Algorithm for Management of Skin |
TREATMENT
Overview
This topic covers management and prevention of skin tears (STs). For an introduction and assessment of STs including epidemiology, risk factors, etiology, pathophysiology, history, physical examination, diagnosis, differential diagnoses, documentation and ICD-10 coding, see topic "Skin Tears - Introduction and Assessment".
Background
STs are acute wounds, best managed with customized treatment plans based on the patient’s input and based on a comprehensive assessment that:
- Identifies/controls any active bleeding
- Identifies presence and repositions any remaining skin flap, which in this context refers to a portion of the skin (epidermis/dermis) that is unintentionally separated from its original place due to shear, friction and/or blunt force (and not intentionally detached from its place of origin for therapeutic use).[7]
- Classifies the ST using a validated classification system, such as the one developed by the The International Skin Tear Advisory Panel (ISTAP)
- Identifies underlying causes and assesses risk factors contributing to the development of STs
- Differentiates from other types of wounds, which require different treatments
- Identifies pertinent patient's/caregiver's concerns
- Determines ulcer prognosis or "healability", that is, the potential of the ulcer to heal with conservative treatment only.[8][9] For details, see 'Ulcer healability (Prognosis)' in "Skin Tear - Introduction and Assessment":
An adequate treatment plan for STs aims to[8][9]:
- Treat the cause and other factors impeding healing
- Assess patient's and caregiver's concerns
- Provide effective local wound care
If a healable ST does not show any signs of improvement within 7-21 days despite adequate treatment, treatment plan should be reassessed, differential diagnoses re-evaluated and adjunctive therapies considered.[2]
See Algorithm for Management of Skin Tears below (Algorithm 1)
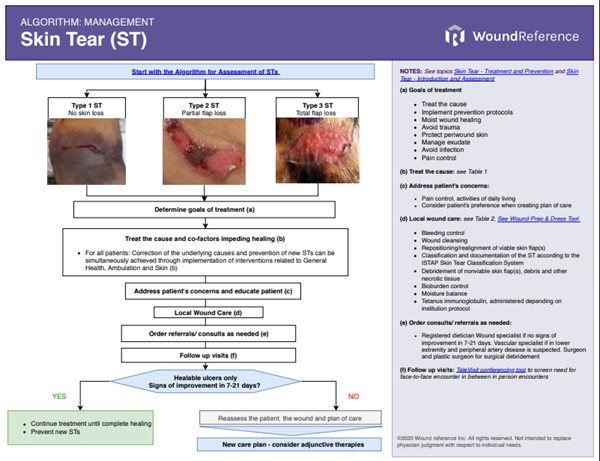
Algorithm 1. Algorithm for Management of Skin Tears (click on this link to enlarge)
Treatment Goals
- General treatment goals for STs include:
-
- Bleeding control
- Identification and prevention of underlying causes
- Maintenance of moisture balance within the wound
- Protection of periwound skin
- Prevention of trauma and infection
- Pain management
- Recovery of skin integrity
- Specific treatment goals vary according to the healability of a ST, that is, the potential for a ST to heal.[2]
-
-
Healable ST:A healable ST has adequate blood supply, and can be healed if the underlying cause is addressed. Treatment goals are listed above.[2]
-
Maintenance ST: Maintenance wounds have the potential to heal. However, the underlying cause of the wound cannot be corrected due to the patient's unwillingness to adhere to treatment, or to limitations, errors or barriers of healthcare resources (e.g., uncontrolled limb edema delaying/preventing healing of the ST, uncontrolled diabetes, malnutrition). These factors may change over time, so they should be monitored and re-evaluated periodically.[8]
-
Non-healable ST: these ulcers lack healing potential due to terminal diseases or end-of-life status; these patients are usually under palliative care.[2]
-
- For patients under palliative care, the general treatment goals listed above also apply, to the extent desired/tolerated by each patient. [2] In addition, the presence of risk factors such as immobility, excessive moisture, nutritional deficiencies, friction and shearing and impaired activities of daily living, exposes these individuals to a high risk of STs. Thus, an individualized plan must be implemented in order to reduce risks associated with STs and prevent infection.[5]
Treat the cause
- For all ST patients (with healable, non-healable or maintenance ST), expert consensus recommends that healthcare professionals identify and implement a personalized treatment plan that corrects the underlying causes or co-factors affecting skin integrity, and that takes into consideration the patient’s needs, the wound, and environmental/system challenges.[2]
Correction of the underlying causes and prevention of new STs can be simultaneously achieved through implementation of the interventions illustrated in Tables 1-3, adapted from the ISTAP Skin Tear Prevention Strategies.[1][2][5]
Table 1. ISTAP Skin Tear Prevention Strategies - General Health
| Risks/ underlying causes |
Comments |
Patient strategies |
Caregiver/ healthcare professional |
|
General Health
|
|
Impaired visual, sensory, auditory status
|
- Chronic and critical diseases can lead to sensory, auditory, and visual changes. Aging is frequently associated with chronic diseases (diabetes, dementia, and vascular disease), and increased risk of skin injury and delayed wound healing.
- Sensory, auditory, and visual changes have a strong correlation with episodes of falls, which lead to the occurrence of STs.
|
- Participate actively in care.
- Be familiar with the surroundings/ environment.
|
- Provide a safe environment.
- Assess footwear.
- Ensure that individuals avoid the use of clothes that can injure the skin (e.g., clothes that are too tight-fitting or that have a zipper).
- Educate caregivers on visual, sensory, auditory impairment, and associated risks for STs.
|
|
Cognitive impairment
|
- Altered levels of cognition in elderly persons, children, and individuals with chronic and critical illnesses lead to an increased risk for STs.
- Aggressive behavior, agitation associated with altered-cognition, and the presence of dementia favor the risk of trauma and self-injury, resulting in STs.
|
- Participate actively in care.
- Be cognizant of environment.
|
- Assess the patient’s cognitive status.
- Educate caregivers concerning safety and the risk of STs.
- Protect the patient from self-mutilation where possible.
|
|
Impaired nutrition
|
- Both obese and undernourished patients may be at risk for STs.
- Upon nutritional assessment and monitoring, look for Braden nutrition subscale values (1: very poor, 2: probably inadequate), body mass index (<20 kg); involuntary weight loss; masticatory difficulties; loss of dentures or teeth; and abnormal laboratory values of zinc, albumin, and prealbumin.
- If a patient consumes less than 50% of his meals for more than three days, consult the nutritionist for evaluation.
- Malnutrition can cause delayed wound healing and increase the risk of infection.
- Assess dehydration by monitoring urine output, skin turgor, and laboratory values of sodium and serum osmolarity.
- Fluid and electrolyte supplementation is often necessary.
- Vitamins A, B, C, D; and iron, zinc, copper are essential to maintaining healthy skin. Supplementation may be necessary. Clinical correlation is required.
|
- Optimize nutrition and hydration.
|
- Refer to a nutritionist to optimize nutrition and hydration.
- Encourage and monitor nutrition and fluid intake Increase fluid intake appropriately.
- Patients in extremes of weight (obese, underweight, or cachectic) require special care to avoid STs
|
|
Polypharmacy
|
- Polypharmacy is the use of multiple medications that can predispose a patient to drug interactions or adverse drug reactions, especially among the elderly.
- Be aware that the use of antibiotics, antihypertensives, analgesics, tricyclic antidepressants, antihistamines, anticoagulants, antineoplastic drugs, antipsychotic drugs, diuretics, oral hypoglycemic agents, non-steroidal anti-inflammatory drugs, oral contraceptives, and steroids can cause skin inflammation or various adverse reactions.
- Use corticosteroids with caution, they can interfere with collagen synthesis, epidermal regeneration, and can increase the incidence of STs.
|
- Patients need to be aware of the potential for skin changes with certain medications.
- Discuss all medications with prescriber or pharmacist
|
- Consult with interprofessional team to monitor the patient’s skin for potential signs of polypharmacy.Discontinue medications that increase the incidence of STs.
|
Table 2. ISTAP Skin Tear Prevention Strategies - Mobility
| Risks/ underlying causes |
Comments |
Patient strategies |
Strategies for caregiver/ healthcare professional |
|
Mobility
|
|
Issues related to mobility
|
- Altered mobility limits normal physical movement of the body.
- Injuries are often related to the use of wheelchairs, falls, transfers, trauma and confusion that arise from bumping into objects.
- Healthcare professionals must be alert and identify patients with changes in mobility
|
- Participate actively in care.
- Be familiar with surroundings/ environment.
|
- Use safe handling techniques and equipment (e.g., use repositioning slings and sliders to reposition patient)
- Monitor and perform daily skin assessments to look for issues caused by mobility devices
- Adjust care for patients in extremes of weight (obese, underweight and cachectic patients).
- Pad hard objects/devices in the patient’s environment (e.g. bedrails, wheelchairs, table corners)
- Encourage the individual to wear long sleeves, pants, socks, etc, to protect the skin against trauma
|
|
Assistance with activities of daily living
|
- Activities of daily living include daily activities such as: putting on /taking off clothes, bathing, positioning, and transfer from one place to another.
- Individuals often develop STs during these daily activities, especially those who depend on caregivers
|
- Participate actively in care.
- Be familiar with the surroundings/ environment
|
- Protect patient against trauma during routine care and activities of daily living.
- Use appropriate techniques for transfers and positioning.
- Promote a safe environment.
- Educate care providers individually on appropriate techniques for transfer and positioning.
|
|
History or risk of falls
|
- A fall is a sudden and unintentional change in the position of the body, which may lead to a ST.
- Several factors predispose an individual to falls. Examples include unstable gait and balance, muscular weakness, impaired vision, side effects of certain medications, dementia, loose carpets, disorganization of the environment, poorly adapted shoes, inadequate illumination, urinary urgency, and neurological diseases.
|
- Participate actively in care.
- Be familiar with the surroundings/ environment
- Use assistive devices (canes and walkers) if those are prescribed.
- Participate s in activities for the prevention of falls
|
- Initiate a program for the prevention of falls for the unit/institution/workplace and involve pediatric and neonatal patients whenever appropriate.
- Create a safe environment.
- Eliminate disorder from the surroundings.
- Get certified in handling techniques and safe equipment
|
|
Mechanical trauma (not related to mobility aids)
|
- Risk for mechanical trauma and a subsequent ST is increased by skin immaturity or skin changes due to aging, acute or chronic illnesses, use of adhesives, repositioning.
- Among neonates, mechanical trauma is the leading cause of STs
- Intrinsic, genetic, biological, and age factors present with susceptibility to developing injuries. Immaturity of the skin, skin changes by aging, skin affected by acute and chronic illnesses, use of adhesives, bathing, and repositioning increase the risk of mechanical trauma.
|
- Participate actively in care.
- Bec familiar with the surroundings/ environment
- Practice care actions for the reduction of the incidence of STs.
- Tidy up/organize the surrounding environment .
- Avoid use of adhesives on the skin.
|
- Provide individuals at risk protective clothing such as long-sleeved shirts, long pants, knee-length socks, and cushions for elbow protection.
- Ensure a safe environment, e.g., pad the bed rails, ensure wheelchair has leg rest, and close drawers and other objects that can cause trauma.
- Remove unnecessary objects from the patient’s environment and keep it well-illuminated.
- Use appropriate positioning/transfer techniques when moving the patient.
- Provide extra skin protection (e.g. with clothing) for individuals at extremes of weight (obese, underweight, or cachectic).
- Educate care providers about the prevention and treatment of STs.
|
Table 3. ISTAP Skin Tear Prevention Strategies - Skin [5]
| Risks/ underlying causes |
Comments |
Patient strategies |
Strategies for caregiver/ healthcare professional |
|
Skin
|
|
- STs often develop in individuals with extremes of age and critically ill patients.
- The skin suffers changes over time; atrophy of the subcutaneous tissue, especially of the hands, legs, and plantar region of the feet.
- The reduction of elasticity, resistance and moisture, senile purpura, and inappropriate skin cleansing products make the skin vulnerable to tears.
|
- Participate actively in care.
- Be familiar with the surroundings/ environment.
- Recognize the risks of STs and how to minimize them.
- Practice behaviors that will decrease the incidence of STs.
|
- Implement safe behaviors, such as applying moisturizer—without perfume after bathing—to damp skin, not wet (moisturize two times per day).
- Use lukewarm water for bathing.
- Use liquid soap with a neutral pH for cleaning the skin.
- Provide protective clothing, such as long-sleeved shirts, long pants or knee-high socks or shin/elbow guard pads.
- Avoid adhesive products on fragile skin. If necessary, use silicon tapes with non-adherent contact layer, non-adherent foam dressing, or other topical dressings specific for STs. Ensure the appropriate removal of all adhesives.
- Both the care provider and patient should keep short nails, removing hardened edges to avoid self-inflicted STs.
- Educate care providers about the importance of “gentle care.”
|
Risk prevention strategies for healthcare facilities include:
- Implementation of a ST risk reduction program that contemplates strategies such as:
-
- Nutrition and hydration optimization
- Increased awareness that some medications/polypharmacy increase risk for ST
- Strategies to minimize risks associated with impaired mobility
- Falls risk assessment and risk reduction
- Assessment of prevalence and incidence of STs in wound rounds/audit program.
Patient-centered concerns
For all patients (with healable, non-healable or maintenance STs), concerns should be addressed and plan of care adjusted accordingly.
Pain control
- It is recommended that clinicians manage pain by addressing the underlying causes of pain and administering analgesics.
- Pain management can be achieved with the following interventions/techniques [10]:
-
- Use of appropriate dressing for each type of wound
- Atraumatic dressing removal:
-
- Consider allowing patients to remove their dressings themselves
- Administer pain medication as per institutional pain protocol
- Bioburden control and prevention of infection, since infection leads to increased pain
Local wound care
STs are acute wounds - thus when treating STs, adequate local wound care is accomplished with the following steps [2][5]:
- Bleeding control
- Wound cleansing
- Repositioning/realignment of viable skin flap(s)
- Classification and documentation of the ST according to the ISTAP Skin Tear Classification System (see section ‘Classification’ in topic "Skin Tear - Introduction and Assessment")
- Debridement of nonviable skin flap(s), debris and other necrotic tissue
- Bioburden control
- Moisture balance
- Tetanus immunoglobulin, administered depending on institution protocol
The sections below elaborate on each step.
Bleeding control (hemostasis)
- As STs are acute wounds, the first step is to control any residual bleeding by:
-
- Applying pressure on the ST, after covering it with saline-moist gauze.
- Raising the affected limb, if the ST is on a limb and if no contraindications are present.
- Applying calcium alginate on the ST in addition to light pressure (calcium alginate has hemostatic properties)
Cleansing
- Expert consensus suggests cleansing STs with non-cytotoxic solutions (e.g. potable water, normal saline, nonionic surfactant cleanser) to remove debris, dried blood and exudate.[11]
-
- Cleansing should be performed at a low pressure (<8 psi) to prevent damage to any remaining fragile skin tear flap (Type 1 or 2 skin tears) or the fragile periwound skin. [2][5]
- For uncomplicated STs, potable water is considered a reliable substitute for sterile saline.[11]
ST skin flap repositioning/realignment
- Upon cleansing, assess presence of any remaining skin flap (i.e. the skin is torn but still attached). If a viable skin flap is present, expert consensus recommends it be rolled back to its original position.[2][5]
-
- Utilize a moist cotton tip applicator, gloved finger or forceps to reposition the skin flap.
- If the skin flap is difficult to align, consider application of a moistened non-woven gauze compress on the ST for 5-10 minutes until the skin flap is rehydrated, and then attempt to reposition it again.
- A viable skin flap may not cover the entire wound bed, but it should be positioned to cover as much area as possible.
- The repositioned skin flap should not be disturbed for at least five days so that it adheres to structures underneath it.
Classification of the ST and documentation
- After cleansing and repositioning any remaining skin flap(s), the ST can be assessed, classified, and documented. See section ‘Classification’ in topic "Skin Tear - Introduction and Assessment")
Debridement
- For all STs, expert consensus recommends debridement of non-viable tissue and debris.[2]
- For STs with skin flaps (ISTAP types 1 and 2), expert consensus recommends debridement of any non-viable portions of the skin flap.[2]
- Rationale: Wound debridement is the process of removing devitalized tissue and infected or foreign material from the wound to promote wound healing.[2] Necrotic tissue serves as a nidus for infection and may lead to increased inflammatory reactions within the wound, which delays healing.
Indications and contraindications to debridement
See debridement indications and contraindications in section ‘General indications and contraindications’ in the topic “Debridement”
Caution
- Prior to debridement, if ST is on a lower limb, a thorough vascular assessment should be performed. See table with values and interpretations of non-invasive arterial tests in the topic "How to Select Adequate Compression Therapy Pressure Levels and Products"
-
Tetanus vaccine prior to debridement: Administer tetanus immunoglobulin, according to institutional policy, to patients who have not been vaccinated with tetanus toxoid in the last ten years. The immunoglobulin should be given before debridement to prevent potential bacterial exotoxin release by wound manipulation.[2]
Debridement techniques
- Wound debridement may occur naturally by autolysis - enhanced by moisture-balanced dressings - or by other methods (chemical/enzymatic, biological/larval, mechanical, sharp conservative debridement, or surgical).[2] For more details on debridement methods, see topics " Debridement" and "How to Perform Conservative Sharp Wound Debridement".
- For STs with skin flaps (ISTAP types 1 and 2), differentiating between viable and non-viable portions of their skin flaps can be challenging. Often, skin flaps may simultaneously present with ecchymosis, senile purpura and hematomas. During debridement, care should be taken to ensure that viable skin flaps are left intact and fragile skin is protected.[2]
-
- To determine if the remaining skin flap is viable:
-
- Inspect skin flap to assess tissue perfusion; viable tissue is warm and pink; however, nonviable tissue is dry and pale.[12]
- Assess blood flow through the capillary refill test and observe bleeding patterns of the skin flap.[12]
- If the ST is on an extremity and its skin flap has a length-to-width ratio beyond 1:1, there might be an inherent higher risk of necrosis.[13] A ST skin flap can be viewed as skin flap with random pattern vascularization. Random pattern flaps are based on the subdermal vascular plexus of the skin. For random pattern flaps to be viable, in the best of conditions, the maximum length-to-width ratio on the extremities should be 1:1.[13]
- If in doubt, consult a wound specialist before debridement.[2]
Bioburden control (infection/ inflammation)
STs are acute wounds. Thus, signs of inflammation on a ST may be due to the trauma that initially caused it. It is important that healthcare professionals be able to distinguish inflammation from infection prior to treating for infection.[2][5]
- For local infection, topical antibiotics and antimicrobial dressings are recommended. Systemic or spreading infection requires treatment with systemic antibiotics in addition to topical antibiotics and antimicrobial dressings. [5]
- Tetanus immunization status should be evaluated as well.[5]
Soft tissue local infection
- The NERDS mnemonic (Nonhealing ulcer, increased Exudate, Red-friable tissue, Debris, Smell;) can help identify soft tissue infection. If any three NERDS are present, soft tissue infection is likely.[8]
- For healable, locally infected STs: Debride the wound and collect wound specimen for culture and sensitivity. Topical antimicrobial agents may be used (e.g., silver sulfadiazine cream and other silver products, medical-grade honey, polyhexamethylene biguanide, cadexomer iodine). Topical dressings must be gentle on fragile skin in order to prevent further trauma.
-
If there are no signs of infection, but no signs of wound healing (contraction or epithelialization) within 2 weeks from debridement: biofilm may be preventing wound healing (biofilm is a cluster of bacteria embedded in a matrix covering the wound).[14]
Systemic infection
- For healable/ non-healable/ maintenance individuals with clinical evidence of spreading or systemic infection (e.g., positive blood cultures and fever, cellulitis, fasciitis, osteomyelitis, systemic inflammatory response syndrome, or sepsis): initiate systemic antibiotics and topical antimicrobials.
Peri-wound care
- Care must be taken not to damage the periwound skin by:
-
- Gently delivering local wound care
- Protecting skin with moisture barrier products based on zinc, silicone or dimethicone, and/or skin protectants based on cyanoacrylate.[15][16]
- Selecting non-adherent dressings that do not cause trauma upon removal.
Moisture balance
-
1CFor non-infected, healable STs, expert consensuses recommend clinicians apply a topical dressing that will maintain a moist wound bed, and protect the fragile periwound skin, with the least amount of dressing changes possible (Grade 1C)[2][5][1]
-
- Rationale: Most STs are dry, but may present as exudative wounds in some cases (e.g. ST on a limb with peripheral edema). Several experimental studies support the hypothesis that a moist wound bed increases wound healing when compared to a dry wound bed, by facilitating cell migration and matrix formation.[17][18] On the other hand, excessive moist/exudate may macerate the fragile periwound skin.
- For infected STs: regular inspection and assessment are needed. See Table 4 for dressing selection
- For non-healable/maintenance STs, if minimal or no exudate, a topical antiseptic may be used. If exudate management is needed, non-adherent dressings may be applied.
Dressing selection
The ISTAP has developed a dressing selection guide based on expert consensus, specific for patients with STs.[1][19] Table 4 incorporates updates to the original version.[1][2]
Table 4. Dressing selection guide for skin tears. Adapted from LeBlanc, 2016 [19][1]
| Type of dressing |
Indication |
Skin tear type |
Considerations |
|
Cyanoacrylate (2 octyl or n-butyl)based topical bandage (skin glue)
|
- If available, may be used to approximate the skin edges of the wound
- For type 1 ST in children, topical skin adhesive has been shown to be a faster and less painful method, with better scar management, compared with sutures or skin closure strips [20]
|
1, 2, and 3
|
- Surgical adhesive, to be used in a similar fashion as sutures, within the first 24 hours after injury.
- Skin sutures and staples are not recommended due to the fragility of the skin [2]
- Relatively expensive
|
|
Solvent free cyanoacrylate based skin protectants
|
- Offers protection from moisture and friction damage, for up to 7 days.
|
1, 2, and 3
|
- Use as a skin protectant. Chemically bonds to skin surface so cannot be removed once applied, will slough off over time
- Does not require secondary dressing
|
|
Non-adherent mesh dressings (silicone mesh dressing, gauze or rayon mesh impregnated with petrolatum or essential fatty acids)
|
Wound exudate level:
- For dry to exudative wounds
|
1, 2, and 3
|
- Maintains moisture balance for STs with different levels of exudate
- Atraumatic removal.
- A secondary dressing may be necessary.
|
|
Foam dressing
|
Wound exudate level:
- Can be suitable to light to heavy exudate, depending on product
|
2 and 3
|
- Wear time: 2 - 7 days, depending on the level of exudate
- Opt for non-adhesive/ silicone borders whenever possible to prevent periwound trauma
|
|
Hydrogel
|
Wound exudate level:
- For dry wounds. Provides moisture to dry wounds
|
2 and 3
|
- Caution with exudative wounds, as hydrogel use on these wounds may lead to periwound skin maceration
- May be used for autolytic debridement on wounds with light exudate.
- Atraumatic removal.
- A secondary dressing may be necessary.
|
|
Calcium alginate
|
Wound exudate level:
Helps control bleeding due to hemostatic properties
|
1, 2 and 3
|
- Caution if wound is dry or lightly exudative, the dressing may adhere to the wound bed.
- A secondary dressing is required.
|
|
Gelling fiber (hydrofiber)
|
Wound exudate level:
|
2 and 3
|
- No hemostatic properties,
- Caution if wound is dry or lightly exudative,, the dressing may adhere to the wound bed.
- A secondary dressing is required.
|
|
Special considerations for infected Skin tears
|
|
Methylene blue and gentian violet dressings
|
Antimicrobial action, broad spectrum efficacy including resistant organisms.
- For wound with suspected or confirmed infection
|
1, 2 and 3
|
- Non-traumatic to wound bed
- Secondary dressing required
|
|
Non-adherent dressing with ionic silver
|
Antimicrobial action, broad spectrum efficacy including resistant organisms.
- For wound with suspected or confirmed infection
|
1, 2 and 3
|
- Do not use for prolonged periods without frequentic re-evaluation,
- Contraindicated in patients with allergy to silver
- Use non-adherent secondary dressing in order to minimize the risk of further trauma.
|
|
Leptospermum honey dressings
|
Wound exudate level:
- Minimal to high levels, depends on the dressing. Underlying dressing may be gauze, alginate, foams, or other types
|
1, 2 and 3
|
- Leptospermum honey dressings provide moist wound healing and autolytic debridement [21]
|
|
Polyhexamethylene biguanide (PHMB) dressings
|
Wound exudate level:
- Minimal to high levels, depends on the dressing. Underlying dressing may be gels, non-adherent contact layers, foams and gauze dressings
|
1, 2 and 3
|
- PHMB is an effective antimicrobial. Healthcare professionals may opt to use PHMB if they deem appropriate for wound bed conditions
|
Products NOT recommended for use in Skin tears
- Iodine-based dressings: while iodine is an effective antimicrobial dressing, it also dries the wound and periwound skin. STs are usually dry, and healable STs require a moist wound bed for healing. [2]
- Film/Hydrocolloid dressings: these dressings have been reported to contribute to medical adhesive-related STs due to their strong adhesive component [2][22]
- Skin closure strips: once accepted method for ST primary closure, skin closure strips are no longer a preferred treatment option for STs. [19][23]
- Gauze: plain gauze does not secure the flap and there is increased risk of flap displacement when changing the secondary dressing, increasing the risk of skin necrosis. [1]
Dressing application/removal techniques
Tips for dressing application
The following tips may be helpful in promoting wound healing and preventing further injuries:
- Use an emollient to soften and smooth wider skin area and prevent further tears. [1]
- Select dressings according to the specific indications described in Table 4. Opt for dressings with longer wear time if feasible/appropriate.
- If a skin flap is present, start application by positioning the dressing over the pedicle of the skin flap first, then proceed towards the end of the skin flap. Mark the dressing with an arrow to indicate the correct direction of removal, along with the ST size, type and shape (Figure 1). Ensure that this is also clearly explained in the notes. [2][1]
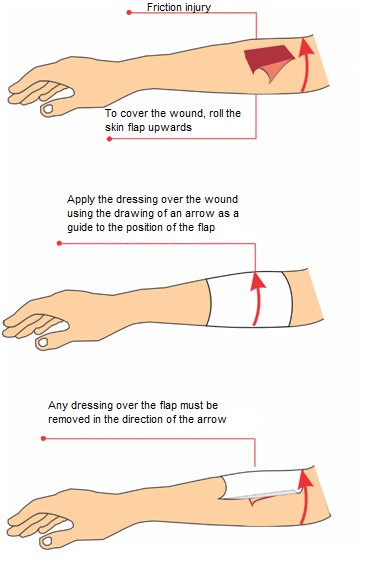
Figure 1 – Dressing application on a skin tear
Tips for dressing removal
- Evaluate the wound upon each dressing change.
- Carefully/slowly lift the dressing, so as to avoid disrupting the skin flap and damaging periwound skin. Consider using adhesive remover or saline to minimize trauma
- Cleanse the wound gently
- If the skin flap seems pale or darker, reassess the ST within 24-48 hours to determine if necrotic tissue is present and debridement is needed.
- Assess for signs of infection
- If there is no improvement after
Surgical procedures
Wound coverage with autologous skin graft
-
2CFor ISTAP type 2 STs, clinicians might opt to cover the wound bed with pieces of the ST skin flap (used as skin grafts) to promote wound healing (Grade 2C). [3][4]
-
-
Rationale: In a ISTAP type 2 ST, there is partial flap loss and the skin flap cannot be repositioned to cover the whole of the wound bed. The remaining raw area is left to heal secondarily and may result in a chronic ulcer. However, case series have shown that when pieces of the ST skin flap are placed on the wound bed as autografts, complete epithelialization of the raw area was observed in most cases after 7-10 days.[3][4] Two techniques are summarized in Table 5.
-
Coverage: authors of one of the case series suggest CPT codes 15110 or 15100, for placement of an epidermal or split-thickness skin graft.[3][4] Depending on the level of trauma, either the epidermis alone or the epidermis and a portioned layer of dermis are included in the graft.
Table 5. Techniques for autologous skin grafting for ISTAP Type 2 Skin Tears [3][4]
1. Minced Expansion Graft of Traumatic Wound Tissue [3][4]
|
|
Overview/
rationale
|
- The entire traumatic skin flap is excised at its base and minced to make grafts that are placed on the wound bed.
- In the authors’ experience, if the ST partial skin flap is simply tacked back it often fails. However, minced grafts are small and thus are more tolerant to shear and subgraft fluid collection. Furthermore, they are subject to exponential skin edge growth/epithelialization. At best, if grafts take, rapid epithelialization occurs. If not, salvaged minced tissue can still serve as an extracellular matrix analog.
|
|
Technique
|
1. Cleanse wound bed and skin flap with surgeon’s preferred wound cleanser.
2. With sharp scissors, excise the traumatized skin flap at its base
3. Transfer the tissue to the dorsum of the surgeon’s glove and repeatedly cut the tissue to create the smallest possible skin pieces
4. With forceps, place minced tissue about the wound bed .
5. Cover the wound with an interface dressing such as Adaptic (Systagenix, an Acelity Company, San Antonio, Texas) or Mepitel (Mo¨lnlycke Health Care, Norcross, Georgia).
a. Interface dressing may be secured in place with cyanoacrylate-based topical bandage (skin glue)
6. Cover interface dressing with double layer of Xeroform (Covidien [now Medtronic], Minneapolis, Minnesota).
7. Cover with gauze pads and rolled gauze.
8. Bandage may be reinforced with loosely rolled Coban Self-adherent Wrap (3M, St Paul, Minnesota).
9. Leave bandage in place for 1 week.
10. At 1-week follow-up, remove everything but the interface dressing.
a. Inspect wound.
b. If healed, remove interface dressing.
c. If islands of skin are present but wound is still healing, replace all layers above interface (repeat steps 6 to 10)
|
2. Mini Patch Grafting From The Flap Edge [3][4]
|
|
Overview/ rationale
|
Variation of the technique above. Partial skin flap is repositioned back to the wound bed. Margins of the skin flap are trimmed and made into small grafts, which are placed on the raw areas.
|
|
Technique
|
- Wash the wound carefully with saline to remove any clots and foreign objects.
- Spread out the flap to cover the defect and fix it to the surrounding skin if possible with cyanoacrylate-based topical bandage (skin glue)
- Hold the free edge of the flap with forceps and trim the edge with curved-tip scissors to get small pieces of skin, measuring approximately 1-3 mm.
- Apply the mini grafts to the skin defect with forceps, leaving free spaces of 3 to 5 mm between each of the grafts.
- Cover the wound with a non-adhesive dressing and gauze pads. Use of a silicone sheet (e.g. Adaptic, Mepitel) as an interface dressing is highly preferred to maintain a slightly moist environment.
- At 10 days follow-up, remove the gauze and dressing and inspect for healing
|
Complications
Complications include:
- Bleeding
- Infection (local, systemic, spreading)
- Delayed wound healing
Plan Reassessment
STs are acute wounds. As such, STs should close in a timely fashion (7-21 days) as long as risk factors that can delay wound healing are controlled, such as: diabetes, nutritional issues and edema.[1] If healing does not occur within 7-21 days, the patient, wound and plan should be reassessed, following the checklist below[2]:
- Check differential diagnoses
- Check if causes and co-factors affecting healing are being addressed:
-
- repeat trauma
- Comorbidities, medications that can be delaying wound healing
- Check for appropriate local wound care
If any goals are not met despite adequate diagnosis and care, more realistic goals should be set. [2]
Adjunctive Therapy
For healable STs that are refractory to adequately delivered comprehensive care, clinicians might consider adjunctive therapies. Comprehensive care includes treating the underlying causes, addressing the patient's concerns and providing adequate local wound care. Evidence supporting adjunctive therapy interventions are not sufficiently strong to justify them as primary therapy. Choice of adjuvant therapy is based on clinical findings, availability of resources, and cost-effectiveness. Patient and wound should be re-evaluated prior to initiating adjunctive therapy. (See 'Plan Reassessment' above). Of note, certain adjunctive therapy interventions might be against a patient's culture and beliefs.
Interventions that have been utilized as adjuncts in ST treatment are summarized below.
Biophysical interventions
Negative pressure wound therapy (NPWT)
-
2CFor healable ISTAP type 1 STs with bleeding that is difficult to control, or with high levels of exudate, clinicians might opt for NPWT as an adjunctive treatment to promote wound healing (Grade 2C)
-
-
Rationale: Although not a true skin graft due to the random pattern blood supply delivered through its pedicle, ST skin flaps can be treated as skin grafts. Low certainty evidence drawn from case reports supports the use of NPWT to bolster the skin flap to the wound bed and prevent shear and fluid accumulation, which can result in graft failure. Use of both traditional and disposable NPWT devices have been shown to result in successful skin flap survival and adherence to the wound bed after 5-6 days. [13][24][25]
Wound Coverage
Cellular and/or tissue based products (CTP)
- For healable but refractory STs, clinicians might consider CTPs to promote healing.
-
- There is a paucity of articles in literature supporting use of CTPs as adjunctive treatment for STs, however several CTPs are approved by the FDA to be used on traumatic wounds. It is important to note however, that Medicare only covers CTPs in the outpatient setting if the wound fails to respond to documented appropriate wound-care measures implemented for at least 4 weeks. See topic “Cellular and/or Tissue Based Products".
PREVENTION
Implementation of preventative measures should start upon first evaluation of each patient. Early recognition of individuals at risk for STs is essential for ST prevention. [5]
Prevention follows the same principles as risk factor identification [1] :
- General health
- Mobility
- Skin
-
1CFor all patients, expert consensus recommends that clinicians perform a risk assessment for skin tears upon admission, daily (or periodically per facility policy) and after identification of an episode of ST to prevent new STs (Grade 1C).[2][1][6]
- A Risk Assessment Pathway adapted from ISTAP is shown in Figure 2. For an overview of ST risk factors, see ‘Risk Factors’ in topic “Skin Tears - Introduction and Assessment”
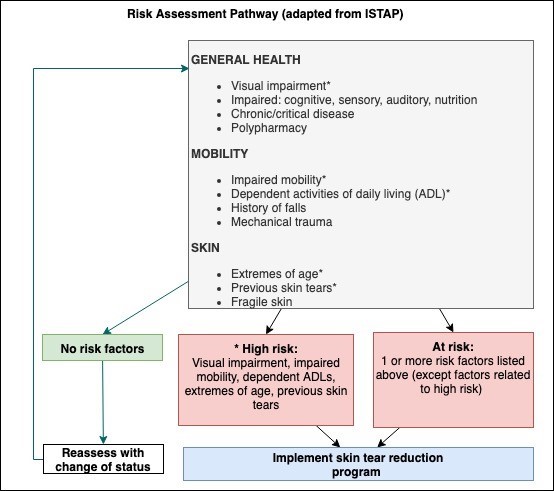
Figure 2 - Skin tear risk assessment pathway (adapted from LeBlanc et al, 2013) [5]
- 1CFor patients at risk for STs, expert consensuses recommend implementation of a ST risk reduction program. (Grade 1C)
-
Tables 1-3 above and Table 6 below summarize recommendations from the ISTAP risk reduction program and expert consensuses.
Table 6. General recommendations for skin tear risk reduction [2][5][1]
| Risk Factor |
Strategies for Risk Reduction |
|
General Health
|
- Educate patient and caregivers on self-care and strategies for ST risk reduction
- Actively involve patients and caregivers in decision making when appropriate
- Refer patient to specialists if visual, cognitive, sensory or auditory impairments are identified
- Optimize nutrition and hydration. Refer to nutritionist as needed
- If polypharmacy, consider potential side effects that may lead to STs
|
|
Mobility
|
- If impaired mobility, encourage active movement/exercises as tolerated by patient
- If patient depends on others for activities of daily living, educate caregivers on safe patient handling/equipment, including proper transferring and repositioning. For instance:
-
- Avoid sharp fingernails/ jewelry
- Avoid shear and friction (e.g. utilize bed sheets that move along with patient)
- Put on/ take off clothes carefully
- Ensure patient’s shoes are comfortable
- Implement a fall prevention program (e.g. remove clutter, ensure proper lighting)
- Pad furniture and assistive devices
- Evaluate potential skin injury by pets
|
|
Skin
|
- Keep skin moisturized with emollients at all times. Use of emollients twice a day has been shown to reduce incidence of skin tears by 50% [26]
- For skin hygiene, use warm/tepid water, soapless pH-neutral cleansers, and skin moisturizers
- Avoid use of adhesives, tapes. Use silicone tape and tubular bandages to secure dressings
- Avoid long fingernails/ jewelry
- Encourage awareness of medication-induced skin fragility
- Encourage use of protective clothing (e.g. long sleeve shirts, paints, socks, etc)
|
APPENDIX
Summary of Evidence
SOE_Negative Pressure Wound Therapy
(back to text)
- In one case report, an 8 cm long and 3.5 cm wide ST was first treated with adequate hemostasis and local wound care. However, the ST bled when skin tape was used. The ST was then covered with Mepitel™, reticulated open cell foam dressing (V.A.C. ® Granufoam™ Dressing, KCI, San Antonio, TX). NPWT ( V.A.C. ® Therapy™, KCI) was applied at - 125 mmHg for 5 days. The skin flap survived in its entirety and completely fixated to the underlying bed after 5 days. [13]
- Another case report illustrates the use of a disposable NPWT (PICO) on a highly exudative ST with a 5 cm long skin flap. Excessive exudate prevented skin tape (SteriStrip™, 3M, Maplewood, MN) and wound dressing (DuoactiveTM, ConvaTec, Flintshire, UK) from staying in place. To control the moist wound-healing environment, NPWT (PICO, Smith & Nephew) was started. Six days later, although minor erosion occurred, the skin flap adhered to the wound bed without necrosis.[24][25]