ABSTRACT
Lymphedema is a chronic disease and the goal of lymphedema treatment is to reset patients to a subclinical or latency stage, regardless of their stage at diagnosis. Conservative management of lymphedema with adequate compression therapy [e.g. typically as part of complex decongestive therapy (CDT)] remains the first-line treatment for chronic lymphedema. Surgical intervention may be of benefit to a few, well-selected individuals who remain refractory to conservative treatment.[1] General surgical indications include lymphedema refractory to conservative management, frequent infections or impaired quality of life. Expert committees and clinical guidelines recommend at least 6 months of conservative management with compression therapy before any surgical intervention for lymphedema is undertaken.[2][3] See algorithm for management of lymphedema in topic "Lymphedema - Surgical Management".
Care coordination: It is recommended that patients under consideration for surgical lymphedema management be referred to centers with an expert in lymphatic microvascular reconstruction and plastic surgeons with experience treating lymphedema patients. For guidance on finding specialists, see "Specialist Finder" or contact authors.
Preoperative assessment: Patients being considered for surgical management should have been following a personalized treatment based on a comprehensive assessment.
Surgical interventions: There are three general surgical approaches to lymphedema: 1) excisional operations (also known as debulking or ablative), 2) liposuction, 3) lymphatic reconstructions (also known as physiologic or functional procedures).[1] Excisional operations and liposuction are reductive techniques that aim at reducing the volume of the affected limb. Lymphatic reconstructions aim at restoring or improving lymph flow in the affected area with microsurgical lymphatic reconstruction procedures.[1] This section provides details on specific indications, patient selection criteria and outcomes for each type of procedure.
In general, the American Venous Forum suggests [3]:
- 2CExcisional operations or liposuction only to patients with late stage non-pitting lymphedema, who fail conservative measures (Grade 2C)
- 2CMicrosurgical lymphatic reconstructions for selected patients with secondary lymphedema, if performed early in the course of the disease, and by centers of excellence (Grade 2C)
Ultimately, all options for surgical management are practitioner dependent and should be suggested according to provider experience and patient preference and specific condition. Surgical procedures require the long-term use of compression therapy to maintain therapeutic results.[4]
SURGICAL MANAGEMENT
Overview
This topic covers surgical management of lymphedema with focus on surgical indications and selection criteria. For management of lymphedema and its complications (e.g., infection, ulcers) see topic "Lymphedema - Treatment and Emerging Strategies for Prevention". For clinical guidelines and quality measures specific to lymphedema, see "Lymphedema - Overview". For evidence-based review and assessment of lymphedema including epidemiology, risk factors, etiology, pathophysiology, history, physical examination, diagnosis, differential diagnoses, documentation and ICD-10 coding, see "Lymphedema - Introduction and Assessment". For patient education, see topic "Patient education - Lymphedema".
Background
Lymphedema is a chronic disease. Management and patient adherence to available treatment modalities is often challenging. The goal of lymphedema treatment is to reset patients to a subclinical or latency stage, regardless of their stage at diagnosis.
Conservative management of lymphedema with adequate compression therapy [e.g. typically as part of complex decongestive therapy (CDT)] remains the first-line treatment for chronic lymphedema. Surgical intervention may be of benefit to a few, well-selected individuals who remain refractory to conservative treatment.[1] Expert committees and the American Venous Forum recommend at least 6 months of conservative management with compression therapy before any surgical intervention for lymphedema is undertaken.[2][3]
Care Coordination
- Patients under consideration for surgical lymphedema management should be referred to centers with an expert in lymphatic microvascular reconstruction and plastic surgeons with experience treating lymphedema patients. These specialists are more likely found in tertiary centers. For guidance on finding specialists, see "Specialist Finder" or contact authors.
- At these centers, patients are evaluated and carefully selected. A summary on indications and selection criteria are provided in this topic.
Preoperative Assessment
Patients being considered for surgical management have should have been following a personalized treatment based on a comprehensive assessment that:
- Includes comprehensive history and physical examination
- Identifies underlying causes of lymphedema (i.e., primary or secondary) and assesses risk factors contributing to development of lymphedema and its complications (e.g. ulcers, infection)
- Identifies conditions associated with lymphedema (e.g. if on lower limbs: chronic venous insufficiency, peripheral artery disease, etc)
- Differentiates from other types of edema (systemic or local), which require different treatments
- Identifies pertinent patient's/caregiver's concerns
- Stages lymphedema using a validated classification system, such as the one created by the International Society of Lymphology (ISL) [2]:
- Stage 0: subclinical disease, swelling absent despite underlying impaired lymphatic transport (at risk for lymphedema)
- Stage I: tissue swelling present but reduced by limb elevation. Pitting may occur
- Stage II: limb swelling partially reduced by elevation, pitting is manifest. Later in stage 2, limb may not pit due to excess subcutaneous fat or fibrosis
- Stage III: lymphostatic elephantiasis with skin changes (e.g., acanthosis, thickening, further fibrosis, warty overgrowths) and recurrent infections
- Assesses lymphedema severity: within each stage above, severity can be assessed and classified according to simple volume differences of the affected part, that is, the change from baseline in swelling not due to weight change (i.e, if limbs, determined by measuring both limbs). Severity can be classified as follows [2]:
- Minimal: typically > 5 to < 20% increase in limb volume
- Moderate: 20-40% increase
- Severe: > 40% increase
- Some clinics prefer to use > 5 to < 10% as minimal and > 10 to < 20% as mild
- Includes imaging modalities if applicable:
- Duplex ultrasonography: if lymphatic reconstruction is under consideration, to rule out venous disease
- Lymphoscintigraphy: allows visualization of lymphatics anatomy and surgical planning.
Surgical Interventions for Lymphedema
There are three general surgical approaches to lymphedema: 1) debulking/ ablative/ excisional operations, 2) liposuction, 3) lymphatic reconstructions.[1] Ultimately, all options for surgical management are practitioner dependent and should be suggested according to provider experience and patient preference and specific condition. Surgical procedures require the long-term use of compression therapy to maintain therapeutic results.[4]
- Excisional surgery (also known as debulking or ablative): aims at reducing volume of the limb by excision of the excess cutaneous and fibrous overgrowth. Excisional procedures may be performed alone or with lymphatic reconstructions.[1]
- Liposuction: aims at reducing volume of the limb by removal of excessive adipose tissue.[1]
- Lymphatic reconstruction (also known as physiologic or functional procedures): aims at restoring or improving lymph flow in patients with lymphedema with microsurgical lymphatic reconstruction procedures.[1] With the advent of advanced microsurgical techniques, surgical management of lymphedema has evolved significantly in the past decades. For selected patients, microsurgical functional procedures may now represent the closest chance for a cure of lymph flow disorders.[2]
In general, the American Venous Forum suggests [3]:
- 2CExcisional operations or liposuction only to patients with late stage non-pitting lymphedema, who fail conservative measures (Grade 2C)
- 2CMicrosurgical lymphatic reconstructions in centers of excellence for selected patients with secondary lymphedema, if performed early in the course of the disease (Grade 2C)
Excisional surgery
indications for Excisional surgery with lymphatic reconstruction
- For patients with advanced lymphedema (ISL Stage 3) that is refractory to conservative management and have recurrent local skin infections in the lower limb (e.g. toe web, foot, lower calf) or recurrent septic attacks of lymphangitis of increasing frequency (>3 per year), clinicians might opt for lymphatic reconstruction followed by excisional surgery and CDT to reduce limb volume and control infection.[1][3]
- Rationale: The therapeutic benefits of surgical management are enhanced when lymphatic reconstruction and excisional procedures are combined to restore lymphatic flow and reduce fibrosis, respectively.[5] After lymphatic flow is established with CDT and a permanent lymphatic reconstruction method, ISL Stage 3 lymphedema usually shrinks leaving excess subcutaneous fat, fibrous tissue and skin. Excisional procedures can then be carried out serially at a minimum interval of 6 months to 3 months. Positive, long-lasting outcomes heavily depend on patient adherence and commitment to life-long CDT.[1]
- Excisional surgery with lymphatic reconstruction for lymphedema is usually covered by Medicare and other private insurance.
indications for Excisional surgery without lymphatic reconstruction
- For well-selected patients with refractory, end-stage chronic lymphedema (ISL Stage 3) and with repeated episodes of infection (>3 year), clinicians might consider excisional surgery without lymphatic reconstruction as an adjunct to CDT to improve efficacy of subsequent CDT.[1]
- Rationale: When advanced lymphedema stops responding to CDT due to excessive fibrosis and edema, frequency of soft tissue infection typically increases. Excisional surgery leads to volume reduction and excision of fibrosis, thereby improving the efficacy of subsequent CDT and compression bandaging.[1] The single most important factor for satisfactory clinical improvement is adherence to post-operative lifelong CDT, thus patients need to be carefully selected.
- Excisional surgery for lymphedema is usually covered by Medicare and other private insurance.
- For pediatric lymphedema, the most commonly reported procedures are excisional, which mirrors the adult literature.[6]
- If patients have advanced end-stage chronic lymphedema and palliative needs, excisional surgery alone over excisional surgery with lymphatic reconstruction is indicated. For other advanced chronic lymphedema cases, the decision to associate lymphatic reconstruction to excisional surgery is made by the lymphedema surgeon and patient, on a case-by-case basis.
Types of excisional surgery
Generally, excisional surgery consists of excision of fibrofatty tissue formed due to lymphatic fluid stasis. Excisional surgery offers the greatest amount of lymphedema reduction by volume.[4] These procedures include:
- Charles procedure (Figures 1 to 4): complete excision of affected skin and subcutaneous tissue of the lower extremity while preserving the deep fascia, with subsequent coverage by skin graft.
- Sistrunk procedure: excision of affected skin, soft tissue and deep fascia of the upper extremity through a medial arm incision.
- Thompson procedure: modified Sistrunk procedure with a lateral arm incision and de-epithelialized skin flaps embedded along the neurovascular bundle.
- More modern techniques that have been employed at lymphedema centers often in association with lymphatic reconstructions include serial elliptical excisions of fibrotic/ hyperkeratotic tissue and resection of redundant skin and subcutaneous tissue (with fibrotic lymphatics) beneath the skin leaving pedunculated flaps.[1] Serial elliptical excisions are more suitable to cases where the lymphedematous limb is not predominantly fibrotic, as fibrosis and skin tension resulting from skin excisions hamper adequate closure of surgical wounds. If the skin of the affected limb is predominantly fibrotic, Charles procedure may offer better results.
Complications of surgical debulking procedures include acute infection, seroma, hematoma, blood loss necessitating transfusions, and graft or minor flap failure requiring more surgery.[7][8]
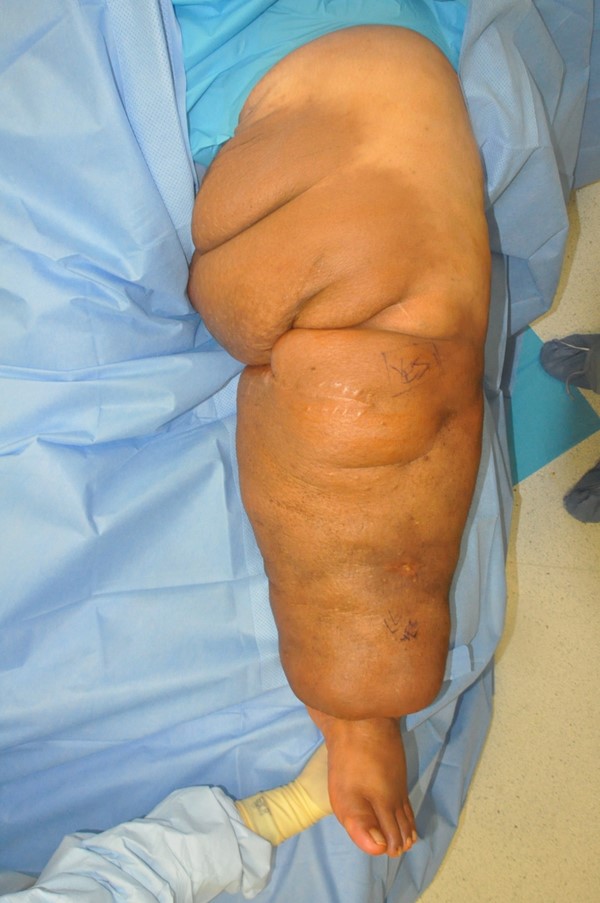
Fig. 1. Charles procedure. Pre-operative frontal view | 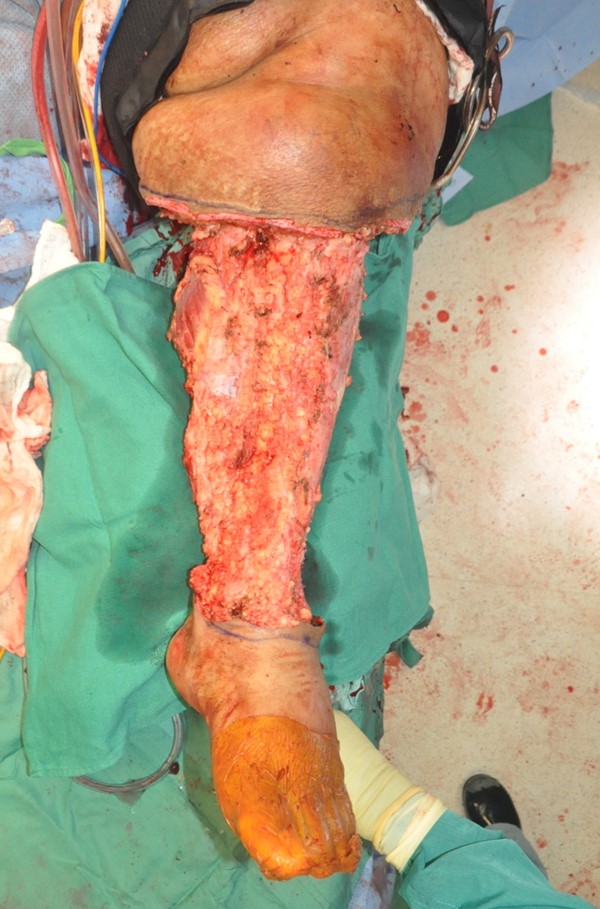 Fig. 2. Charles procedure. Intra-operative frontal view
|
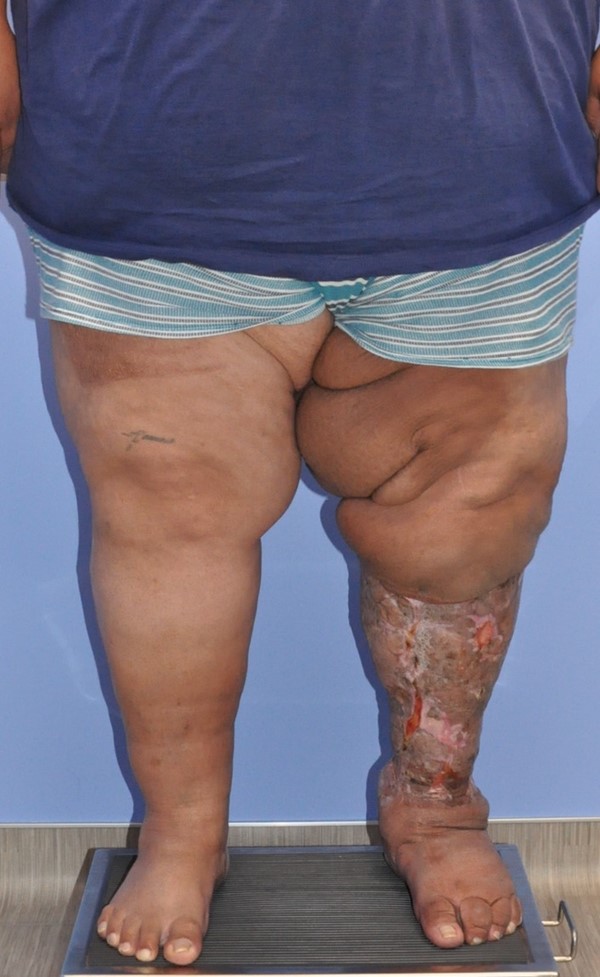
Fig. 3 Charles procedure. Post-operative frontal view | 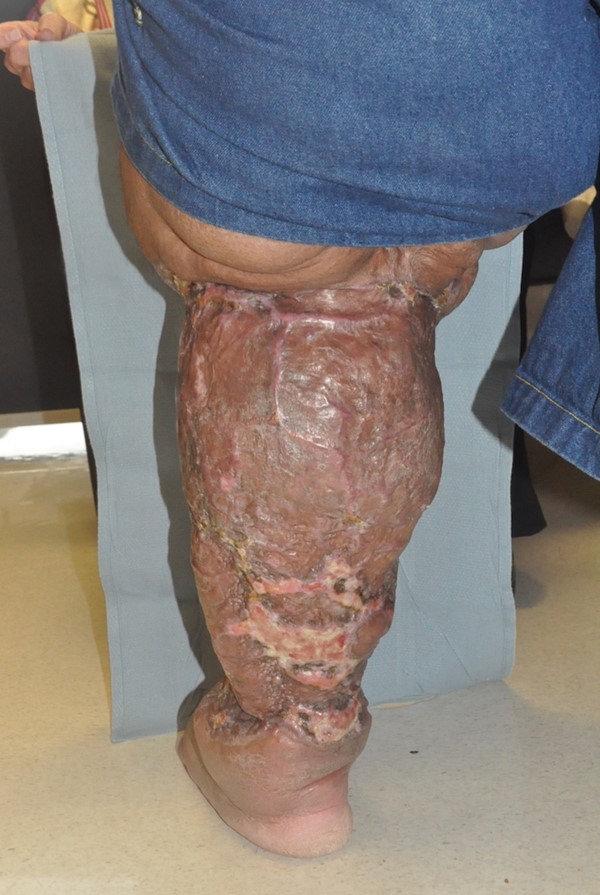
Fig. 4 Charles procedure. Post-operative back view |
Liposuction
Liposuction consists in aspiration of subcutaneous fat using a suction cannula.
Indications for liposuction in patients with lymphedema
- For patients with upper or lower extremity late-stage lymphedema and hypertrophied adipose tissue, without pitting edema or fibrotic changes, and that are refractory to conservative management, clinicians might opt for liposuction with or without lymphatic reconstruction, accompanied by life-long compression garments.[1][9]
- Rationale: chronic lymphedema is frequently associated with hypertrophied adipose tissue. Adipose tissue that remains after a period of adequate CDT may be removed with liposuction and is associated with improved patients’ quality of life and reduced incidence of erysipelas (i.e. infection of the upper dermis and superficial lymphatics).[1][10][11][12] Liposuction has been used in both upper and lower limbs, but it is said to be more effective in the upper extremity.[13] A study showed that liposuction combined with controlled compression therapy reduces arm lymphedema more efficiently than the therapy alone.[14] Liposuction should never be performed in a patient with pitting edema. Accumulated lymph should be removed with CDT pre-operatively. After liposuction, patients are required to undergo lifelong compression therapy with compression garments.[15] Liposuction is frequently associated with lymphatic reconstruction. Surgical planning (i.e., liposuction first followed by lymphatic reconstruction or the other way around) is individualized and decided by the lymphedema surgeon and patient.
- Liposuction for lymphedema is typically not covered by Medicare and other private health insurers.
- Complications: Liposuction is considered a relatively safe procedure with quick recovery and few complications, usually limited to paresthesias.[7]
Lymphatic reconstruction
Physiological treatment is based on microsurgery techniques that aim at establishing lymph flow in a lymphedematous limb. Three main techniques of lymphatic reconstructions have been developed: lymphovenous anastomosis, lymphatic grafting, and lymph node transplantations.[1] Evidence supporting use of lymphatic reconstructions to reduce lymphedematous limb volume is mainly derived from uncontrolled or retrospective studies and remains of low certainty.
General Indications for lymphatic reconstruction
The American Venous Forum suggests that microsurgical lymphatic reconstructions be performed in centers of excellence for selected patients with secondary lymphedema, early in the course of the disease.[3] Patients need to be carefully selected to enhance chances of successful outcomes.
- Indications include [1]:
- Less than 50% reduction of lymphedema with medical and physical therapy correctly conducted (generally regarded as 4-6 weeks of intensive CDT and 6-12 months of maintenance CDT)
- Recurrent episodes of lymphangitis
- Intractable pain
- Worsening limb function
- Patient preference
- Contraindications include:
- Lymphatic fibrosis (advanced lymphedema)
- Congenital hypoplasia, or even aplasia of the lymph vessels, as seen in those with primary lymphedema
types of Lymphatic reconstruction techniques
Lymphovenous anastomosis
- Specific indications: Lymphovenous anastomosis has been used to treat iliac lymphatic obstruction in patients with distal lymphatic patency.[7][16] Better results are obtained if performed in early stages of lymphedema.[3][17] It has been the most frequently used physiological procedure for pediatric lymphedema, although prospective data on the efficacy of this procedure is sparse.[6]
- Techniques (Figures 5 to 7): Two techniques have been introduced, lymph node-to-vein and lymph vessel-to-vein anastomoses [1]:
- The lymph node-to-vein technique consists in anastomosing healthy transected inguinal lymph nodes distal to the lymphatic obstruction to the saphenous or the femoral vein.
- The lymph vessel-to-vein technique consists in anastomosing healthy lymphatic vessels distal to the lymphatic obstruction to neighboring veins (e.g. saphenous, femoral vein). The latest techniques using super-microscopic surgery use very high power magnification and enable anastomosis of smaller lymph vessels (< 1mm in diameter). See Figures 5 to 7
- Outcomes: Clinical improvement after lymph-node-to-vein anastomoses has been reported in a few uncontrolled studies. As for lymph vessel-to-vein techniques, some uncontrolled studies have shown long term clinical improvement. A case series of 655 patients showed subjective improvement in 87% of the patients, reduction in limb volume in 69% patients, and discontinuation of conservative treatment for 85% patients.[1][18][19] For lower extremity lymphedema, lymphovenous anastomosis has been reported to result in reduced above-knee circumference, body weight, episodes of cellulitis, and improved Lymphedema Quality of Life questionnaire scores.[20]
- Complications: The most common complications reported following lymphovenous anastomosis are re-exploration of the anastomosis, venous reflux, and surgical site infection.[8]
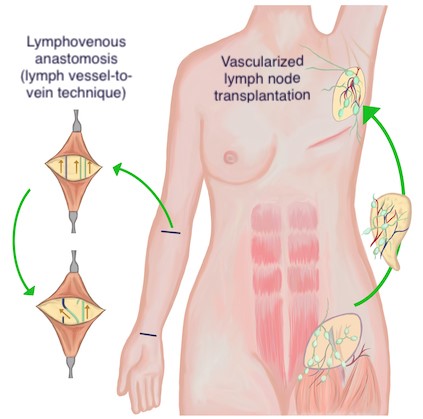 Fig. 5. Lymphatic reconstruction techniques. Left: Lymphovenous anastomosis (LVA), lymph vessel-to-vein. Right: Vascularized lymph node transplantation Fig. 5. Lymphatic reconstruction techniques. Left: Lymphovenous anastomosis (LVA), lymph vessel-to-vein. Right: Vascularized lymph node transplantation
| 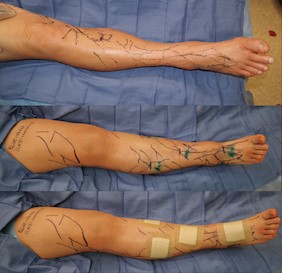
Fig. 6. Intraoperative surgical planning shows mapping of veins (purple) and mapping of incisions used to look for lymphatic vessels to anastomose to venous system (green). Five LVA’s were performed in total. Source: Dr. Ketan M. Patel. |
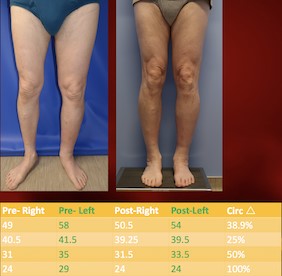
Fig. 7. Pre and post-operative aspect of patient who underwent LVA for the treatment of lymphedema secondary to pelvic lymph node dissection and radiation therapy for bladder cancer. Source: Dr. Ketan M. Patel. |
|
Lymphatic grafting
- Specific indications: Lymphatic grafting has been used to bypass axillary, iliac or iliofemoral lymphatic obstructions.[1] Better outcomes have been reported when lymphatic grafting is performed in early stages of lymphedema.[17]
- Techniques: Techniques include lymphatic-lymphatic anastomosis and lymphatic-venous-lymphatic anastomosis.[7]
- Lymphatic-lymphatic anastomosis (LLA): healthy lymphatic vessels are harvested from a donor site and anastomosed to recipient lymphatics in the affected limb
- Lymphatic-venous-lymphatic anastomosis (LVLA): an autologous vein graft is harvested from a donor site and anastomosed to lymphatic vessels proximal and distal to the site of lymphatic blockage
Outcomes: Case series have reported sustained long-term improvement in limb volume. For instance, a study with 55 patients reported continued limb volume improvement in 80% of patients 3 years after surgery.[1][21] Drawbacks to bypass procedures include long incisions at donor and recipient sites, and the predisposition to lymphedema in the lymphatic donor region.[7] Complications for bypass procedures are low, including primarily nonhealing wounds, cellulitis and lymphatic fistulas.[7]
Lymph node transplantation
- Specific indications: Lymph node transplantation has been used for patients with earlier stages of reversible lymphedema and unaffected lymph nodes at distant sites.[16]
- Technique (Figure 5): Vascularized lymph node transplant (VLNT) involves free tissue transfer of vascularized lymph nodes from accessible donor sites, with subsequent anastomosis to recipient blood vessels in the affected region.[7]
- Outcomes: A case series has reported decreased limb volume 5 years after transplantation in 22 out of 24 patients.[1][22] Another small case series that compared lymph node transplantation and lymphovenous anastomosis in lower limb lymphedema suggested that lymph node transplantation may result in better outcomes as measured by limb volume reduction, fewer episodes of infection and improved qualify of life.[20]
- Complications: Most common complications include delayed wound healing, seroma and hematoma formation, lymphatic fluid leakage, iatrogenic lymphedema, soft-tissue infection, venous congestion, marginal nerve pseudoparalysis, and partial flap loss.[8]
EMERGING SURGICAL PREVENTATIVE INTERVENTIONS
- 2CFor patients undergoing lymphadenectomy, we suggest consultation with a specialized reconstructive microsurgeon for consideration of prophylactic lymphovenous bypass to prevent development of lymphedema (Grade 2C). See section 'Prevention - Emerging Strategies' in topic "Lymphedema - Treatment and Emerging Strategies for Prevention".