Last updated on 5/27/25 | First published on 2/8/23 | Literature review current through Mar. 2026
[cite]
Authors:
Jeff Mize RRT, CHT, UHMSADS,
Tiffany Hamm BSN, RN, CWS, ACHRN, UHMSADS,
Topic editors:
Elaine Horibe Song MD, PhD, MBA,
Mike White MD, UHM, MMM, CWS,
more...
Coauthor(s)
Jeff Mize, RRT, CHT, UHMSADS
Disclosures: Nothing to disclose
Tiffany Hamm, BSN, RN, CWS, ACHRN, UHMSADS
Chief Nursing Officer, Wound Reference, IncDisclosures: Nothing to disclose
Editors
Elaine Horibe Song, MD, PhD, MBACo-Founder and Editor, Wound Reference, Inc;
Professor (Affiliate), Division of Plastic Surgery, Federal University of Sao Paulo;
Volunteer, Association for the Advancement of Wound Care;
Google Scholar Profile
Disclosures: Nothing to disclose
Mike White, MD, UHM, MMM, CWS
Disclosures: Nothing to disclose
INTRODUCTION
Background
The year 1662 marked the first documented usage of medical hyperbaric therapy: British physician Rev. Nathaniel Henshaw created the "domicilium" which consisted of a pressurized airtight chamber in which pressure could be increased using a bellows. Using the "domicilium", numerous conditions were treated with unknown results. Forlanini's pneumatic institute is an example of the use of hyperbaric therapy to treat tuberculosis (Figure 1). Of note, the first clinical use of medical hyperbaric therapy took place years before hyperbaric oxygen therapy (HBOT)-related gas laws were described. For instance, it was not until the 1670s when Robert Boyle confirmed the relationship between volume and pressure of gases. Subsequently, the actual discovery of oxygen by Joseph Priestly happened in 1774, and the development of Dalton’s and Henry’s laws of gases occurred in the early 1800s.[1][2]
Gas laws such as Boyle’s, Dalton’s and Henry’s laws have significant implications in the use of hyperbaric oxygen therapy (HBOT). As gases are compressible, they are subject to three inter-related factors: volume, pressure, and temperature, all of which are critical elements in HBOT.[1][2] Understanding gas laws in the context of HBOT is essential for enhanced safety in the hyperbaric environment. This topic provides essential information on gas laws and their role in HBOT.

Fig. 1. "Forlanini’s Pneumatic Institute"
BOYLE'S LAW
Who was Robert Boyle?
- Sir Robert Boyle was an English chemist and physicist who lived between 1627 and 1691 (Figure 2).
- In the 1670s, Boyle reported the pressure-volume law commonly called Boyle’s law, the first gas law to be described. For details, see Table 1.

Fig. 2. Sir Robert Boyle; English chemist and physicist, 1627-1691
Table 1. What history did not tell: an interesting note regarding the origins of the Boyle’s Law
About Henry Power and Richard Towneley
- Robert Boyle was not the original author of the Boyle’s law. A description of the reciprocal relation between the volume of air and its pressure appeared first in a book written by Boyle ("New Experiments Physico-Mechanical"), where he refers to it as “Mr Towneley’s hypothesis”, incorrectly crediting only Richard Towneley. [3]
- However, history has never associated the names of Henry Power and Richard Towneley with the gas law they discovered. Perhaps because the news was first given to, and first reported by, Robert Boyle.
The experiment - Henry Power and Richard Towneley original research was performed on April 27th, 1661. On that date they introduced a quantity of air above the mercury in a Torricellian tube. They measured its volume, and then using the tube as a barometer, they measured the air pressure. They then ascended Pendle Hill in Lancashire, England. Pendle Hill’s summit is 1,827 ft above mean sea level. At the summit they repeated the measurements of volume and air pressure. As expected, there was an increase in volume and a decrease in pressure. Although their measurements were believed to be roughly accurate and affected by temperature differences between the base and summit of the hill, the numerical data was sufficient to give them the intuitive realization of a reciprocal relationship between the pressure and volume of air. [3]
|
Boyle's Law
Using Hooke’s pump, Boyle and Hooke carried out experiments to investigate the properties of air and the vacuum, which resulted in the Boyle’s Law (Table 2).[4] They made their discovery using a glass tube similar to the one shown in Figure 2 above. Inside the tube, scientists used different volumes of liquid mercury to vary the pressure on a fixed weight of air. Boyle saw that pressure multiplied by volume is a constant. In other words, when one increases the pressure on a gas, the gas volume shrinks in a predictable way. This was the first gas law to be discovered. Over a hundred years were to pass before the next gas law, Charles’ Law, was discovered in 1787.[4]
Table 2. Boyle's Law
Boyle’s Law states that "the volume of a gas at constant temperature varies inversely with the pressure exerted on it”.[5] Or simply stated: When temperature is constant, pressure and volume are inversely proportional. Boyle's Law can be stated as: P1 x V1 = P2 x V2 where P is the absolute pressure of the gas, V is the volume of the gas, and variables marked 1 are the initial conditions, and variables marked 2 are the final ones
Boyle saw that pressure multiplied by volume is a constant. In other words, when one increases the pressure on a gas, the gas’ volume shrinks in a predictable way. For a discussion on Boyle's Law, see Video 1. For a demonstration on Boyle's Law, see Video 2.
|
Boyle's Law Videos
Below are a selection of curated external videos related to Boyle's Law.[6]
Video 1. Demonstration of Boyle's Law
Video 2. Discussion on Boyle's Law
The role of Boyle's Law in Hyperbaric Medicine
Boyle's Law is directly related to the situations below:
- When HBOT is used as an intervention to treat accidents and events that produce intravascular and/or other tissue gas embolization, including [7]:
- Decompression sickness
- Decompression-induced arterial gas embolism
- Iatrogenic arterial gas embolism
- Pneumotosis cystoides intestinalis (gas bubbles in the lining of the intestine)
- When the following clinical manifestations and complications occur in the hyperbaric environment:
- Ear, sinus, dental barotrauma
- Pulmonary barotrauma
- Damage to equipment with closed gas filled spaces
CHARLES' LAW (Charles and Gay-Lussac's Law)
Who was Jacques-Alexandre-César Charles?
- Jacques-Alexandre-César Charles was a French mathematician and physicist who lived between 1746 and 1823 (Figure 3).
- Charles was a pioneer balloonist who in 1787 did experiments on how the volume of gases depends on temperature. Unfortunately, Charles never published the work for which he is remembered; it was Joseph Gay-Lussac in 1808 who made definitive measurements and published results, crediting to Jacques Charles. As a result, Charles' Law is also known as "Charles and Gay-Lussac's Law" or "law of the volumes".[8] For details, see Table 3.

Fig. 3 Jacques-Alexandre-César Charles
Table 3. Whose Law is it anyways?
Charles Law: Whose Law is it anyways? - It is argued that Charles’s Law should be more appropriately attributed to Joseph Gay-Lussac.[9] Gay-Lussac correctly proposed a direct proportionality between volume and temperature. Though Charles carried out experiments on the relationship between the volume and temperature of gases before Gay-Lussac, Charles did not publish his results, perhaps because he had not taken care to dry his gases and his results were inconsistent. However, Gay-Lussac’s acknowledgement of Charles’s unpublished work led to the relationship becoming known as Charles’s Law. [9]
|
Charles' Law
As mentioned above, Gay-Lussac confirmed Jacques Charle's observation that all gases expand equally over the temperature range 0-100 °C (32-212 °F), law that became known as Charles' Law (See Table 4).[10]
Table 4. Charles' Law
Charles' Law describes the volume and temperature relationship of a gas at a constant pressure. The volume of a fixed mass of a gas at constant pressure is directly proportional to the temperature in kelvin scale.[11] See Figure 4 and Video 3 below. Charles' Law can be stated as: V 1/ T 1 = V 2 / T 2 where V is the volume of the gas, T is the temperature of the gas, and variables marked 1 are the initial conditions, and variables marked 2 are the final ones |
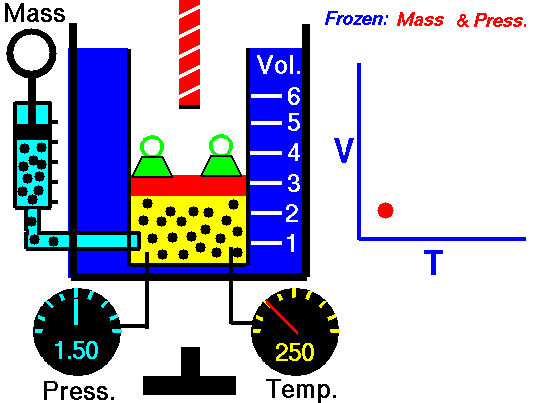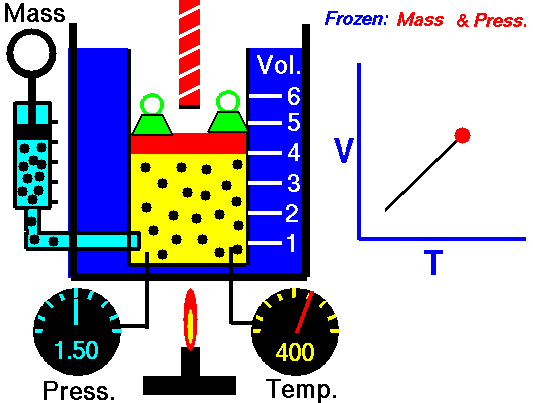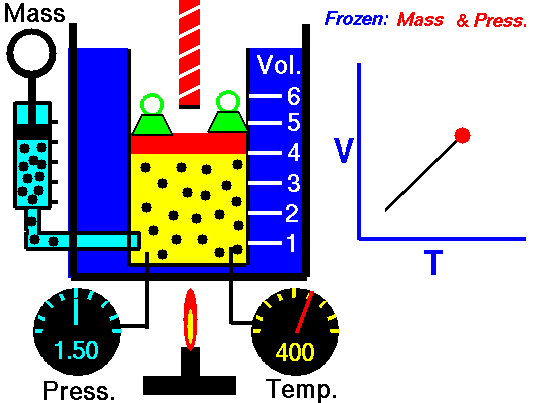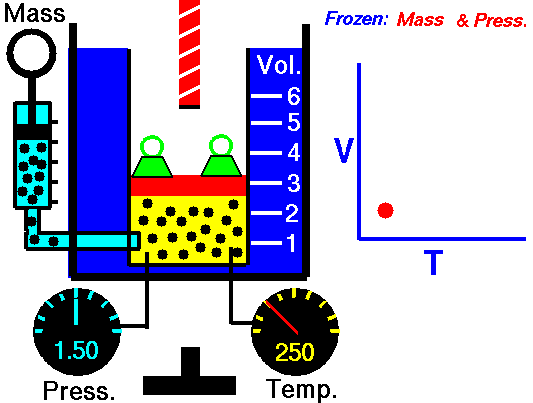
Fig. 4. Charles' Law
Video 3. Discussion on Charles' Law
GAY-LUSSAC'S LAW
Who was Joseph Louis Gay-Lussac?
- Joseph Louis Gay-Lussac was a French chemist who lived between 1778 and 1850 (Figure 5).
- Gay-Lussac is known for several discoveries, including that water is made of two parts hydrogen and one part oxygen, Charles' Law and Gay-Lussac's Law (Table 5) [8]

Fig. 5. Joseph-Louis Gay-Lussac
Gay-Lussac's Law
Table 5. Gay-Lussac’s Law
Gay-Lussac’s Law describes the pressure and temperature relationship of gases at a constant volume. The absolute pressure of a gas at constant volume is directly proportional to the temperature in Kelvin scale.[11] Gay-Lussac's Law can be stated as:
P 1 / T1 = P 2 / T 2 where P is the absolute pressure of the gas, T is the temperature of the gas, and variables marked 1 are the initial conditions, and variables marked 2 are the final ones
|
The role of Gay-Lussac’s Law in hyperbaric medicine
- In a hyperbaric chamber, as the pressure is increased there is a corresponding and proportional increase in temperature. Patients will sense this slight warming. The greater the rate of compression the higher the generated temperature. Once at treatment pressure, temperature will fall as it equilibrates with ambient surroundings. As the chamber is decompressed there is a corresponding fall in chamber temperature.[7]
DALTON'S LAW
Who was John Dalton?
- John Dalton was an English chemist, meteorologist and physicist who lived between 1766 and 1844 (Figure 6).
- In 1794, he wrote his first scientific paper which he called "Extraordinary Facts Relating to the Vision of Colours". This was the first ever paper to discuss color blindness. Dalton had realized the condition was hereditary, because he and other members of his family had it. Ultimately, Dalton’s theory for color blindness was wrong, but as he was the first person ever to research it, the condition became known as Daltonism. [12]
- In 1803, Dalton published his Law of Partial Pressures, also known as Dalton's Law [12]

Fig. 6. John Dalton, English chemist, meteorologist and physicist
Dalton's Law
The Behavior of Gases
- In 1801, Dalton gave a series of lectures in Manchester whose contents were published in 1802. In these lectures he presented research he had been carrying out on gases and liquids. This research was groundbreaking, offering great new insights into the nature of gases. Dalton stated correctly that he had no doubt that all gases could be liquefied provided their temperature was sufficiently low and pressure sufficiently high. He then stated that when its volume is held constant in a container, the pressure of a gas varies in direct proportion to its temperature.
- In 1803, Dalton published his Law of Partial Pressures (Dalton's Law), still used by every university chemistry student, which states that in a mixture of non-reacting gases, the total gas pressure is equal to the sum of the partial pressures of the individual gases (Table 6). Dalton’s work distinguished him as a scientist of the first rank, and he was invited to give lectures at the Royal Institution in London.[13]
Table 6. Dalton’s Law
According to Dalton’s Law, the total pressure exerted by a gaseous mixture is equal to the sum of the partial pressures of each individual component in a gas mixture (Figure 7 and Video 4). Dalton's Law can be stated as:
P total = p1+p2+p3… where p1, p2, p3..., represent the partial pressures of each component.[1] The pressure that gas exerts = % gas (in a decimal form) X Pressure [14] Example:
- P1 = nitrogen 78%; 0.78 ATA x 760 mmhg = 593 mmHg
- P2 = oxygen 21%; 0.21 ATA; 160 mmHg
- P3 = argon >1%; >0.01 ATA; 8 mmHg
- P total = 1.0 ATA or 760 mmHg
|

Fig. 7. Demonstration of Dalton's Law
Video 4. Gas Mixtures and Partial Pressure Discussion
The role of Dalton’s Law in hyperbaric medicine
- The process where high (hyperbaric) oxygen pressures are delivered to the lung with increases in ambient pressure.
- Refer to external video for details Dalton's Law, Henry's Law Discussion.
HENRY'S LAW
Who was William Henry?
- William Henry was an English physician and chemist who lived between 1775 and 1836 (Figure 8) [15]
- In 1803, Henry proposed what is now known as "Henry’s law" (Table 7).

Fig. 8. William Henry
Henry's Law
Henry’s Law states that at constant temperature, the amount of a gas absorbed by a liquid is directly proportional to the absolute pressure of the gas above the liquid, provided that no chemical action occurs.[16]
Table 7. Henry’s Law
According to Henry’s Law, a gas will dissolve into a liquid in direct proportion to its partial pressure. An increase in ambient pressure (partial pressure) results in more gas dissolving into the liquid portion of blood and tissues (Video 5).[1]
Henry's Law can be stated as:
p = kc where p is the partial pressure of the solute above the solution, c is the concentration of solute in the solution, and k = Henry’s Law constant |
Video 5. Henry's Law - Simple Demonstration
The Role of Henry’s Law in Hyperbaric Medicine
Hyperbaric oxygen dissolves any extra oxygen into the plasma (Henry's law). The total oxygen carried to the tissues is about 10 times more than breathing air at sea level. The increase in the oxygen concentration in the blood stream promotes the healing. Hyperbaric Oxygen therapy [1]:
- Increases the oxygen-carrying capacity of blood by increasing the oxygen concentration of the plasma.
- Henry’s law states, more gas will be dissolved in solution under higher pressures; this describes the effect of delivering more oxygen to tissues with HBOT.
- At rest, tissues need approximately 60mL/L of oxygen. At 1 ATA, plasma oxygen levels are approximately 3ml/L, however, with HBOT at 3 ATA this will increase to 60mL/L. This is in addition to the oxygen-carrying capacity of the hemoglobin present.
- HBOT will promote further diffusion of oxygen in tissues across a distance of approximately four times the normal distance. This is vital for the treatment of numerous conditions including select wounds, burns, CO poisoning, acute anemia, and ischemia.
THE IDEAL GAS LAW
The ideal gas law, also called perfect gas law, is the relation between the pressure P, volume V, and temperature T of a gas (see Table 8). The ideal gas law is a generalization containing both Boyle's Law and Charles' Law as special cases. This law can be derived from the kinetic theory of gases and relies on the assumptions that [17]:
- the gas consists of a large number of molecules, which are in random motion and obey Newton’s laws of motion,
- the volume of the molecules is negligibly small compared with the volume occupied by the gas, and
- no forces act on the molecules except during elastic collisions of negligible duration.
Although no gas has these properties, the behavior of real gases is described quite closely by the ideal gas law. A gas does not obey the equation when conditions are such that the gas, or any of the component gases in a mixture, is near its condensation point, the temperature at which it liquifies. [17]
Table 8. The Ideal Gas Law
The Ideal Gas Law can be stated as:
PV = nRT where P is absolute pressure, V is volume, n is the amount of substance, R is the ideal gas constant, and T is temperature See Videos 6 and 7 |
Video 6. Ideal Gas Law Discussion
Video 7. Ideal Gas Law Calculations
CATEGORY A CONTINUING EDUCATION CREDIT
This topic has been reviewed and approved by the National Board of Diving and Hyperbaric Medical Technology (NBDHMT) for one (1) Category A Credit.
To claim the credit:
- 1. Read the topic
- 2. Answer the examination and course critique questions.
- 3. After passing, click the provided link to access your certificate. If prompted, sign into your WoundReference account.
For more information on Category A continuing education credits see blog post " Hyperbaric Certification and Continuing Education for Technicians & Nurses".
REVISION UPDATES
| Date | Comments |
| 3/4/23 | Added section 'Category A Continuing Education Credit' |
Official reprint from WoundReference® woundreference.com ©2026 Wound Reference, Inc. All Rights Reserved
NOTE: This is a controlled document. This document is not a substitute for proper training, experience, and exercising of professional judgment. While every effort has been made to ensure the accuracy of the contents, neither the authors nor the Wound Reference, Inc. give any guarantee as to the accuracy of the information contained in them nor accept any liability, with respect to loss, damage, injury or expense arising from any such errors or omissions in the contents of the work.