing practices. Organizations may consider reviewing their prescribing practices or consulting available national data to identify areas of inappropriate antimicrobial medication use. The organization’s protocols and guidelines, education resources, and measurement activities should align with the annual goal.[11]
There are several possible choices for EP-2. Examples of antimicrobial stewardship goals may include decreasing the use of antibiotics or addressing overuse of a specific medication.[11]
After establishing the goal, the next step is to select parameters to measure progress towards the goal. Examples of measures to be tracked include:
- Number of intravenous antibiotic days [35]
- Number of oral antibiotic days [35]
- Justified use of antibiotic [35]
- Number of antibiotic prescriptions written per patient and per clinic [34]
- The use of topical antimicrobials in the clinic and prescribed [34]
- The combination of oral antibiotics and topical antimicrobials [34]
- The reduction in bacterial fluorescence in patients over time [34]
- Treatment failures [34]
- Healing outcomes [34]
Implementing evidence-based practice guidelines related to the antimicrobial stewardship goal
It is important for organizations to implement evidence-based practice guidelines or to develop protocols to improve the appropriateness of their antimicrobial prescribing. Using protocols and guidelines will help ensure that patients who need antibiotics receive them while those without an indication do not. [11]
- For a list of evidence-based practice guidelines see topic "Wound Infection - Overview".
Providing clinical staff with educational resources related to the antimicrobial stewardship goal
Clinical staff need to be educated about the organization’s annual antimicrobial stewardship goal so that patients receive clear, consistent recommendations when antimicrobials are not indicated. Educational resources may include recommended prescribing practices (including medication selection, dosing, or duration) and strategies and interventions to explain why some patients may not need an antibiotic, such as addressing patient expectations and discussing possible adverse effects of antibiotics.[11]
Box 2. The Joint Commission's expectations for an antimicrobial stewardship program - Education (Manual: Critical Access Hospital) [5]
Education - Although Joint Commission surveyors will not be reviewing staff or medical staff/licensed independent practitioner records on education received regarding antimicrobial resistance and antimicrobial stewardship, they will inquire about the type of education provided by the organization. During patient tracers, surveyors may ask staff and licensed independent practitioners about the education they have received. Organizations may consider providing written material such as the organization's antibiogram (if available) to demonstrate compliance.
|
Educational resources within WoundReference include topics, algorithms and competencies such as:
- Algorithm 'Antibiotic Therapy for Wounds'
- How to Assess a Patient with Chronic Wounds
- Wound Culture - Swabs, Biopsies, Needle Aspiration
- How to Collect a Wound Swab (Levine Technique) for Culture
- How to Perform a Wound Biopsy
- Acute Burns: for assessment, see section 'Infection Assessment' in topic "Acute Burns - Introduction and Assessment", for management, see section 'Infection Management' in topic "Acute Burns - Treatment"
- Arterial Ulcers: for assessment, see section 'Ulcer Complications' in topic "Arterial Ulcers - Introduction and Assessment", for management see section 'Infection' in topic "Arterial Ulcer - Treatment"
- Venous Ulcers: for assessment, see section 'Ulcer Exam' in topic "Venous Ulcers - Introduction and Assessment", for management, see section 'Local Wound Care - Infection Management' in topic "Venous Ulcers - Treatment and Prevention"
- Diabetic Foot Ulcers: for assessment, see section 'Diabetic Foot Infection' in topic "Diabetic Foot Ulcer - Introduction and Assessment", for management, see topic "Diabetic Foot Ulcer Associated with Infection - Management" and algorithm "Empiric Antibiotic Therapy for Diabetic Foot Infection"
- Pressure Ulcers/Injuries: for assessment, see section 'Infection Associated with PU/PI' in topic "Pressure Ulcers/Injuries - Introduction and Assessment", for management see section 'Local Wound Care - Infection Management' in topic "Pressure Ulcers/Injuries - Treatment"
Collecting, analyzing, and reporting data related to the antimicrobial stewardship goal
It is important to evaluate whether an organization’s antimicrobial stewardship activities are successful and to share with leadership any progress toward achieving the antimicrobial stewardship goal. Organizations may incorporate electronic or manual data collection and analysis methods to identify improvements, though a sustained effort is often required before organizations see change in the culture of antimicrobial prescribing.[11] Of note, data may include antimicrobial medication prescribing patterns, antimicrobial resistance patterns, or an evaluation of the antimicrobial stewardship activities implemented.[11]
Box 3. The Joint Commission's expectations for an antimicrobial stewardship program - Data Collection and Analysis and Improvement Opportunities (Manual: Critical Access Hospital) [5]
Data Collection and Analysis - The Joint Commission does not specify the type of antimicrobial stewardship data that organizations should collect. It is the organization's responsibility to identify which antimicrobial stewardship data it will collect, analyze, and report. Organizations are encouraged to review the CDC's Core Elements of Hospital Antibiotic Stewardship Programs at https://www.cdc.gov/getsmart/healthcare/pdfs/core-elements.pdf and The Core Elements of Antibiotic Stewardship for Nursing Homes at https://www.cdc.gov/longtermcare/prevention/antibiotic-stewardship.html for examples of measures that can be used to collect antimicrobial stewardship data.
Improvement Opportunities - Organizations need to use the antimicrobial stewardship data they have collected and analyzed to identify improvement opportunities for their antimicrobial stewardship program. If the data demonstrate that antimicrobial stewardship improvements are not necessary, the organization should share the data with the surveyor. If improvements are identified, the organization should be prepared to discuss the actions taken to improve the program.
|
Sample Framework
A sample framework for implementation of an antimicrobial stewardship program can be summarized as follows:
- Choose goals and measures as listed in section 'Establishing an annual antimicrobial stewardship goal' above.
- Perform a baseline assessment of the current status, to compare with data collected after the intervention is completed. For instance, an organization might opt to pull data related to the selected metrics corresponding to the preceding 2-6 months.
- Implement intervention to achieve goal: interventions might include education, training, competencies as described in section 'Providing clinical staff with educational resources related to the antimicrobial stewardship goal' above, or other types of interventions such as methods to improve diagnosis of infection, or antibiotic prescribing practices
- After the intervention is completed, perform a post intervention assessment: re-evaluate the same metrics evaluated during the baseline assessment. For instance, an organization might opt to pull data related to the selected metrics corresponding to 2-6 months after intervention was implemented, and compare with the data obtained with the baseline assessment.
Other Resources
- Resources from the Joint IDSA-SHEA-PIDS Task Force on Antimicrobial Stewardship: Antimicrobial stewardship resources
- Patient education materials (The Joint Commission): Speak Up Antibiotics Know the Facts
- Decision support tool (AHRQ): AHRQ Minimum Criteria for Antibiotics Tool
- Toolkit (World Health Organization): Antimicrobial stewardship programmes in health-care facilities in low- and middle-income countries. A practical toolkit.