ABSTRACT
Around 40% of diabetic foot ulcers (DFUs) without infection may develop infection within 1 year.[1] Infection associated with DFU is a risk factor for lower extremity amputation [2], and the risk increases significantly if peripheral arterial disease is also present.[3][4][5][2]
Management of DFU associated with infection includes standard DFU care with appropriate debridement, local wound care, and offloading, coupled with interventions to treat infection such as systemic antibiotic therapy, antimicrobial dressings, and surgical procedures when indicated (e.g, incision and drainage for abscesses). Wound culture, obtained right after debridement and before initiation of empiric antibiotic therapy, guides definitive antibiotic therapy.
This topic focuses on topical antimicrobial agents, systemic antibiotics and surgical interventions for DFUs with soft tissue infection and/or osteomyelitis. An algorithm to guide choice of empiric antibiotic therapy is provided.
|
Algorithm for Selection of Empiric Antibiotic Therapy for Diabetic Foot Ulcer Associated with Infection
|
CLINICAL
Overview
This topic focuses on topical antimicrobial agents, systemic antibiotics and surgical interventions for diabetic foot ulcers (DFUs) with soft tissue infection and/or osteomyelitis. It is important to note that management of healable infected DFUs also includes standard diabetic foot ulcer (DFU) care with appropriate debridement, offloading, wound care. For a comprehensive overview on DFU management see "Diabetic Foot Ulcer - Treatment". For assessment and diagnosis of infected DFU, see sections 'Diagnosis: Wound Cultures' and 'Diagnosis: Bone biopsy' in "Diabetic Foot Ulcer - Introduction and Assessment".
DFU with Soft Tissue Infection
Besides standard DFU care as described in the topic "Diabetic Foot Ulcer - Treatment", management of healable infected DFUs includes debridement, topical antimicrobial agents, systemic antibiotics and surgical interventions, as summarized below:
-
Debridement and removal of devitalized tissue reduce bacterial bioburden.[6]
- Wound culture, obtained right after debridement and before initiation of empiric antibiotic therapy, guides definitive antibiotic therapy. When choosing antibiotics for empiric therapy, clinicians should consider several key factors such as: likely etiologic agents, past antibiotic usage and severity of infection, history of methicillin-resistant Staphylococcus aureus (MRSA) colonization or infection, risk factors for Pseudomonas aeruginosa infection, local prevalence of antibiotic-resistant microorganisms, patient history of allergic reactions to antibiotic agents, and presence of renal or hepatic impairment.
- Surgical procedures are needed for deep abscess, compartment syndrome and virtually all necrotizing soft tissue infections.
More details on topical antimicrobial agents, systemic antibiotics and surgical interventions can be found below:
Antimicrobial topical agents
-
2CWe suggest use of antimicrobial dressings (e.g., impregnated with medical-grade honey, silver, iodine, polyhexamethylene biguanide or PHMB) for clinically infected DFUs and for DFUs that fail to show progress in healing despite standard care (Grade 2C)
-
-
Rationale: Clinical guidelines recommend against use of antimicrobial dressings to promote healing in the majority of cases with uninfected DFU.[6][3][7] Instead, topical antimicrobial agents are recommended for DFUs with high level of bacteria (>10^5 CFU/g of tissue) post debridement [6] and for non-healing wounds.[8][9] Despite these recommendations, a recent meta-analysis [10] concluded that there is low-certainty evidence (evidence level C) that more DFUs - infected or not infected - may heal when treated with an antimicrobial dressing than with a non-antimicrobial dressing (relative risk 1.28; 95% confidence interval 1.12 to 1.45). It is not clear if a specific type of antimicrobial dressing is superior to another. However, clinicians should note that evidence is still of low certainty, and use of antimicrobials may result in local adverse effects (e.g., allergy, unknown effect on wound healing) and increased risk of bacterial resistance.[7][11] Furthermore, as it relates to coverage, topical antiseptics and topical antibiotics do not have a special coverage arrangement under the Medicare part B surgical dressing benefit.[12] Also, many antimicrobial impregnated dressings have similar HCPCS codes as their non-antimicrobial impregnated counterparts, even though they are more costly to manufacture. As a result, if an antimicrobial impregnated dressing is ordered for a Medicare part B covered patient, the DME supplier might substitute it for another lower cost product with similar HCPCS.[13] When paid out-of-pocket, antimicrobial impregnated dressings are typically more expensive than their non-antimicrobial impregnated counterparts.
- For patients with signs of DFU infection, topic and systemic antibiotic therapy should be initiated promptly
Systemic antibiotics
- 1CIn the absence of infection, the IDSA/IWGDF recommend against treating wounds with antibiotics (Grade 1C)
-
-
Rationale: In the absence of infection, no evidence has been found in the literature supporting use of antibiotics to promote DFU healing or prevent infection. Furthermore, the IWGDF considers bacterial ‘bioburden’ a poorly defined concept that does not justify use of antibiotics in clinically uninfected wounds. [6][7][14][15][16]
- The IDSA/IWGDF state that patients with infected DFUs should have their ulcers cultured before initiation of antibiotics if possible (See 'Diagnosis: Wound Cultures' and 'Diagnosis: Bone biopsy' in "Diabetic Foot Ulcer - Introduction and Assessment").
-
1CFor infected DFUs, we recommend clinicians start empiric antibiotic regimen on the basis of the severity of the infection and the likely etiologic agents, and then implement definitive therapy based on culture/ sensitivity results and the patient’s clinical response to the empiric regimen (Grade 1C)
-
-
Rationale: Infected DFUs can turn into a limb and life-threatening condition, and must be treated promptly. The IDSA/IWGDF [14][7] and other clinical guidelines [6][3] recommend starting empiric antibiotic regimen after a specimen for culture is properly collected, and adjusting the regimen based on culture and sensitivity results and response to the initial course of antibiotics. Antibiotic therapy alone is insufficient, unless combined with standard wound care (debridement, offloading, moisture balance).[7]
Deciding on an empiric antibiotic regimen
-
1CWhen deciding on an empiric antibiotic regimen to treat DFU infections, consider several key factors such as: severity of infection, likely etiologic agents, past antibiotic usage, history of methicillin-resistant Staphylococcus aureus (MRSA) colonization or infection, risk factors for Pseudomonas aeruginosa infection, local prevalence of antibiotic-resistant microorganisms, patient history of allergic reactions to antibiotic agents, and presence of renal or hepatic impairment (Grade 1C).
-
- Rationale: As for choice of specific antibiotic agents for empiric therapy, low certainty evidence shows that no one antibiotic treatment is superior to others in promoting infection resolution or in having fewer side effects (evidence level C).[7][15][16][17] One RCT suggested that ertapenem with or without vancomycin is more effective in achieving clinical resolution of infection than tigecycline.[15][18] When selecting an antibiotic regimen, clinicians need to decide on antibiotic agents, route of administration and duration of treatment. An empiric regimen should virtually always include an antibiotic active against standard strains of staphylococci and streptococci. Other agents can be combined as needed, depending on the key factors above.[7][14] In the absence of a compelling reason to choose a specific antibiotic, the one with the lowest acquisition cost is preferred.[14]
- Algorithm 1 offers a framework to help clinicians consider these factors in this decision process. Table 1 lists the IDSA classification of infection severity. Table 2 details type of antibiotic, route and duration based on infection severity.
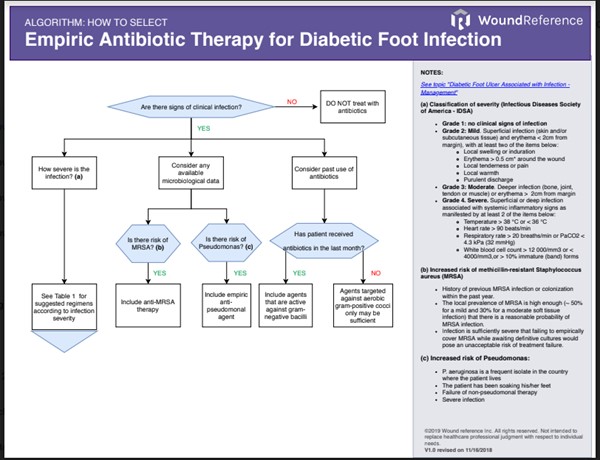
Algorithm 1. Considerations when selecting an empiric antibiotic regimen for infected DFUs (click this link to enlarge)
- Consider any available microbiological data that can help uncover the probable etiologic agent:
-
- Methicillin-resistant Staphylococcus aureus (MRSA): MRSA should be covered in any of the circumstances below:
-
- The patient has a history of previous MRSA infection or colonization within the past year.
- The local prevalence of MRSA (ie, percentage of all S. aureus clinical isolates in that locale that are methicillin resistant) is high enough (perhaps 50% for a mild and 30% for a moderate soft tissue infection) that there is a reasonable probability of MRSA infection.
- The infection is sufficiently severe that failing to empirically cover MRSA while awaiting definitive cultures would pose an unacceptable risk of treatment failure.
- Pseudomonas: P. aeruginosa should be covered in any of the circumstances below
-
- P. aeruginosa is a frequent isolate in the country where the patient lives
- The patient has been soaking his/her feet
- Failure of non-pseudomonal therapy
- Severe infection
- Consider past antibiotic usage: antibiotic usage in the month previous to the episode of infection affects etiologic agents
-
- Acute infections in patients not treated with antibiotics in the past month are most likely caused by aerobic Gram-positive cocci, especially staphylococci.
- Chronic infections or infections occurring after antibiotic treatment are often polymicrobial (aerobic Gram-negative bacilli, aerobic Gram-positive cocci, anaerobes)
- Consider infection severity:
-
- See Table 1 for classification of severity of diabetic foot infections, by the Infectious Diseases Society of America (IDSA).[7]
- See Table 2 for general guidance on type of antibiotic, route and duration of therapy according to site of infection and severity. Agents that are not listed in the table still can be used, depending on safety, clinical, availability and financial considerations.[7][16]
-
Antibiotic administration route: Mild and most moderate DFU infection can be treated with oral antibiotics. Some patients with moderate DFU infection who cannot tolerate oral antibiotics or who are infected with pathogens insensitive to available oral agents require parenteral antibiotics. Severe infection should be treated initially with parenteral antibiotics, and then with oral once the patient no longer shows systemic inflammatory response syndrome as described by the IDSA classification.[14] When available and patient’s conditions allow, clinicians might consider parenteral therapy in an outpatient setting to reduce costs.[14]
Table 1. Classification of severity of diabetic foot infections, by the Infectious Diseases Society of America (IDSA) [7]
| Clinical Criteria |
Grade/ Severity |
No clinical signs of infection
|
Grade 1/ uninfected |
|
Superficial infection (skin and/or subcutaneous tissue) and erythema < 2cm from margin), with at least two of the items below:
- Local swelling or induration
- Erythema > 0.5 cm* around the wound
- Local tenderness or pain
- Local warmth
- Purulent discharge
|
Grade 2/ mild |
Deeper infection (bone, joint, tendon or muscle) or erythema > 2cm from margin
|
Grade 3/ moderate |
|
Superficial or deep infection associated with systemic inflammatory signs as manifested by at least 2 of the items below:
- Temperature > 38 °C or < 36 °C
- Heart rate > 90 beats/min
- Respiratory rate > 20 breaths/min or PaCO2 < 4.3 kPa (32 mmHg)
- White blood cell count > 12 000/mm3 or < 4000/mm3,or > 10% immature (band) forms
|
Grade 4/ severe |
Table 2. General guidance on type of antibiotic, route and duration of therapy according to infection severity [14][7]
|
IDSA Infection Classification
|
Route and setting
|
Suggested antibiotics according to probable agents
|
Duration
|
|
Soft tissue
|
|
Grade 2. Mild
|
Topical or oral. Outpatient
|
Did the patient receive antibiotics in the past month? If did not receive, target aerobic gram-positive cocci. If received, include agents against gram-negative bacilli in regimen
- Staphylococcus aureus (MSSA); Streptococcus spp: dicloxacillin, clindamycin, cephalexin, levofloxacin, amoxacilin-clavulanate*
- Methicillin-resistant S. aureus (MRSA): Doxycycline , Trimethoprim/ sulfamethoxazole
* Relatively broad spectrum
Active against some gram-negative
|
1-2 weeks (may extend to 4 weeks if slow to resolve)
|
|
Grade 3. Moderate
|
Oral, or initiate with parenteral then switch to oral. Outpatient/inpatient
|
Did the patient receive antibiotics in the past month? If did not receive, target aerobic gram-positive cocci. If received, include agents against gram-negative bacilli in regimen
If extensive, chronic moderate infection, promptly commence therapy with a broad-spectrum regimen (activity against gram-positive cocci, common gram-negative and obligate anaerobic organisms)
- MSSA; Streptococcus spp; Enterobacteriaceae; obligate anaerobes: levofloxacin, cefoxitin, ceftriaxone, ampicillin-sulbactam, moxifloxacin*, ertapenem*, tigecycline **, levofloxacin or ciprofloxacin with clindamycin, imipenem-cilastatin**
- MRSA: Linezolid, Daptomycin, Vancomycin
- Pseudomonas aeruginosa: Piperacillin-tazobactam **
* Relatively broad spectrum
** Broad spectrum
|
1-3 weeks
|
|
Grade 4. Severe
|
Initiate with parenteral, then switch to oral when patient is systemically well and culture results are available to guide antibiotics selection. Inpatient then outpatient
|
Start with broad-spectrum empiric antibiotic therapy (activity against gram-positive cocci, common gram-negative and obligate anaerobic organisms)
Consider broad spectrum antibiotics described above under "moderate" as well
- MRSA, Enterobacteriacae, Pseudomonas, and obligate anaerobes: Vancomycin plus one of the following: ceftazidime, cefepime, piperacillintazobactam, aztreonam,bor a carbapenem
|
2-4 weeks
|
|
Bone or Joint Infection (classified by IDSA as moderate if no systemic signs, or severe if systemic signs of infection)
|
|
No residual infected tissue
|
Parenteral or oral
|
See suggested antibiotics for “moderate” or for “severe” infection cases above. Bone or joint infection is considered moderate in the absence of systemic signs of infection and severe in the presence of systemic signs of infection
|
2-5 days
|
|
Residual infected soft tissue (but not bone)
|
Parenteral or oral
|
1-3 weeks
|
|
Residual infected (but viable) bone
|
Initially parenteral, then consider switch to oral
|
4- 6 weeks
|
|
No surgery, or residual dead bone postoperatively
|
Initially parenteral, then consider switch to oral
|
6 weeks [14]
|
Switching to definitive antibiotic therapy
- Once culture and sensitivity results are available, the IDSA/IWGDF recommend a switch to narrower spectrum agents to reduce the likelihood of antibiotic resistance. However it is important to evaluate patient’s response to initial therapy and follow steps below [14][7]:
-
- If the infection responded well to the initial empiric regimen, clinicians might opt to keep the same antibiotics, even if some or all bacteria are resistant to the prescribed antibiotics.
- If infection is not responding or worsening and initial therapy does not cover isolated pathogens, therapy should be broadened to cover all isolated pathogens
- If infection is not responding or worsening despite the fact that initial therapy covers isolated pathogen:
-
- Reassess need for surgical intervention (e.g, abscess drainage)
- Reassess indication for coverage of MRSA, pseudomonas, or fastidious infecting organisms (e.g., Mycobacterium tuberculosis)
- Check patient adherence to treatment and if there is any drug interaction causing rapid metabolism or low absorption of the antibiotic.
-
Duration: Duration of antibiotic therapy for a DFI should be based on the severity of the infection, the presence or absence of bone infection, and clinical response to therapy (Table 2). Antibiotics can usually be discontinued once the clinical signs and symptoms of infection have resolved. There is no good evidence to support continuing antibiotic therapy until the wound is healed in order to either accelerate closure or prevent subsequent infection.[14][7]
Surgical procedures for soft tissue infections
- 1C In most cases of deep abscesses, compartment syndrome and virtually all necrotizing soft tissue infections, the IWGDF recommends urgent surgical intervention. (Grade 1C)
- Interventions can range from minor debridement or drainage to extensive resections, revascularization or major amputation, and should be performed by a surgeon with thorough knowledge of anatomy of the foot and its compartments [14] :
-
- For DFU with dry eschars: avoid debridement and allow for autoamputation
- Non-viable limb, affected by a potentially life-threatening infection (e.g. gas gangrene or necrotizing fasciitis), extensive soft tissue necrosis: may need amputation, but best to avoid major amputations
- Severely ischemic limb: may need revascularization
- For non-urgent infections with abscess: limit to incision and drainage. The aim of surgical treatment is to drain any deep pus and to minimize tissue necrosis by decompressing foot compartments and removing devitalized and infected tissue.
DFU with Osteomyelitis
Surgical and Medical Approaches
-
2B For patients with DFU and forefoot osteomyelitis but no ischemia or necrotizing soft tissue infections, clinicians may opt to use either primarily surgical or primarily medical strategies to treat osteomyelitis (Grade 2B).
-
2CFor patients with DFUs and osteomyelitis in any foot location, with or without comorbidities, clinicians may also opt to use either primarily surgical or primarily medical strategies (Grade 2C).
- Selection of approach should be based on several factors such as patient condition and preference, mobility, comorbidities and others (Table 3)
-
-
Rationale: While many cases of diabetic foot osteomyelitis require, or benefit from, surgical debridement or resection of bone, some can be treated successfully by medical therapy alone. Retrospective series have shown success rates with prolonged antibiotic therapy of 65-80%.[7][14][19][20] However, both interventions have pros and cons that need to be carefully weighed. Prolonged use of antibiotics leads to increased likelihood of drug-resistance, which is a risk factor for development of diabetic foot osteomyelitis.[21] Definitive surgical interventions such as as ray and transmetatarsal amputations, may risk architectural reorganization of the foot, resulting in altered biomechanics and additional cycles skin breakdown at a new high-pressure locations.[7] Moderate quality evidence (evidence level B) [22] and low quality evidence (evidence level C) [23] indicate that either intervention may be equally effective in patients with forefoot osteomyelitis, without ischemia or necrotizing soft tissue infections and in the general population of patients with diabetic foot osteomyelitis without exclusion criteria, respectively. Table 3 illustrates factors that can guide clinicians in choosing between primarily surgical or primarily medical strategies in the treatment of diabetic foot osteomyelitis. If based on these factors a primarily surgical strategy is elected and there is indication for conservative surgery (e.g. ostectomy, metatarsal head resection, distal phalangectomy, interphalangeal joint resection, metatarsal resection, metatarsal-phalangeal, joint resection, sesamoidectomy, partial calcanectomy, and exostectomy), this approach in combination with antibiotics may reduce the changes in the biomechanics of the foot and minimize the duration of antibiotic therapy.[21]
Table 3. Considerations when opting between medical or surgical approaches in the treatment of diabetic foot osteomyelitis[14]
| When to consider medical (nonsurgical) treatment |
- Patient is not a good surgical candidate (medically unstable)
- Location of infection:
-
- Not favorable for adequate postoperative mechanics (e.g., infection in midfoot or hindfoot)
- Confined to small, forefoot lesion
- No other surgical procedures on foot are needed
- Lack of resources:
-
- No skilled surgeon available
- Surgery costs are prohibitive
- Patient preference: strong preference to avoid surgery
|
| When to consider surgical treatment (bone resection) |
- Foot infection associated with substantial bone necrosis or exposed joint
- Foot appears to be functionally non salvageable
- Patient is already non ambulatory
- Patient is at high-risk for antibiotic-related problems
- Infecting pathogen resistant to antibiotics
- Limb with uncorrectable ischemia (making it difficult for systemic antibiotic delivery)
- Patient preference: strong preference for surgery
|
-
Choice of antibiotic agent: should be based on the results of a bone culture, especially because of the need for long-duration therapy [14], and evidence that culture-directed antibiotic treatment results in enhanced outcomes in osteomyelitis. [6] If empiric therapy is needed, S. aureus - the most common pathogen, should be covered. However, there might be a need to broaden coverage depending on patient’s history or previous culture results.[6] See Algorithm 1, Tables 1 and 2.
- Route of administration: If there is no residual infected tissue or the residual infected tissue is soft tissue only, antibiotics can be given orally or parenterally. Any selected oral antibiotics should have good bioavailability. If there is residual infected bone or if the patient has not undergone surgical resection or debridement, antibiotics should be initially parenteral, then switched to oral when the patient stops showing signs of systemic inflammatory response syndrome. See Table 2.
-
Duration of antibiotic therapy: Table 2 also illustrates duration of therapy as suggested by the IDSA/IWGDF. Basically, in cases treated primarily with surgical resection of the infected tissue, antibiotic therapy should last no longer than 1 week. For cases treated primarily with antibiotics and no bone surgical resection, patients should receive antibiotics for 6 weeks. This IWGDF recommendation is based on a recent randomized controlled trial that compared 6 versus 12 weeks of antibiotic therapy for nonsurgically treated diabetic foot osteomyelitis (DFO) in 40 patients found no significant difference in the remission rate (60% versus 70%). There were significantly fewer adverse effects with the shorter treatment (evidence level B).[24][14]
Adjunctive therapy for DFU with soft tissue infection or osteomyelitis
-
2CThe IDS A/IWGDF suggest against using any adjunctive treatments for DFI (Grade 2C).
-
- Rationale: More evidence is needed before these guidelines can recommend use of adjunctive therapies such as negative pressure wound therapy (NPWT), systemic hyperbaric oxygen therapy (HBOT), granulocyte colony-stimulating factors (G-CSF), larval (maggot) therapy, and platelet-derived growth factors to treat infection of DFU. While there is evidence that some of these adjunctive therapies are effective in promoting healing of DFUs, there is currently no strong evidence that can justify use of adjunctive therapies to resolve infection or osteomyelitis of DFU.[7][14][16]
APPENDIX
Summary of Evidence
|
SOE: Antimicrobial dressing
(back to text)
- Antimicrobial dressings: Low-certainty evidence (evidence level C) suggests that more DFUs - infected or not infected - may heal when treated with an antimicrobial dressing than with a non-antimicrobial dressing.
-
- A 2017 Cochrane systematic review and meta-analysis [10] included 5 RCTs (945 participants) that compared effectiveness of antimicrobial dressings and non-antimicrobial dressings in DFUs healing, and found that more wounds may heal when treated with an antimicrobial dressing than with a non-antimicrobial dressing [RR: 1.28, 95% confidence interval (1.12 to 1.45)]. Authors considered this finding as low-certainty evidence (evidence level C) due to risk of bias. It was uncertain whether antimicrobial dressings can aid in infection healing or prevent infection in DFUs (low-certainty evidence, level C). Where reported, the grade of ulcer in the studies ranged from I to III. Some studies enrolled patients with ulcers with duration longer than 30 days, others did not specify duration of the ulcer at enrollment. Three studies evaluated silver-containing dressings ( Gottrup 2013 ; He 2016 ; Jude 2007 ), one a honey-containing dressing ( Imran 2015 ), and one an iodine-containing dressing ( Jeffcoate 2009 ). Wounds were not infected at baseline in one study ( Gottrup 2013 ); mixed infected and not infected in one study ( Jude 2007 ); and not reported in the remaining three studies. Authors concluded that antimicrobial dressings probably increase the number of healing events in the medium term compared with non-antimicrobial dressings (evidence level C). However, the effect antimicrobial dressings on the incidence of infection, other outcomes, and adverse events is unclear (evidence level C)
- As for cost-effectiveness, one small, manufacturer sponsored RCT (41 participants) [25] compared treatment with a cadexomer iodine ointment to “standard treatment”, which included a gentamicin solution, in people with a grade I or II ulcer and followed them for 12 weeks. Cadexomer iodine was shown to be dominant compared to standard treatment. [26] However, according to the 2017 Cochrane review, it is uncertain whether there was a difference in the risk ratio of healing between these treatments: RR 2.16, 95% CI 0.47 to 9.88 (very low-certainty evidence - downgraded twice for imprecision and once for risk of bias)
The 2016 Wound Healing Society (WHS) [6], the 2012 Infectious Disease Society of America (IDSA) [7] and the 2016 Society for Vascular Surgery in association with American Podiatric Medical Association (SVS) [3] guidelines do not advocate for use of antimicrobial dressings for most uninfected DFU. The 2012 Wound and Ostomy and Continence Nurses Society (WOCN) [27] recommends instituting a short course of antimicrobial agent along with careful daily monitoring of the DFU for signs of infection
|
Intervention
|
SVS
|
WHS
|
IDSA
|
WOCN
|
|
Use antimicrobial dressings to treat DFU
|
“There is minimal evidence for increased rate of healing with other popular wound dressings, including honey and topical silver”
|
“Selectively use adjuvant agents (topical, device, and/or systemic) after evaluating a patient and their ulcer characteristics and when there is a lack of healing progress in response to more traditional therapies”
|
“We do not advocate using topical antimicrobials for most clinically uninfected wounds”
|
“Institute a short course of a topical antimicrobial agent along with careful daily monitoring of the neuropathic ulcer for signs of infection. Level of Evidence = B”
|
- A 2006 Cochrane review of treatment with silver-based wound dressings or topical agents for DFU [28] found no RCTs reporting outcomes on healing rates or infection resolution, and thus could not draw any conclusion related to their effectiveness on healing DFU
- A 2015 Cochrane systematic review on topical honey for treating wounds [29] concluded that it may reduce healing time for mild-to-moderate superficial and partial-thickness burns and infected postoperative wounds, but did not significantly hasten leg ulcer healing
- A 2016 systematic review of the effectiveness of interventions in the management of diabetic foot infections [17] found six studies that investigated the use of topical agents, but the methods and results did not allow the authors to draw any definitive conclusions. [10]
- A 2017 Cochrane systematic review and meta-analysis [10] included 5 RCTs (945 participants) that compared effectiveness of antimicrobial dressings and non-antimicrobial dressings in DFUs healing, and found that more wounds may heal when treated with an antimicrobial dressing than with a non-antimicrobial dressing [RR: 1.28, 95% confidence interval (1.12 to 1.45)]. Authors considered this finding as low-certainty evidence (evidence level C) due to risk of bias. It was uncertain whether antimicrobial dressings can aid in infection healing or prevent infection in DFUs (low-certainty evidence, level C). Where reported, the grade of ulcer in the studies ranged from I to III. Some studies enrolled patients with ulcers with duration longer than 30 days, others did not specify duration of the ulcer at enrollment. Three studies evaluated silver-containing dressings ( Gottrup 2013 ; He 2016 ; Jude 2007 ), one a honey-containing dressing ( Imran 2015 ), and one an iodine-containing dressing ( Jeffcoate 2009 ). Wounds were not infected at baseline in one study ( Gottrup 2013 ); mixed infected and not infected in one study ( Jude 2007 ); and not reported in the remaining three studies. Authors concluded that antimicrobial dressings probably increase the number of healing events in the medium term compared with non-antimicrobial dressings (evidence level C). However, the effect antimicrobial dressings on the incidence of infection, other outcomes, and adverse events is unclear (evidence level C). Given the weak available evidence, authors could not draw a firm conclusion on the role of any topical antimicrobial in the treatment or prevention of wound infection in people with foot ulcers and diabetes.
- As for cost-effectiveness, one small, manufacturer sponsored RCT (41 participants) published in 1996 [25] compared treatment with a cadexomer iodine ointment to “standard treatment”, which included a gentamicin solution, in people with a grade I or II ulcer and followed them for 12 weeks. Cadexomer iodine was shown to be dominant compared to standard treatment. [26] However, according to the 2017 Cochrane review, it is uncertain whether there was a difference in the risk ratio of healing between these treatments: RR 2.16, 95% CI 0.47 to 9.88 (very low-certainty evidence - downgraded twice for imprecision and once for risk of bias)
|
SOE - Systemic Antibiotics
(back to text)
The 2016 Wound Healing Society (WHS) [6], the 2012 Infectious Disease Society of America (IDSA) [7] and the 2016 Society for Vascular Surgery in association with American Podiatric Medical Association (SVS) [3] guidelines recommend implementing definite antibiotic therapy
based on results of appropriately collected culture and sensitivity testing as well as the patient’s clinical response to the empiric regimen.
|
Intervention
|
SVS
|
WHS
|
IDSA
|
|
Use empiric antibiotic regimen on the basis of the severity of the infection and the likely etiologic agents
|
Supports IDSA
|
n/a
|
Strong, low
|
|
Definitive therapy be based on the results of an appropriately obtained culture and sensitivity testing of a wound specimen as well as the patient’s clinical response to the empiric regimen
|
Supports IDSA
|
Level II
|
Strong, low
|
- A 2015 Cochrane systematic review that included 20 RCTs (3791 participants) concluded that evidence for the relative effects of different empiric systemic antibiotics for the treatment of foot infections in diabetes is very heterogeneous and generally at unclear or high risk of bias (evidence level C) due to limitations in the design of the RCTs. Therefore, it is not clear if any specific empiric systemic antibiotic treatment is better than others in resolving infection or in terms of safety. One RCT suggested that ertapenem with or without vancomycin is more effective in achieving clinical resolution of infection than tigecycline. [15]
- Low-certainty evidence (evidence level C) suggests that there is no specific systemic antibiotic treatment that is better than others in resolving infection or in terms of safety.[18]
|
SOE - Osteomyelitis
(back to text)
- Moderate certainty evidence (evidence level B) supported by one RCT [22] suggests that for DFU patients with forefoot osteomyelitis, without ischemia or necrotizing soft tissue infections, either prolonged antibiotic therapy or surgical treatment combined with a short course of antibiotics have similar outcomes in terms of healing rates, time to healing, and short-term complications. Low certainty evidence (evidence level C) supported by a retrospective observational study suggest that there is no difference in DFU healing rates in DFU patients (including patients with DFU with peripheral artery disease, hind and midfoot infections) who undergo prolonged antibiotic therapy only compared to patients who undergo surgical resection and short course of antibiotic therapy. [23]
-
- The 2012 Infectious Disease Society of America (IDSA) [7], the 2016 International Working Group on the Diabetic Foot [14] and the 2016 Wound Healing Society (WHS) [6] include recommendations regarding when to choose surgical (debridement with antibiotics) and when to choose medical (antibiotic only) interventions in the treatment of osteomyelitis, as well as duration of antibiotic therapy. The IDSA/IWGDF guidelines suggest clinicians choose either surgical or medical, depending on several factors such as patient preference, availability of skilled surgical team, and others. The WHS guideline favors surgical intervention over prolonged antibiotic therapy. As for duration of antibiotic therapy, the IDSA/IWGDF guidelines have different strength of recommendation, but both support use of antibiotics for less than a week if all the infected tissue has been removed, and 4-6 weeks of antibiotics if the patient does not undergo resection. The WHS suggests 2-4 weeks followed proper removal of infected tissue, and at least 6 weeks if there is residual infected tissue.
|
Intervention
|
IDSA
|
IWGDF
|
WHS
|
|
Surgical (debridement with antibiotic) or medical (prolonged antibiotic therapy)
|
Surgical or medical, depending on case
“Clinicians can consider using either primarily surgical or primarily medical strategies for treating diabetic foot osteomyelitis (DFO) in properly selected patients (weak, moderate).”
|
Surgical or medical, depending on case
“While many cases of DFO require, or benefit from, surgical debridement or resection of bone, some can be treated successfully by medical therapy alone”
|
Favors surgical with antibiotics
“Osteomyelitis is best treated by surgical intervention and antibiotics. However if not possible, treat with prolonged antibiotic therapy” (Level II)
|
|
Length of antibiotic therapy
|
“When a radical resection leaves no remaining infected tissue, we suggest prescribing antibiotic therapy for only a short duration (2 – 5 days) (weak, low). When there is persistent infected or necrotic bone, we suggest prolonged ( = 4 weeks) antibiotic treatment (weak, low)”
|
“6 weeks of antibiotic therapy for patients who do not undergo resection of infected bone and no more than a week of antibiotic therapy if all infected bone is resected” (Strong, moderate)
|
“When debridement has been adequate, a 2–4-week course of antibiotics is adequate. If the infected bone is not totally resected, a longer course (at least 6 weeks) is usually required.”
|
- A 2014 RCT [22] compared outcomes of the treatment of diabetic foot osteomyelitis in patients treated exclusively with antibiotics (AG) versus patients who underwent conservative surgery (SG), with follow up of 12 weeks after healing. Antibiotics were given for a period of 90 days in the AG. Patients in the SG received conservative surgery with postoperative antibiotic treatment for 10 days. Eighteen patients (75%) achieved primary healing in the AG, and 19 (86.3%) in the SG (P = 0.33). The median time to healing was 7 weeks (quartile [Q] 1 to Q5, Q3-Q8) in the AG and 6 weeks (Q1-Q3, Q3-Q9) in the SG (P = 0.72). Authors concluded that antibiotic therapy and surgical treatment had similar outcomes in terms of healing rates, time to healing, and short-term complications in patients with neuropathic forefoot ulcers complicated by osteomyelitis without ischemia or necrotizing soft tissue infections. [22]. This RCT had some limitations: osteomyelitis was diagnosed with by a validated combination of plain radiographs plus the probe-to-bone test [30], the criterion standard for this diagnosis is still bone culture and histology. Also, sample size calculations were not demonstrated and population studied was small. (evidence level B) This result cannot be extrapolated to cover DFU patients with severe or necrotizing soft-tissue infection, peripheral arterial disease and infection other than in the forefoot [31]
- A 2014 retrospective study [23] compared outcomes of patients with bone culture-proven S . aureus DFO who were treated by either ‘ medical ’ (just antibiotic therapy, other than soft tissue debridement at the bedside) or ‘ surgical ’ (operative treatment combined with prolonged antibiotic therapy). DFU healing outcomes were similar for the two groups (favourable in 80% in the surgical group and 87% in the medical group). However, patients in the medical group were less frequently hospitalized, had a shorter length of hospital stay and had more treatment related side effects
|
SOE - Adjunctive Therapy in infected DFU and osteomyelitis
(back to text)
- G-CSF: a 2013 Cochrane meta-analysis [32] included 5 RCTs (167 participants) that analyzed adjunctive therapy with G-CSF concluded that G-CSF did not appear to increase the likelihood of resolution of the infection, wound healing or duration of systemic antibiotic therapy. However, it seemed to be associated with shorter hospital stay and reduced rates of amputation, especially in patients with limb-threatening infections [16]
- Hyperbaric oxygen therapy: a 2015 Cochrane meta-analysis [33] concluded that HBOT may facilitate the healing of chronic foot ulcers, but it has no effect on promoting healing of diabetic foot infections [16]
|