CLINICAL
Overview
This topic provides a review and updates on offloading devices for diabetic foot ulcers (DFUs), from the clinical, coverage and reimbursement perspectives. For a framework for assessment of DFUs, see Diabetic Foot Ulcer - Introduction and Assessment. For DFU management, see "Diabetic Foot Ulcer - Treatment", "Diabetic Foot Ulcer Associated with Infection - Management", "Diabetic Foot Ulcer Associated with Ischemia - Management". For DFU prevention, see "Diabetic Foot Ulcer - Prevention". For decision support on selecting offloading interventions, see "How to Select Offloading Devices".
Background
Local factors that lead to the development of calluses and diabetic foot ulcers
- The combination of foot deformity, loss of protective sensation, inadequate offloading, and minor trauma leads to tissue damage and ulceration.[2][3]
- The foot has natural bony prominences that are susceptible to pressure and friction. Typically, in healthy active individuals, excessive pressure and friction on these prominences cause painful blisters. However, in the setting of diabetic peripheral neuropathy (or neuropathy of any etiology where the sensory feedback of pain is eliminated), the pressure and friction forces will continue until they erode through the dermis. The edges of the ulcer will typically present with heavy callus buildup as a result from friction, whereas the center will typically be the deepest part of the ulcer. In contrast, pressure ulcers/injuries (also known as decubitus ulcers) are typically a result of constant pressure with or without shear (see topic "Pressure Ulcers/Injuries - Introduction and Assessment").
- Friction on feet are often caused by shear forces between the skin and a hard surface, such as the ground or a shoe. Shoes tied loosely or sandals tend to cause the foot to slip around, which increases friction stress to the foot.
- Diabetic neuropathy can cause foot deformity, most commonly hammer toes (Figure 1). Foot deformity is caused by denervation of the distal muscles of the foot first, leading to a muscle imbalance. The foot muscles stabilize the toes, so after denervation and muscle atrophy, the leg muscles will overpower the smaller foot muscles causing contracture over time, also known as an “intrinsic-minus” foot type. Hammer toes have more accentuated bony prominences, but unfortunately most regular shoes are not designed to accommodate this deformity.
- Another less common but more severe form of foot deformity is Charcot foot (Figure 2), where the foot is undergoing subluxation and fracturing. This typically happens in the midfoot, where a breakdown of the arch is known as a “rocker-bottom” deformity.
- For more information on the etiology of DFUs, see section 'Etiology' in the topic "Diabetic Foot Ulcers - Introduction and Assessment".
Fig. 1. Hammer toes | 
Fig. 2. Charcot foot |
Management of diabetic foot ulcers
- For adequate DFU management, it is essential to address the underlying causes and co-factors impeding healing, provide appropriate local wound care and address the patient's and caregiver's concerns. As seen above, DFU results from a combination of factors. Offloading is the mainstay among multiple interventions in DFU management. Of note, standard care includes not only offloading, but also optimal diabetes control, nutritional optimization, assessment of the patient’s vascular status, correction of any vascular problems, infection/ bioburden control and wound bed preparation. For more on standard care, see topic "Diabetic Foot Ulcer - Treatment".
Offloading
- Definitions:
-
- Offloading in the context of this topic refers to pressure modulation or redistribution throughout the surface of the foot and leg.[4] Common methods to offload the foot include non-weight bearing (e.g., bed rest, wheel chair, crutch-assisted gait), offloading devices, other non-surgical techniques and offloading surgical procedures.[4]
- Offloading devices are specialized products, such as casts, removable casts or specialized shoes that relieve pressure on foot ulcers to help them heal and reduce the risk of amputation. [5]
- Offloading devices for DFUs can be categorized as non-removable, removable, and other assisted devices.[1]
Offloading Principles
- There are multiple methods to offload foot ulcers, including combinations of offloading devices, dressings, and techniques. Understanding offloading principles and goals allows the clinician to develop patient-specific offloading treatment plans. In general, there are two goals in offloading:
-
- Reducing the total amount of friction
-
- The tighter the shoe or device, the less the foot slips within the shoe/device, and therefore the less friction there is against the foot.
-
- Pay attention to shoe fit and lacing habits. Shoes that are too large slip more. Laces left untied will be loose.
- Conversely, excessive tightness could lead to excessive pressure on the skin. Thus, it is important to follow up closely whenever a new offloading device is applied.
- Offloading devices that cannot be removed (non-removable) are more effective than removable devices because it forces compliance.
- Distribution of friction through an interface
-
- Slippery surfaces or lubricants (e.g. ointments or creams) are useful to reduce friction
- Multiple layers between the skin and the ground help decrease friction forces that reach the foot. Friction forces must travel from the ground to the foot, so the more layers between the ground and skin, the more likely those friction forces are dissipated amongst those layers before reaching the foot. For example, layers can include application of an ointment, a non-adherent wound dressing (e.g. Covaderm ?), moleskin (thin but heavy cotton adhesive fabric), sock, multidensity insole, and shoe. Each offloading method will be described below with its pros and cons, but the idea is to use these methods in combination to maximize efficacy.
- Micro traumas to the foot that occur when walking contribute to the development of DFUs. The easiest way to offload would be to recommend non-weight bearing. However, that is not feasible because foot ulcers tend to heal slowly, and non-weight bearing for several months is difficult to adhere to. DFUs have a high recurrence rate (ranging from 8–59% [6]), so treatment should also incorporate preventative measures.
General Indications and Contraindications
In general, offloading is indicated to heal and/or prevent DFUs. Indications that are specific to certain types of offloading devices are listed below.
Non-removable knee-high offloading devices
INDICATIONS
Non-removable knee-high offloading devices are indicated for offloading/healing neuropathic plantar forefoot ulcers in patients with diabetes that [2][3]:
- Do not have ischemia or uncontrolled infection.
- Are mildly infected, that is, under control with topical and/or oral antibiotic treatment, and with none to only small amounts of exudate.
- Have mild peripheral arterial disease (PAD), that is, PAD is present to a level where there is still potential for wound healing, as determined by non-invasive arterial exams such as ankle brachial pressure index (ABI), transcutaneous oxygen measurement (TCOM), etc. See exams and their interpretation in section ‘Peripheral Artery Disease (PAD) in patients with DFU’, in “Diabetic Foot Ulcer - Introduction and Assessment”
contraindications
Non-removable knee-high offloading devices are contraindicated for offloading neuropathic plantar forefoot ulcers if [2][3]:
- There is any doubt on the potential for wound healing (i.e., non-healing DFUs). See topic “How to Determine Healability of a Chronic Wound”.
- Both mild infection and mild PAD are present.
- The DFU is severely infected and/or severely ischemic. In those cases, infection and ischemia should first be resolved before offloading can be applied.
Possible adverse effects of non-removable knee-high devices include [2][3]:
- Ankle joint immobilization, reduced activity level, potential risk of falls, knee or hip complaints due to asymmetry in walking from the unilaterally increased sole height, and pressure ulcers due to poor casting or fitting.
Removable knee-high offloading devices
INDICATIONS
Removable knee-high offloading devices are indicated for offloading/healing neuropathic plantar forefoot ulcers in patients with diabetes that [2][3]:
- Present with contraindications to a non-removable knee-high device and can be expected to be adherent to wearing the device.
- Have heavily exuding plantar foot ulcers or ulcers with active mild infection that is not yet under control, which requires frequent local care or inspection;
- Present with mild PAD and some doubt regarding the potential for wound healing;
- Present with both mild infection that is under control and mild PAD, but the potential for healing is still present.
contraINDICATIONS
Removable knee-high offloading devices are contraindicated for offloading/healing neuropathic plantar forefoot ulcers in patients with diabetes that [2][3]:
- Have severely infected and/or severe ischemic foot ulcers. In those cases, the infection and ischemia should first be resolved before offloading can be applied.
Possible adverse effects of non-removable knee-high devices include [2][3]:
- Ankle joint immobilization, reduced activity level, potential risk of falls, or knee or hip complaints due to asymmetry in walking from the unilaterally increased sole height.
Forefoot offloading shoe, cast shoe, or custom-made temporary shoe
Indicated for offloading/ healing neuropathic plantar forefoot ulcers in patients with diabetes for whom a knee-high device is contraindicated or cannot be tolerated [2][3].
Offloading Devices - Types
Several offloading devices exist. They can be roughly divided as non-removable, removable and other assisted devices.[1] Types are listed below. For a concise glossary of offloading devices, see Table 1 in this section.
Non-removable offloading devices:
- Cast supplies:
-
- Knee-high non-removable device: total contact cast (TCC)
-
- TCC should be considered for:
- Severe foot and ankle deformities where fit into a controlled ankle movement (CAM) boot is questionable or impossible (i.e. Charcot deformity of the midfoot and ankle)
- Treatment of ulcers associated with instability, such as fractures or Charcot foot. If leg swelling is present however, TCC is contraindicated, because as the edema subsides, the cast will become loose and cause iatrogenic ulceration.
- If prescribing TCC for patients that need to climb stairs in their home, a physical therapy consult is appropriate to evaluate and improve gait and balance.
-
- TCC are considered the gold standard for offloading non-complex DFUs, however they remain vastly underutilized in practice.[2][3] Barriers to a more widespread use of non-removable knee-high devices include the need for specialized cast technicians who can apply TCCs, the relatively longer duration of the procedure to apply a TCC, and issues with Medicare reimbursement. See section 'Coding, Coverage and Reimbursement' below.
- Soft casting: also known as “football dressing” soft casting is composed of multiple layers of cast padding to the foot, followed by a layer of gauze bandage roll (e.g. Kerlix) and a layer of self-adherent wrap (e.g. Coban) (Figures 3 to 5).[7] This technique creates a snug bulky dressing with multiple layers of material between the foot and the ground, almost like a cast without the hard outer layer. It is not supposed to be removed, and is changed weekly. This is a good option for patients with knee, hip, and back problems because it does not alter their gait or limb length dramatically. It is also a good option for patients who are unable to don or doff devices. It should not be applied as tightly in the setting of vascular disease and thin fragile skin.
Fig. 3. Soft casting (football dressing), layer of gauze bandage roll | 
Fig. 4. Soft casting (football dressing), layer of self-adherent wrap | 
Fig. 5. Soft casting (football dressing) |
- Walking boot:
-
- Knee-high removable cast walker (RCW) rendered non-removable (instant TCC or iTCC)
Removable offloading devices
- Cast supplies:
-
- Ankle-high removable cast (e.g., Scotchcast Softcast™, Ransart boot) [8][9]
- Walking boot:
-
- Charcot Restraint Orthotic Walker (CROW): considered a custom fabricated ankle foot orthosis (AFO)
- Knee-high removable cast walker (RCW)
-
- Removable offloading walking boots such as CAM boots (Figure 6) are effective - as long as the patient is adherent to wearing them - because these boots keep the foot very snug and allow pressure to be redistributed from the foot and into the shin with push off. Some boots allow for exchange of the insole to an offloading insole. The boot creates a limb length discrepancy, so if the patient is to be walking in a boot as the primary mode of offloading, it is helpful to have the patient purchase a shoe leveler (e.g. EvenUP™, Procare® ShoeLift™) which is not covered by insurance, but is inexpensive. Patients with knee, hip, and back problems may not tolerate long term use of the boot due to the limb length discrepancy.
- If prescribing CAM boots for patients that need to climb stairs in their home, a physical therapy evaluation is appropriate.
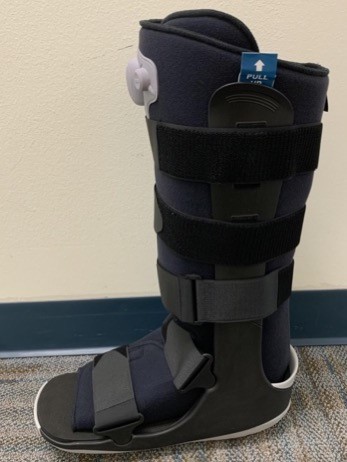
Fig. 6. Knee-high removable walker - controlled ankle movement (CAM) boot
- Shoes/ sandals
-
- Orthopedic footwear: includes orthopedic shoes, cast shoes, half-shoes (e.g. OrthoWedge™), heel-relief shoes, post-operative shoes and others [10]
- The insole of some post-operative shoes (e.g. Darco Med-Surg™) can be easily modified with a scalpel or grinder. Some insoles (e.g. PegAssist™) have pre-cut plugs that can be easily removed (Figures 7-10). The fifth metatarsal head, fifth metatarsal base, and first metatarsal head are susceptible to ulceration on the side against the straps. In that case, adjust the straps or cut a hole in it to reduce pressure. Those shoes can be effective if the patient secures the straps with each use.

Fig. 7. Post-operative shoe with removable insole
| 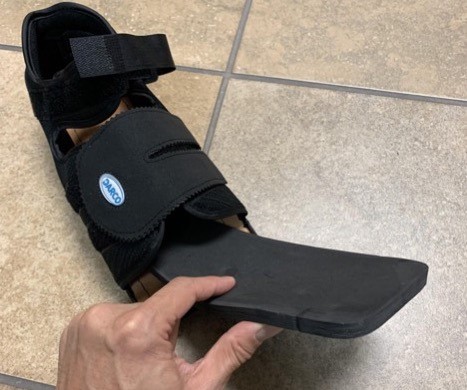
Fig. 8. Removing insole
|

Fig. 9. Insole with pre-cut plugs
| 
Fig. 10. Insole with pre-cut plugs, lateral view
|
- Therapeutic shoes (also known as 'diabetic shoes'): a depth-shoe or custom-molded shoe that can accommodate multi-density inserts.[11]
- Wearing a shoe that is stiff and does not bend easily reduces forefoot motion, and thus reduces friction to the metatarsals and toes. Some shoes have a built-in shank in the outsoles or have a rocker bottom. A carbon fiber insert can be purchased and inserted underneath the insole to provide extra rigidity.
- Multidensity inserts can be purchased or prescribed. They are created with increasing softness, with the softest layer closest to the foot. This allows for reduction of pressure and a bit of friction reduction between the layers of the insole.
Other assisted devices
- Crutches
- Wheelchair
- Walkers, rolling walkers
Glossary - Offloading devices
Table 1. Glossary for Offloading Devices. Adapted from The International Working Group on the Diabetic Foot (IWGDF) [2][3]
| Glossary for Offloading Devices (IWGDF) |
- Ankle-high offloading device: an offloading device that extends no higher up the leg than just above the ankle level. Includes ankle-high walker, forefoot offloading shoe, cast shoe, healing sandal, post- operative healing shoe.
- Cast shoe: in the U.S. the term cast shoe usually refers to a flat sandal that is worn outside of a total contact cast as a way to protect the cast.
- Conventional shoe: ‘Off-the-shelf ’ mass-produced shoe without any intended therapeutic effect.
- Custom-made insole: For the purpose of this paper, this term refers to a custom-made multi-density insole that serves to accommodate foot deformities.
- Custom-made shoe: A unique, usually handmade shoe built on a positive model of the patient’s foot to accommodate deformity and relieve pressure over at-risk sites on the plantar and dorsal surfaces of the foot.
- Customized insole: Loosely used term in American English to denote an over-the-counter orthotics or prefabricated orthotics/insoles, to which some minor modifications specific to the patient’s foot may have been made. This term is not synonymous with Custom-made insole.
- Depth-shoe: American English synonym for extra-depth shoe or depth inlay shoe. Shoe constructed with additional depth and volume in order to accommodate deformity such as claw/hammer toes and/or to allow for space for a thick insole. Usually a minimum of 5 mm (~3/ 16 in.) depth is added compared with a conventional shoe. Even greater depth is sometimes provided in shoes that are referred to as double depth or super extra-depth.
- Felted foam: A fibrous, unwoven material backed by foam with absorbing and cushioning characteristics.
- Forefoot offloading shoe: Prefabricated shoe especially designed for relieving forefoot locations on the foot. The footwear has a specific shape with a wedge design and the outsole portion missing in the forefoot. These shoes are usually worn unilaterally.
- Half-shoe: Prefabricated shoe designed to offload the forefoot. The anterior part of the shoe is cut out, leaving the heel and the midfoot as the only weight-bearing surfaces.
- Healing shoe/sandal: Specially designed sandals with insoles that reduce pressure.
- Heel-relief shoe: Shoe designed to offload the heel. The heel part is missing from the footwear, and its sole arrangement is constructed in such a way that the heel is not loaded when walking.
- Liner: American English synonym for insole.
- Non-removable (cast) walker : Same as removable (cast) boot/walker but then with a layer(s) of fiberglass cast material circumferentially wrapped around it, rendering it irremovable (also known as ‘instant total contact cast ’).
- Post-operative shoe: Prefabricated shoe with roomy and soft upper worn after an operation to the foot.
- Prefabricated insole: An ‘off-the-shelf ’ flat or contoured insole made without reference to the shape of the patient's foot.
- Ransart boot: Removable fiberglass combi-cast shoe extending to just below the ankle, existing of minimal padding and moulded to the shape of the foot with total contact of the entire plantar surface. A window is cut over the ulcer area.
- Removable (cast) boot/walker: Prefabricated removable knee-high boot with a rocker or roller outsole configuration, padded interior, and an insertable and adjustable insole, which may be total contact.
- Rocker bottom shoe: Shoe with rigid outsole designed with a sharp transition on the outsole. The shoe rocks forward in the late support phase to allow walking without extension of the metatarsal-phalangeal joints.
- Roller shoe : Shoe with rigid outsole similar to the rocker shoe, but with a contoured outsole to provide a smoother transition during walking.
- Scotch-cast boot : A removable well-padded cast cut away at the ankle. Windows are cut over the ulcers if needed. For large heel ulcers, a removable heel cap of fiberglass is added. The boot is worn with a cast sandal to increase patient mobility.
- Shoe modification: Modification to an existing shoe with an intended therapeutic effect, for example, pressure relief.
- Standard therapeutic shoe: Prefabricated form of therapeutic shoe, without any customization to the patient’s foot.
- Temporary shoe: Prefabricated shoe that is used temporarily to treat a foot ulcer.
- Therapeutic shoe: Generic term for footwear designed to allow some form of treatment to be applied to the foot that cannot be applied by or in a conventional shoe. Extra-depth shoes, custom-made shoes, and so on are all examples of therapeutic shoes.
- Toe orthosis: An in-shoe orthosis to achieve some alteration in the function of the toe.
- Total contact cast: A well-moulded, minimally padded, knee-high non-removable fiberglass or plaster cast that maintains total contact with the entire plantar surface and lower leg. The cast is often worn with an attachable sole to facilitate walking.
|
Other non-surgical offloading techniques
- Ointment followed by an adhesive bandage (e.g Mepilex™, Covaderm™) allows for a clean low friction interface. This can be augmented by adding a donut or U-shaped felt pad, fixated with a large sturdy adhesive such as moleskin to reduce pressure. This bandage can be left alone for several days and fits in shoes, which is a good option for patients that need to wear specific shoes, such as for work or formal events.
- Teaching patients to perform the heel lock lacing technique improves shoe fit and reduces friction. This involves creating a loop through the hole nearest the ankle and locking the laces across the loop as demonstrated in the images below (Figure 11).
- If tying shoe laces is a problem because of musculoskeletal problems such as arthritis in the back or hands, a steel lacing system (e.g., Boa™, UTurn™) is helpful. Elastic laces, although convenient, do not give a snug fit.
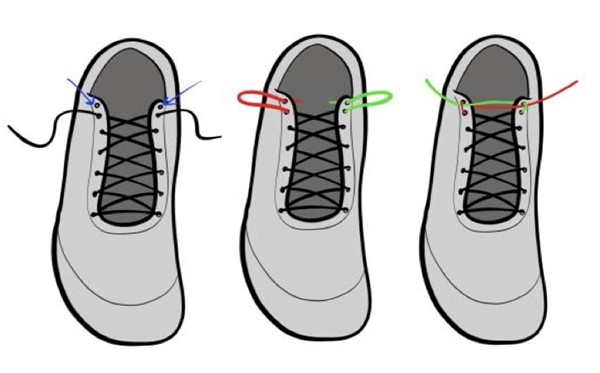
Fig. 11. Shoe lacing technique - heel lock lacing for better shoe fit
Offloading based on ulcer location
According to the author’s experience, in general for a foot ulcer that is shallow (1mm depth), 100% granular, and with minimal hyperkeratosis, clinicians could opt to initially treat without offloading, and only with debridement, ointments, and dressings. Candidates for this approach would be patients who are usually less active, but developed an ulcer because of excess activity such as attending a social event or going on a trip. Some of these ulcers will heal with little effort once the patient returns to baseline activity levels.
- A good way to screen simple ulcers from those likely to recur would be to see which ulcers improve with regular shoes and insoles. If debridements, ointments, dressings, and prescription or regular shoes are sufficient to see improvement of the foot ulcer, then we know that once healed the DFU is not likely to recur. The key is to accurately measure the ulcer, and continue with the same treatment for at least 2 weeks to observe change. The change (or lack thereof) in wound size will determine whether treatment be continued or changed. Thus, measurements should be obtained by the same clinician performing treatment in order to reduce inter-rater variability. If needed, an ideal offloading plan would include something that the patient is willing to wear indefinitely, beyond healing of the ulcer.
Ulcers with exposed tendons or joints and/or ulcers with heavy periwound hyperkeratoses should be offloaded at the first visit. The following are guidelines for initiating offloading based on ulcer location. As a reference, Figure 12 illustrates the foot anatomy, and Figure 13 shows the parts of a shoe.
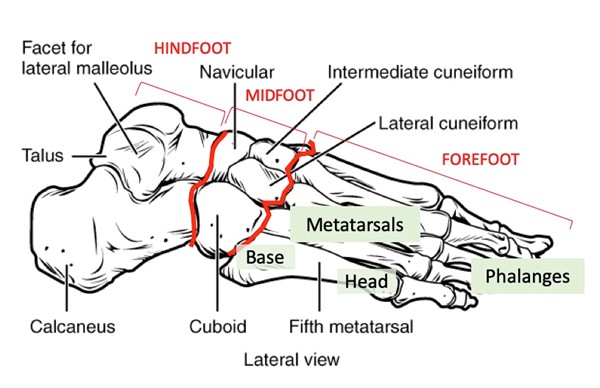
Fig. 12 Foot anatomy - bones
| 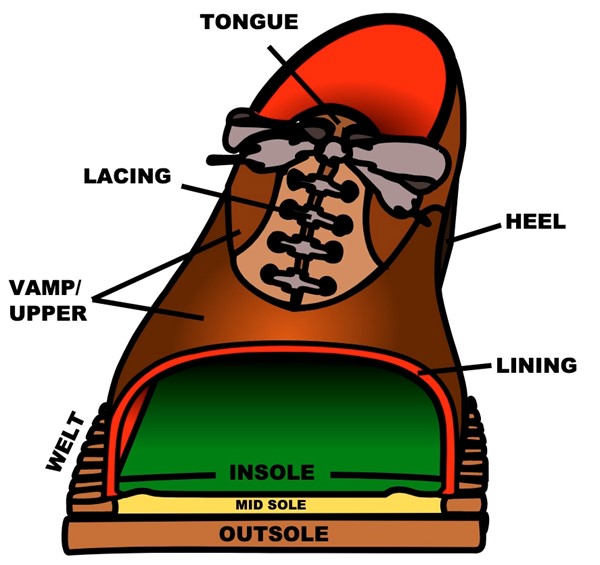
Fig. 13. Parts of a shoe
|
Forefoot ulcers
- Toe ulcers at the dorsal proximal interphalangeal joint (PIPJ) knuckle are caused by a hammer toe deformity rubbing against the vamp of the shoe. Simple surgical shoes/ sandals can be effective. Prescribe a therapeutic shoe with a Lycra ? cover on the first visit so by the time it heals, the patient will have a shoe to transition into.
- If the patient does not want to wear therapeutic shoes, clinicians might opt to cut an “X” into the area of the shoe adjacent to the ulcer. Typically the toe has worn a dent into the shoe so that it is obvious where to cut the shoe. Alternatively, patients can bring their shoes to a cobbler to have it stretched.
- Lesser toe ulcers at the distal tip can be offloaded by prescribing any therapeutic shoe with a multidensity insert. Patient education on appropriate shoe lacing is important to reduce foot slippage in the shoe (see section 'Other offloading techniques' above). If these interventions fail, an offloading shoe/sandal is appropriate. These shoes/sandals can be more effective than therapeutic shoes because the insoles are thicker and can be offloaded by removing plugs or cutting out the area of insole adjacent to the ulcer. If this fails to improve the ulcer, refer to a podiatrist for surgical options. Flexor tenotomy is a common and effective surgical procedure with little risk to the patient for lesser toe ulcers at the distal tip. See "Case: Surgical offloading of a lesser toe DFU with flexor tenotomy".
- For plantar forefoot DFUs with no ischemia or no uncontrolled infection: clinical guidelines recommend offloading with a non-removable knee-high offloading device, or with a removable knee-high or ankle-high offloading device if the former is contraindicated.[2][12][3] However, in practice a significant number of patients exists for whom such non-removable or removable knee-high/ ankle-high devices would not be appropriate. Reasons include perfusion, infection, depth of wound, drainage, mental status, balance, patient adherence, etc. For those patients, the International Working Group on the Diabetic Foot (IWGDF) suggests use of felted foam in combination with appropriate footwear to offload and heal neuropathic foot ulcers.[2][3] In practice, the use of felt for offloading DFUs is widespread.[2][13] See section 'Evidence and Recommendations'
- Hallux tip ulcers are under significantly more pressure than lesser toes as this is the last part of the body to leave the ground when pushing off during walking.
- If using a knee-high offloading device, consider applying a football dressing or prescribing a CAM boot. Football dressings are preferable over CAM boots because it is a non-removable and inexpensive offloading device.
- If a knee-high offloading device is contraindicated, not tolerated or not appropriate for the patient, clinicians might opt to initiate offloading with a surgical sandal, therapeutic shoe with a steel or carbon fiber shank, or rocker bottom shoe (e.g, Drew Lightning™).
- If the patient does not want to wear therapeutic shoes, an accommodative custom foot orthoses with Morton’s extension could be another option.
- If the patient’s insurance does not cover custom foot orthoses or cannot afford it, they may purchase a carbon fiber insert applied underneath their shoe’s factory insole. The idea is increasing rigidity to the forefoot to decrease motion and friction. For appropriate shoe lacing techniques, see section (see section 'Other offloading techniques' above)
- If the above interventions fail, refer to a podiatrist.
-
- Metatarsal head ulcers typically result from subtle foot deformity. If a knee-high offloading device is contraindicated, not tolerated or not appropriate for the patient, clinicians might opt to start with modifications to pre-existing insoles such as a metatarsal pad, which is an adhesive pad made of wool felt applied to an insole proximal to the ulcer (Figures 14 to 16). All metatarsal head ulcers can benefit from a prescription for a therapeutic shoe with a stiff shank or rocker bottom shoe. If insole modifications are ineffective, refer to a podiatrist.
-
- Ulcers located on the lateral side of the 5th metatarsal head: these ulcers are usually associated with a tailor’s bunion and shoe pressure, so attention should be paid to the vamp area of the shoe corresponding to the ulcer, similar to dorsal PIPJ toe ulcers.
- Ulcers located on the 5th metatarsal base: these ulcers can be located plantarly or laterally, or a bit of both (Figure 17).
-
- Plantar ulcers benefit from custom or over-the-counter orthotics, as the arch will take up some of the pressure from the ulcer. The same can be accomplished with felt padding to the arch area of insoles. Sometimes this can be confused with a prominent cuboid bone from Charcot foot deformity.
- Lateral ulcers in this location means the shoe is too narrow. The patient should get professionally measured and fitted.
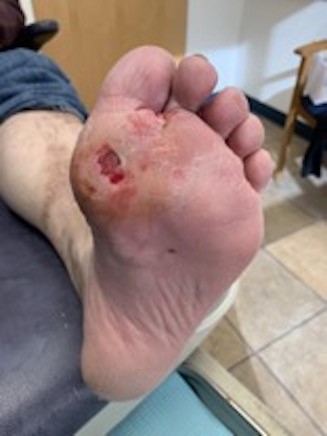
Fig. 14. Metatarsal head ulcer
| 
Fig.15. Insole of an appropriate footwear
|
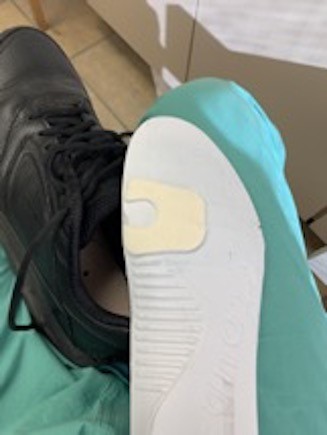
Fig. 16. Metatarsal pad (felt)
| 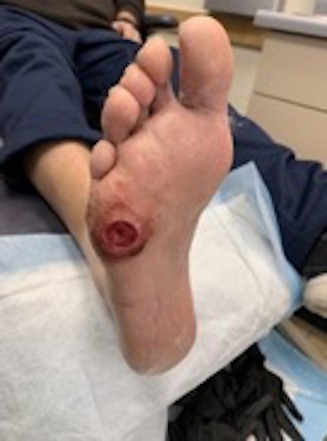
Fig. 17. 5th metatarsal base ulcer
|
Midfoot ulcers
-
- For plantar midfoot DFUs with no ischemia or no uncontrolled infection: clinical guidelines recommend offloading with a non-removable knee-high offloading device, or with a removable knee-high or ankle-high offloading device if the former is contraindicated.[2][12][3] However, in practice a significant number of patients exists for whom such non-removable or removable knee-high/ ankle-high devices would not be appropriate. Reasons include perfusion, infection, depth of wound, drainage, mental status, balance, patient adherence, etc. For those patients, the International Working Group on the Diabetic Foot (IWGDF) suggests use of felted foam in combination with appropriate footwear to offload and heal neuropathic foot ulcers.[2][3] In practice, the use of felt for offloading DFUs is widespread.[13] See section 'Evidence and Recommendations'
- Foot ulcers located in the midfoot secondary to Charcot neuroarthropathy or severe flat foot deformity are caused by breakdown of the arch through fractures, dislocation, or dysfunction of tendons and ligaments.
-
- Flat foot deformities are due to posterior tibial tendon dysfunction, not fracture or dislocation, so they are more stable than ulcers associated with Charcot foot. These ulcers can be treated with TCC, football dressing, CAM boot, CROW boot or felt pad offloading. A prescription for custom accommodative insoles and therapeutic shoes are helpful to prevent recurrence. It is important for the patient to be carefully measured because these feet are typically much wider than normal. Once the deformity is stable, an ankle-foot-orthosis (AFO) for flat foot may be prescribed.
- Charcot deformity occurs due to neuropathy in active patients, causing fracture and dislocation (Figure 2). Ulcers associated with Charcot foot will not heal if the fractures and dislocations are not stabilized, so first line of treatment typically involves a TCC, sometimes with a brief period of non-weight bearing. Alternatives include CAM boot or CROW boot (Figures 18-20).[14][15] If the foot is too swollen for a TCC or boot, first consider a compression dressing and non-weight bearing. Co-management with a podiatric or orthopedic surgeon is recommended. Once the deformity is stable and swelling minimized, a CROW boot for Charcot foot may be prescribed.
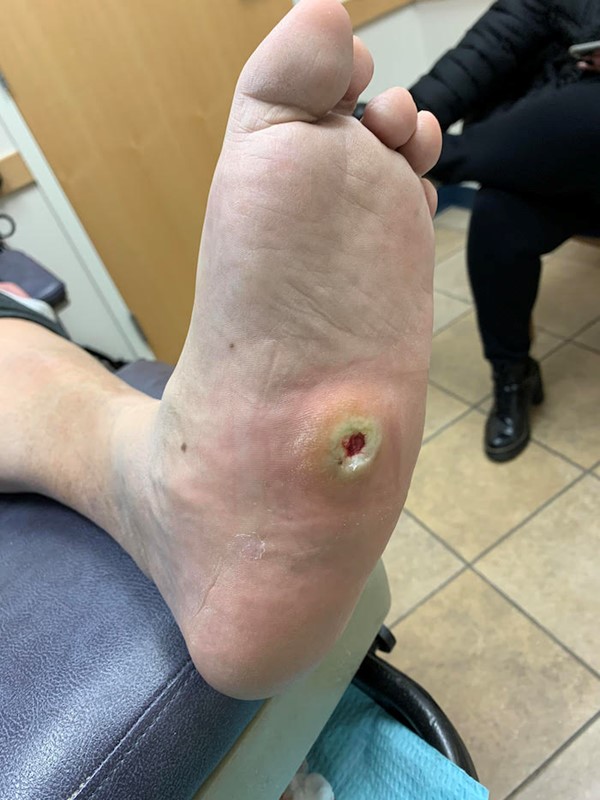
Fig. 18. Charcot foot and ulcer
| 
Fig.19. CROW boot
| 
Fig. 20. Patient wearing CROW boot |
Hindfoot ulcers
- Plantar heel ulcers are secondary to a calcaneal gait. It is common to find a functional deficit of the Achilles’ tendon such as a neglected rupture, history of Achilles’ tendon lengthening surgery, or spinal pathology involving the S1 nerve root. However, it is difficult to improve the function of an Achilles’ tendon that has been torn or cut, therefore the author typically indicates an AFO brace on the first visit.
- Posterior heel ulcers usually result from prolonged bed rest. Often these patients are not very ambulatory. For the limited ambulation that they can perform, they can wear anything they want. The focus should be on what they wear when laying in bed. Options include placing legs on a pillow or using a heel protector or suspension device that offloads the heel.[16] See heel protectors. If the patient is non-adherent to wearing these devices, a posterior splint can be made and reused with dressing changes.
Plan reassessment
The above mentioned is an appropriate first line of treatment. A normal healing rate of a foot ulcer is 1mm/week.[17][18][19] (Note: Healing rate can be assessed by accurately measuring the maximum length of the ulcer and subtracting this value from the maximum length of the ulcer measured in the prior week). If no improvement is found, it is recommended that clinicians reassess the patient, the ulcer and the treatment/ offloading plan. As for offloading, an offloading shoe, boot, weekly football dressing changes, or weekly TCC may be appropriate if no contraindications are present. These devices may be used for ulcers in any location of the foot.
- For new DFUs that failed to show adequate healing progress within 12 weeks of appropriate conservative management and offloading, consider referring to a podiatrist for evaluation of surgical offloading.[20]
Practice Tips
- Among options for knee-high offloading devices, the author’s preference is the football dressing. Patients do not need to do dressing changes, it rarely causes iatrogenic ulcers, and is appropriate for swollen legs. It does not cause limb length discrepancies so it’s easier to tolerate for those with knee, hip, or back pain. It is easy to apply such that medical assistants can be trained to apply, and requires very inexpensive materials. A multilayer dressing can also be added above the football dressing for swollen legs.
- For any ulcer that is not healing, or expanding despite optimal offloading, refer to a podiatric surgeon to evaluate for surgical offloading.
- For ulcers where a prevention plan involves some type of custom made device such as CROW boot, AFO, or custom insole, consider prescribing the device when the ulcer is about 1cm in diameter and less than 1mm deep. That way, when the ulcer heals, the device will be available for use. Consider discussing with your prosthetist their level of comfort casting molds in the presence of ulceration.
- After the ulcer heals, regular follow up visits are recommended, to make sure the callus is maintained and the epidermis matures to handle the stress and strain of weight bearing in the preventive device. With subsequent visits, clinicians can start stretching out the appointments, such as doubling the duration each visit (I.e. 1 week, 2 weeks, 4 weeks, 8 weeks). Refer the patient to establish care with a podiatrist for routine foot care. See topic “Diabetic Foot Ulcers - Prevention”.
- This document is only a guideline on the principles of offloading. These patients have different comorbidities, levels of activity, and levels of compliance and disability. As such, the optimal offloading plan can vary greatly between patients, and the clinician should be flexible to meet the patient’s needs to improve compliance. Keep in mind that the patient will have to have some form of offloading for the rest of their life to prevent recurrence.
Evidence and Recommendations
Below is a summary of the current evidence on offloading devices.[3][12] See details in section ‘Offloading’ in “Diabetes Foot Ulcer - Treatment”.
- 1BFor patients with DFU, we recommend offloading to promote faster DFU healing (Grade 1B)
Plantar forefoot or midfoot
- 1BAs the first choice to promote healing of plantar forefoot or midfoot DFUs with no ischemia or uncontrolled infection, clinical guidelines recommend offloading with a non-removable knee-high offloading device such as total contact cast (TCC) or fixed ankle knee-high walking boot rendered non-removable (instant TCC or iTCC) (Grade 1B)
- 2BTo promote healing of plantar forefoot or midfoot DFUs with no ischemia or uncontrolled infection in patients who do not tolerate non-removable knee-high offloading devices, or who require frequent dressing changes (e.g. heavy exudate, uncontrolled mild infection, mild PAD), consider offloading with a removable knee-high cast walker (RCW) instead of a removable half-shoe. (Grade 2B)
- 2CTo promote healing of plantar forefoot or midfoot DFUs with no ischemia or uncontrolled infection in patients who cannot tolerate non-removable or removable knee-high offloading devices, or when such devices are contraindicated, consider offloading with a removable ankle-high offloading device (e.g. cast shoes, half-shoes, forefoot offloading shoes, custom-made temporary shoes). (Grade 2C)
- 1BFor plantar forefoot or midfoot DFUs, clinical guidelines recommend against using conventional or standard therapeutic shoes for offloading, unless none of the above-mentioned offloading devices is available.(Grade 1B)
- 2C
If a removable/ non-removable knee-high or removable ankle-high offloading device is not available, to promote healing of plantar forefoot or midfoot DFUs with no ischemia or uncontrolled infection, consider using felted foam in combination with appropriate footwear. (Grade 2C) - 2CTo promote healing of plantar forefoot or midfoot DFUs with either mild infection or mild ischemia, consider using a non-removable knee-high offloading device (Grade 2C)
- 2CTo promote healing of plantar forefoot or midfoot DFUs with both mild infection or mild ischemia or with either moderate infection or moderate ischemia, consider using a removable knee-high offloading device (Grade 2 C)
- 2CTo promote healing of plantar forefoot or midfoot DFUs with both moderate infection and moderate ischemia, or with either severe infection or severe ischemia, primarily address the infection and/or ischemia, and then consider using a removable offloading device based on the patient’s functioning, ambulatory status and activity level (Grade 2C)
Plantar heel
- 2CTo promote healing of a plantar heel ulcer, consider using a knee-high offloading device or other offloading intervention that effectively reduces plantar pressure on the heel and is tolerated by the patient (Grade 2C)
Non-plantar
- 1CFor non-plantar DFU with no ischemia or uncontrolled infection, the IWGDF and the Society for Vascular Surgery (SVS) suggest clinicians use removable ankle-high offloading device, shoe modifications, temporary therapeutic footwear, toe spacers or orthoses depending on the type and location of the foot ulcer, to promote DFU healing (Grade 2C)
How to Select Offloading Devices
For guidance on selection of offloading devices see algorithm "How to Select Offloading Devices".
Prescribing/ Ordering
A prescription is usually required for offloading devices such as non-removable offloading devices, removable offloading devices and offloading shoes/sandals. Prescription needs to list the patient’s information, diagnosis supporting medical necessity, the exact type of offloading device being prescribed (e.g, walking boot, therapeutic shoe, orthopedic footwear, ankle foot orthosis, etc) anticipated length of need, primary goals of treatment (e.g. offload a foot ulcer, or primarily treat an orthopedic condition), instructions for use. Some DME suppliers have their own template/form to facilitate prescription. In those cases, it might be helpful to meet with the supplier to understand what types of devices they carry.
Documentation Requirements
For reimbursement and medical-legal purposes, it is important that practitioners adequately document each patient’s assessment and plan, so that medical necessity for offloading devices is supported. See coverage indications/requirements for each type of offloading device in section ‘Coding, Coverage and Reimbursement’ below.
CODING, COVERAGE AND REIMBURSEMENT
There are two sets of codes used for coding and billing of application of offloading devices:
- Current Procedural Terminology (CPT) codes, used for provider and facility fees, and
- Healthcare Common Procedure Coding System (HCPCS) Level II, used to report cost of devices (these will be used either by the DME provider who is billing the payor directly, or by facility applying the device)
Typical Medicare coverage for offloading devices is summarized below.
Non-removable offloading devices
Cast supplies - total contact cast (TCC)
- Settings: Application of total contact cast (TCC) is covered by Medicare for patients in the office and facility (hospital outpatient/ambulatory surgical center) settings.
medicare Covered indications/ requirements
- Presence of a plantar foot ulcer and/or diagnosis of Charcot [21] OR
- Diagnosis of a fracture or dislocation [21]
CPT® and HCPCS Coding
To apply a TCC, providers should use CPT 29445 and a Q code for the supplies. Application of the TCC and their materials (Q codes) should be billed to the local Medicare carrier and not the durable medical equipment (DME) Medicare Administrative Contractor (MAC).
- CPT 29445 Application of rigid total contact leg cast
- Q4037 Cast supplies, short leg cast, adult (11years+), plaster
- Q4038 Cast supplies, short leg cast, adult (11years+), fiberglass
Cast Supplies
- Cast supplies (Q codes) are always separately billable, in addition to CPT 29445.[22] However, as per Medicare Unlikely Edits (MUE), Medicare only permits for 1 supply unit to be billed per application, no matter how much material is utilized. If the Q code is rejected, practitioners are encouraged to file for a redetermination when a TCC is billed for offloading a DFU, with a diagnosis other than fracture or dislocation.[21]
CAM boot for TCC
- Some TCC manufacturers include a CAM boot in the TCC kit as well. If a provider opts to have the patient utilize the CAM boot, the boot will act as a cast shoe to provide support to the TCC, and not as an immobilizing device.[23]
-
- Off-the-shelf CAM boot [23]: whether the boot is purchased separately or included in the TCC kit, the boot should not be billed to the DME MAC as anything but a cast shoe (L3260), which is a non-covered item.
- Custom-fitted CAM boot [23]: for Medicare patients, the boot should be billed to the DME MAC with a miscellaneous HCPCS code L2999 Lower extremity orthoses, not otherwise specified, which is a non-covered item.
Frequency of replacement
- Policies may vary across MACS. For National Government Services, Inc (NGS), when billing for application of casts (CPT 29445), more than 8-10 visits for evaluation, treatment, modification and caregiver education would not be considered reasonable and necessary without significant documentation.[24]
Same visit : TCC and debridement or TCC and application of cellular and/or tissue-based products (CTP, or “skin substitutes”)
For patients covered by Medicare and by payers that abide by the Medicare The National Comprehensive Coding Initiative (NCCI):
- CPT 29445 is bundled into debridement CPT codes (e.g., 11042-11047, 97597, 97598) and grafting CPT codes (e.g., 15040-15776). Therefore, those codes shall not be reported with a casting/splinting/strapping CPT code (e.g., 29445, 29580, 29581) for the same anatomic area on the same visit. [25]
- CPT 29445 for application of TCC may be reimbursed separately if the provider appends the “59” modifier. However, modifier “59” would only be allowed if another ulcer is being treated on another site or limb where the TCC is being applied (e.g., TCC is applied on the left lower extremity and debridement is performed on the right foot) [21]
-
- Per CPT guidance, in order to use modifier “59”, "documentation must support a different session, different procedure or surgery, different site or organ system, separate incision/excision, separate lesion, or separate injury (or area of injury in extensive injuries) not ordinarily encountered or performed on the same day by the same individual."
- Note 1: Some commercial payers may reimburse both TCC and debridement performed on the same anatomical location and reported on the same visit, when modifier “59” is used. Providers should confirm directly with the commercial payer.
- Note 2 : upon first look, CPT coding guidance by “CPT Assistant” seems to conflict with reimbursement guidance provided by the NCCI Edits. According to CPT Assistant, if a wound debridement is performed (codes 11042-11047, 97597-97598) and TCC is applied on the same anatomical location during the same visit, the TCC application should be coded in addition to the debridement codes, as a “TCC is not considered a wound dressing and is not included in the debridement procedure” REF. However, it is important to highlight that while “CPT Assistant” states that these codes can be reported together, it does not instruct coders to append modifier “59” in to obtain reimbursement. Medicare and payers that abide by the NCCI Edits consider the cost of TCC bundled into that of debridement and will consider it an overpayment if the modifier is not supported by documentation (i.e., if TCC application and debridement are performed on the same anatomical site during the same visit).[26]
Cast supplies - instant total contact cast (iTCC)
An instant total contact cast (iTCC) or irremovable cast walker is a removable cast walker rendered irremovable by cohesive wrapping or casting.
CPT ® and HCPCS Coding
The same guidance provided above for TCC applies (in terms of CPT and Q codes). However, the removable cast walker (HCPCS codes L4360, L4361, L4386 and L4387 ) is not covered by Medicare when the purpose is to offload a foot ulcer.[27]
- HCPCS codes L4360, L4361, L4386 and L4387 describe an ankle-foot orthosis commonly referred to as a walking boot. Walking boots that are used to provide immobilization as treatment for an orthopedic condition or following orthopedic surgery are eligible for coverage under the Brace benefit. When walking boots are used primarily to relieve pressure, especially on the sole of the foot, or are used for patients with foot ulcers, they are non-covered - no benefit category.[27][28]
Removable offloading devices
Walking boot
Walking boots are considered ankle-foot orthoses (AFO). Their coverage (or lack thereof) is described by the Medicare Braces Benefit (Social Security Act §1861(s)).[27][29]
Medicare covered indications/ requirements
- Walking boots (L4360, L4361, L4386, L4387, L4631) are not covered when used primarily to relieve pressure, especially on the sole of the foot, or are used for patients with foot ulcers. [27][28]
- Walking boots (L4360, L4361, L4386, L4387, L4631) are covered for ambulatory beneficiaries with weakness or deformity of the foot and ankle, who [29]:
-
- Require stabilization for medical reasons, and,
- Have the potential to benefit functionally.
- Per Medicare: “HCPCS codes L4360, L4361, L4386, L4387 and L4631 describe an ankle-foot orthosis commonly referred to as a walking boot. Walking boots that are used to provide immobilization as treatment for an orthopedic condition or following orthopedic surgery are eligible for coverage under the Brace benefit. When walking boots are used primarily to relieve pressure, especially on the sole of the foot, or are used for patients with foot ulcers, they are non-covered - no benefit category”.[27][28]
- A foot pressure off-loading/ supportive device is denied as non-covered (no Medicare benefit), because it does not support a weak or deformed body member or restrict or eliminate motion in a diseased or injured part of the body.[27]
- The Charcot Restraint Orthotic Walker, also referred to as CROW boot or walker, is a custom fabricated AFO developed for patients with severe deformity of the foot and ankle due to a sensory neuropathic arthropathy, most commonly caused by diabetes. The CROW boot can be modified to accommodate changes by flaring, adding padding, and trimming where and when appropriate. No other codes may be billed for a CROW boot. [30] There is no separate billing for any modifications, fitting, or adjustments.[30] If the CROW boot is used for treatment of edema or for a lower extremity ulcer or for the prevention of ulcers and the patient does not have Charcot arthropathy, then it should be coded A9283 (non-covered).[30]
CPT ® and HCPCS Coding
- When used as a brace for the treatment of an orthopedic condition, prefabricated walking boots are coded L4360, L4361, L4386, L4387. When used as an AFO for a patient with Charcot arthropathy, CROW boot is coded L4631. However, walking boots must be coded A9283 when used solely for the prevention or treatment of a lower extremity ulcer or pressure reduction.[27]
-
- A9283: Foot pressure off loading/supportive device, any type, each
- L4360: Walking boot, pneumatic and/or vacuum, with or without joints, with or without interface material, prefabricated item that has been trimmed, bent, molded, assembled, or otherwise customized to fit a specific patient by an individual with expertise
- L4361: Walking boot, pneumatic and/or vacuum, with or without joints, with or without interface material, prefabricated, off-the-shelf
- L4386: Walking boot, non-pneumatic, with or without joints, with or without interface material, prefabricated item that has been trimmed, bent, molded, assembled, or otherwise customized to fit a specific patient by an individual with expertise
- L4387: Walking boot, non-pneumatic, with or without joints, with or without interface material, prefabricated, off-the-shelf
- L4387: Ankle foot orthosis, walking boot type, varus/valgus correction, rocker bottom, anterior tibial shell, soft interface, custom arch support, plastic or other material, includes straps and closures, custom fabricated.
- For examples of brands, go to WoundReference Product Navigator and enter the HCPCS code in the search box on the right side of the screen.
For DFU patients with concomitant weakness or deformity of the foot and ankle
- Patients with orthopedic conditions that meet the coverage requirements for walking boots (e.g., lateral ankle instability, tendinopathy of ankle, etc) may have their devices covered by Medicare.[21] In those cases, practitioners need to adequately document medical necessity, including distinct assessment and plans for the orthopedic condition and the DFU. However, it is recommended that providers confirm with their DME MACs first.
Shoes/ sandals - Orthopedic Footwear
Orthopedic footwear is covered under the leg, arm, back, and neck braces, and artificial legs, arms and eyes benefit (Social Security Act §1861(s)).[22][10]
Medicare covered indications/requirements
Shoes, inserts, and modifications are covered in limited circumstances:
- They are covered in selected beneficiaries with diabetes for the prevention or treatment of DFUs. However, different codes (A5500-A5511) are used for orthopedic footwear provided under this benefit. [10] For instance, Alice ™ by Propet Inc, and Alla™ by Pilgrim Shoes are coded A5500 when used as therapeutic shoes for diabetes, but are coded L3216 and L3217 when used as orthopedic footwear. (see DME PDAC)
- Besides the above, an orthopedic shoe and related modifications, inserts, and heel/sole replacements, are covered only when the shoe is an integral part of a brace.
- When an orthopedic footwear is used for DFU offloading, it is coded as A9283, that is, a foot pressure off-loading/ supportive device. The footwear in this case is denied as non-covered because there is no Medicare benefit category for these items.[10] For instance, these products would be coded L3260 when used for orthopedic conditions, but when used for DFU offloading, they should be coded as A9283: Darco Orthowedge Healing Shoe, Cast Boot, Economy Closed Toe Cast Boot
CPT ® and HCPCS Coding
Orthopedic footwear items are billed using HCPCS codes L3000-L3649 when not used primarily as an offloading device.
Code A9283 (foot pressure off-loading/ supportive device) is used for an orthopedic footwear item that is designed primarily to reduce pressure on the sole or heel of the foot but that does not meet the definition of [10]:
- A therapeutic shoe for diabetics or related insert or modification; or
- An orthopedic shoe or modification; or
- A walking boot for non offloading purposes
It may be a shoe-like item, an item that is used inside a shoe and may or may not extend outside the shoe, or an item that is attached to a shoe. It may be prefabricated or custom fabricated.
Shoes/ sandals - Therapeutic Shoes
Therapeutic shoes and inserts are covered under the Therapeutic Shoes for Individuals with Diabetes benefit (Social Security Act §1861(s)).[27][31]
Medicare covered indications/requirements
Therapeutic Shoes, inserts and/or modifications to Therapeutic Shoes are covered if all of the following criteria are met [31]:
- The beneficiary has diabetes mellitus; and
- The certifying physician has documented in the beneficiary's medical record one or more of the following conditions:
- Previous amputation of the other foot, or part of either foot, or
- History of previous foot ulceration of either foot, or
- History of pre-ulcerative calluses of either foot, or
- Peripheral neuropathy with evidence of callus formation of either foot, or
- Foot deformity of either foot, or
- Poor circulation in either foot; and
- The certifying physician has certified that indications (1) and (2) are met and that he/she is treating the beneficiary under a comprehensive plan of care for his/her diabetes and that the beneficiary needs diabetic shoes. For claims with dates of service on or after 01/01/2011, the certifying physician must:
- Have an in-person visit with the beneficiary during which diabetes management is addressed within 6 months prior to delivery of the shoes/inserts; and
- Sign the certification statement (refer to the Policy Specific Documentation Requirements section below) on or after the date of the in-person visit and within 3 months prior to delivery of the shoes/inserts.
- Prior to selecting the specific items that will be provided, the supplier must conduct and document an in-person evaluation of the beneficiary.
- At the time of in-person delivery to the beneficiary of the items selected, the supplier must conduct an objective assessment of the fit of the shoe and inserts and document the results. A beneficiary’s subjective statements regarding fit as the sole documentation of the in-person delivery does not meet this criterion.
If criteria 1-5 are not met, the Therapeutic Shoes, inserts and/or modifications will be denied as noncovered. When codes are billed without a KX modifier, they will be denied as noncovered.
In order to meet criterion 2, the certifying physician must either:
- Personally document one or more of criteria a – f in the medical record of an in-person visit within 6 months prior to delivery of the shoes/inserts and prior to or on the same day as signing the certification statement; or
- Obtain, initial, date (prior to signing the certification statement), and indicate agreement with information from the medical records of an in-person visit with a podiatrist, other M.D or D.O., physician assistant, nurse practitioner, or clinical nurse specialist that is within 6 months prior to delivery of the shoes/ inserts, and that documents one of more of criteria a – f.
The requirement that the in-person visit(s) be within 6 months prior to delivery of the shoes/inserts is effective for claims with dates of service on or after 1/1/2011.
Note: The certification statement is not sufficient to meet the requirement for documentation in the medical record.
Depending on the items ordered, both the evaluation and delivery could occur on the same day if the supplier had both a sufficient array of sizes and types of shoes/inserts and adequate equipment on site to provide the items that meet the beneficiary’s needs. Both components of the visit (criteria 4 and 5 above) must be clearly documented.
For claims with dates of service on or after 1/1/2011, there must be an in-person visit with the prescribing practitioner within 6 months prior to delivery of the shoes/inserts.
CPT ® and HCPCS Coding [31]
|
A5500
|
For diabetics only, fitting (including follow-up), custom preparation and supply of off-the-shelf depth-inlay shoe manufactured to accommodate multi-density insert(s), per shoe
|
|
A5501
|
For diabetics only, fitting (including follow-up), custom preparation and supply of shoe molded from cast(s) of patient's foot (custom molded shoe), per shoe
|
|
A5503
|
For diabetics only, modification (including fitting) of off-the-shelf depth-inlay shoe or custom-molded shoe with roller or rigid rocker bottom, per shoe
|
|
A5504
|
For diabetics only, modification (including fitting) of off-the-shelf depth-inlay shoe or custom-molded shoe with wedge(s), per shoe
|
|
A5505
|
For diabetics only, modification (including fitting) of off-the-shelf depth-inlay shoe or custom-molded shoe with metatarsal bar, per shoe
|
|
A5506
|
For diabetics only, modification (including fitting) of off-the-shelf depth-inlay shoe or custom-molded shoe with off-set heel(s), per shoe
|
|
A5507
|
For diabetics only, not otherwise specified modification (including fitting) of off-the-shelf depth-inlay shoe or custom-molded shoe, per shoe
|
|
A5508
|
For diabetics only, deluxe feature of off-the-shelf depth-inlay shoe or custom-molded shoe, per shoe
|
|
A5510
|
For diabetics only, direct formed, compression molded to patient's foot without external heat source, multiple-density insert(s) prefabricated, per shoe
|
|
A5512
|
For diabetics only, multiple density insert, direct formed, molded to foot after external heat source of 230 degrees fahrenheit or higher, total contact with patient's foot, including arch, base layer minimum of 1/4 inch material of shore a 35 durometer or 3/16 inch material of shore a 40 durometer (or higher), prefabricated, each
|
|
A5513
|
For diabetics only, multiple density insert, custom molded from model of patient's foot, total contact with patient's foot, including arch, base layer minimum of 3/16 inch material of shore a 35 durometer (or higher), includes arch filler and other shaping material, custom fabricated, each
|
|
A5514
|
For diabetics only, multiple density insert, made by direct carving with cam technology from a rectified cad model created from a digitized scan of the patient, total contact with patient's foot, including arch, base layer minimum of 3/16 inch material of shore a 35 durometer (or higher), includes arch filler and other shaping material, custom fabricated, each
|
Frequency of replacement
For beneficiaries meeting the coverage criteria, coverage is limited to one of the following within one calendar year (January – December):
- One pair of custom molded shoes (A5501) (which includes inserts provided with these shoes) and 2 additional pairs of inserts (A5512, A5513, or A5514); or
One pair of depth shoes (A5500) and 3 pairs of inserts (A5512, A5513, or A5514) (not including the non-customized removable inserts provided with such shoes)